Scalable Synthesis of Optically Active 3-Amino-N-Cyclopropyl-2-Hydroxyl Hexanamide for Antiviral APIs
Scalable Synthesis of Optically Active 3-Amino-N-Cyclopropyl-2-Hydroxyl Hexanamide for Antiviral APIs
The pharmaceutical industry's relentless pursuit of potent antiviral therapies has placed a premium on high-quality chiral intermediates, specifically those capable of inhibiting viral proteases with high specificity. Patent CN101279929A introduces a groundbreaking methodology for the preparation of optically active 3-amino-N-cyclopropyl-2-hydroxyl hexanamide, a pivotal building block in the synthesis of next-generation Hepatitis C inhibitors. This technical disclosure represents a significant paradigm shift from traditional amino acid derivatization, offering a route that is not only chemically elegant but also industrially robust. By leveraging a strategic combination of asymmetric epoxidation, regioselective ring-opening, and classical resolution, the inventors have solved long-standing issues regarding cost and scalability. For R&D directors and procurement specialists alike, this patent outlines a pathway to secure a reliable supply of critical antiviral intermediates while drastically simplifying the manufacturing footprint.
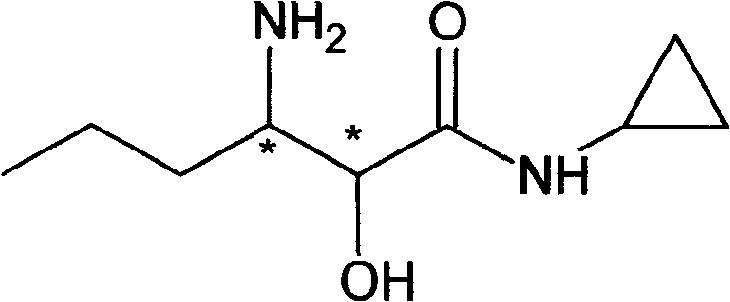
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those detailed in WO2005058821A and WO2007083620A, suffer from severe economic and operational bottlenecks that render them suboptimal for modern commercial scale-up. The most glaring deficiency lies in the reliance on expensive chiral pool starting materials like L-norvaline, the price volatility of which can destabilize long-term supply contracts and inflate the cost of goods sold (COGS). Furthermore, these legacy routes frequently employ lithium aluminum hydride (LiAlH4) for critical reduction steps, a reagent known for its pyrophoric nature and demanding requirement for strictly anhydrous conditions. The use of such hazardous reagents necessitates specialized reactor infrastructure, rigorous safety protocols, and complex quenching procedures, all of which contribute to substantial capital expenditure and extended batch cycle times. Additionally, the coupling steps often involve costly condensing agents like BOP or HOBT/EDC, generating significant stoichiometric waste that complicates downstream purification and environmental compliance.
The Novel Approach
In stark contrast, the method disclosed in CN101279929A circumvents these pitfalls by utilizing a "build-up" strategy starting from simple, achiral alkenes. The core innovation involves the epoxidation of N-cyclopropyl-trans-2-hexenamide followed by a highly regioselective ring-opening with an azide source. This approach eliminates the need for precious chiral starting materials, instead introducing chirality through a final, highly efficient resolution step using abundant organic acids like tartaric or mandelic acid. The replacement of LiAlH4 with catalytic hydrogenation or mild metal reductions (e.g., zinc powder) dramatically lowers the barrier to entry for manufacturing, allowing the process to be run in standard stainless steel reactors without exotic safety modifications. This transition from a "chiral pool" dependency to a "synthetic chiral" strategy ensures that the cost reduction in API manufacturing is structural and sustainable, rather than dependent on fluctuating raw material markets.
Mechanistic Insights into Epoxidation and Azide Ring-Opening
The chemical elegance of this synthesis lies in its precise control over regiochemistry and stereochemistry during the early stages of the sequence. The process initiates with the epoxidation of the alpha,beta-unsaturated amide using oxidants such as hydrogen peroxide or peracetic acid, often catalyzed by sodium tungstate. This step converts the planar alkene into a strained oxirane ring, setting the stage for nucleophilic attack. Subsequently, the epoxide undergoes ring-opening with sodium azide in the presence of a Lewis acid promoter like magnesium sulfate or aluminum chloride. The Lewis acid coordinates with the epoxide oxygen, activating the adjacent carbons for nucleophilic attack by the azide ion. Crucially, the reaction conditions are tuned to favor attack at the beta-position relative to the carbonyl, ensuring the formation of the 3-azido-2-hydroxy motif required for the final pharmacophore. This regioselectivity is paramount, as any deviation would lead to isomeric impurities that are difficult to remove later in the synthesis.
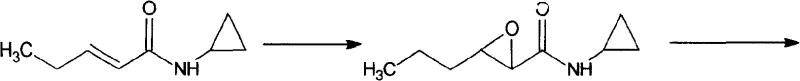
Following the installation of the azide functionality, the intermediate is subjected to reduction to reveal the primary amine. This transformation can be achieved via catalytic hydrogenation over palladium on carbon or Raney nickel, or alternatively through chemical reduction using zinc powder in ammonium chloride. The choice of reduction method offers flexibility depending on the available plant infrastructure; hydrogenation provides a clean atom-economical route, while zinc reduction offers a non-pressure alternative. The resulting racemic amino-alcohol is then subjected to diastereomeric salt formation. By reacting the racemate with a single enantiomer of a chiral acid, such as L-tartaric acid, the (S,S)-enantiomer preferentially crystallizes out of solution due to differences in lattice energy and solubility. This classical resolution technique, when optimized with specific solvent systems like methanol-water or acetone, consistently delivers product with an enantiomeric excess (ee) exceeding 99.6%, meeting the rigorous purity standards demanded by regulatory agencies for antiviral drug substances.
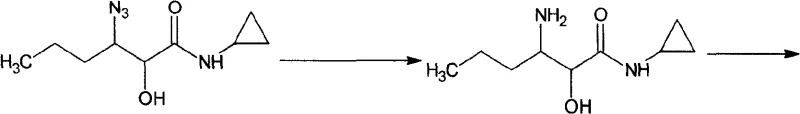
How to Synthesize 3-Amino-N-Cyclopropyl-2-Hydroxyl Hexanamide Efficiently
The synthesis protocol described in the patent is designed for operational simplicity, avoiding cryogenic temperatures and ultra-high vacuum conditions that often plague fine chemical manufacturing. The process flows logically from the inexpensive alkene precursor through oxidation, functionalization, and finally purification. Each step has been optimized in the examples to maximize yield while minimizing the formation of byproducts, ensuring a robust process suitable for technology transfer. For process chemists looking to implement this route, the key parameters to monitor include the pH control during epoxidation to prevent hydrolysis of the amide bond, and the temperature control during resolution to maximize the recovery of the desired enantiomer. The detailed standardized synthesis steps see the guide below.
- Epoxidation of N-cyclopropyl-trans-2-hexenamide using hydrogen peroxide or peracids with a tungstate catalyst to form the epoxy intermediate.
- Regioselective ring-opening of the epoxide using sodium azide and a Lewis acid (e.g., magnesium sulfate) to generate the azido-alcohol.
- Catalytic hydrogenation or metal reduction of the azide group to yield the racemic amino-alcohol intermediate.
- Optical resolution of the racemic amine using chiral acids like L-tartaric acid or L-mandelic acid to isolate the desired (S,S) enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic advantages that extend beyond mere technical feasibility. The primary value driver is the decoupling of production costs from volatile chiral feedstock markets. By starting with commodity chemicals, the manufacturer gains significant leverage in negotiating raw material prices, leading to substantial cost savings over the lifecycle of the product. Furthermore, the elimination of hazardous reagents like lithium aluminum hydride reduces the complexity of waste disposal and safety compliance, directly lowering the overhead associated with environmental health and safety (EHS) management. This streamlined operational profile translates into a more resilient supply chain, capable of withstanding market shocks and regulatory scrutiny.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the substitution of expensive stoichiometric reagents with catalytic systems and cheap inorganic salts. The removal of lithium aluminum hydride not only saves on reagent costs but also eliminates the need for specialized dry solvents and inert atmosphere equipment, which are capital-intensive to maintain. Additionally, the use of classical resolution with recycled mother liquors allows for the recovery of the unwanted enantiomer, potentially improving the overall mass balance and further driving down the effective cost per kilogram of the active isomer. These factors combine to create a highly competitive cost structure that is essential for generic API production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the scarcity of specialized chiral building blocks. This method mitigates that risk by relying on N-cyclopropyl-trans-2-hexenamide, a derivative of widely available hexenoic acid. The robustness of the reaction conditions—operating effectively at ambient or moderate temperatures—means that production is less susceptible to disruptions caused by utility failures or equipment limitations. The ability to source raw materials from multiple global suppliers ensures that the supply chain remains agile and responsive to demand fluctuations, securing the continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns with the principles of green chemistry by minimizing the use of toxic heavy metals and reducing the generation of hazardous waste streams. The aqueous workups and standard organic solvents employed are easily managed in existing wastewater treatment facilities, facilitating rapid regulatory approval for new manufacturing sites. The scalability is proven by the linear nature of the reaction steps, which do not exhibit exothermic runaway risks typical of hydride reductions, allowing for safe scale-up from pilot plant to multi-ton commercial production without significant process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, purity, and scalability concerns that often arise during vendor qualification.
Q: How does this new route improve safety compared to traditional lithium aluminum hydride methods?
A: The conventional route relies on lithium aluminum hydride (LiAlH4), a hazardous pyrophoric reagent requiring strict anhydrous conditions and specialized handling equipment. The novel method utilizes catalytic hydrogenation or mild metal reductions (like zinc powder) for the azide-to-amine conversion, significantly lowering operational risks and eliminating the need for expensive moisture-free infrastructure.
Q: What optical purity can be achieved with this resolution strategy?
A: By employing chiral resolving agents such as L-tartaric acid, L-mandelic acid, or camphorsulfonic acid, the process consistently achieves an enantiomeric excess (ee) value greater than 99.6%. This high level of stereochemical control is critical for meeting the stringent impurity profiles required for antiviral active pharmaceutical ingredients.
Q: Are the starting materials for this synthesis readily available for large-scale production?
A: Yes, the synthesis begins with N-cyclopropyl-trans-2-hexenamide, which is derived from simple, commodity-grade trans-hexenoic acid derivatives. This contrasts sharply with prior art methods that depend on expensive, non-commodity chiral pools like L-norvaline, ensuring a more robust and cost-effective supply chain for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-N-Cyclopropyl-2-Hydroxyl Hexanamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral therapies depends on a partner who can deliver both technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-amino-N-cyclopropyl-2-hydroxyl hexanamide meets the highest international standards. Our facility is equipped to handle the specific unit operations required by this patent, including safe azide handling and precise chiral resolution, providing you with a dependable source for this critical intermediate.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring a partnership built on transparency, quality, and mutual success.
