Advanced Pd-Catalyzed Synthesis of Multi-substituted Alkenyl Cyanides for Commercial Pharmaceutical Applications
The chemical industry is constantly seeking more efficient pathways to access high-value building blocks, and the recent disclosure in patent CN114539097B presents a significant breakthrough in the synthesis of multi-substituted alkenyl cyanides. These compounds serve as ubiquitous scaffolds in natural products, pharmaceuticals, and agrochemicals, acting as versatile precursors for amines, amides, and carboxylic acids. Traditionally, accessing these structures has been fraught with challenges regarding reagent stability and harsh reaction environments. However, this novel methodology utilizes easily prepared polysubstituted alkenyl sulfonium salts as key starting materials, reacting them with cuprous cyanide under mild alkaline conditions. By leveraging a robust palladium-catalyzed system, this approach not only simplifies the operational workflow but also dramatically expands the functional group tolerance, offering a reliable pharmaceutical intermediate supplier with a distinct competitive edge in producing complex molecular architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl cyanides has relied on a limited set of methodologies that often impose severe restrictions on process scalability and substrate compatibility. Classical approaches, such as the reaction of allyl halides with potassium cyanide, frequently require hazardous reagents and generate significant waste streams, complicating environmental compliance. Furthermore, conjugate addition reactions involving alkynyl nitriles and organic Grignard reagents demand strictly anhydrous conditions and cryogenic temperatures, which drastically increase energy consumption and operational costs. Other reported methods utilizing nickel or cobalt co-catalysts for alkyne hydrocyanation introduce concerns regarding heavy metal toxicity and the difficulty of removing trace metal residues from the final active pharmaceutical ingredients. Additionally, copper trifluoroacetate catalyzed condensation reactions often suffer from narrow substrate scopes and poor stability of the required alpha-iminonitrile precursors, limiting their utility in diverse medicinal chemistry campaigns.
The Novel Approach
In stark contrast to these legacy techniques, the innovative strategy outlined in the patent employs polysubstituted alkenyl sulfonium salts as stable and structurally diverse electrophiles. These sulfonium salts can be readily synthesized from common olefins and sulfoxides, providing a modular entry point for introducing various substituents at the alpha-position of the nitrile group. 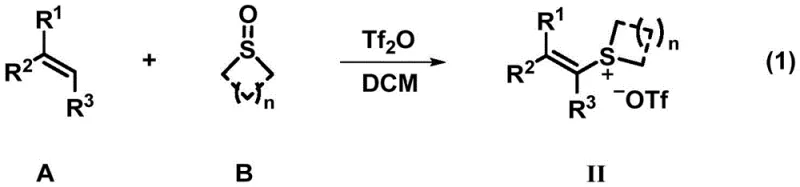 The core transformation involves a palladium-catalyzed cross-coupling with cuprous cyanide, which proceeds under significantly milder thermal conditions compared to traditional nucleophilic substitutions. This shift in synthetic logic allows for the preservation of sensitive functional groups that would otherwise decompose under the harsh basicity or high temperatures of conventional methods. By utilizing cheap and commercially available cuprous cyanide as the cyanide source, the process achieves high atom economy while avoiding the use of volatile and toxic hydrogen cyanide gas, thereby enhancing workplace safety and reducing the burden on specialized containment infrastructure required for industrial manufacturing.
The core transformation involves a palladium-catalyzed cross-coupling with cuprous cyanide, which proceeds under significantly milder thermal conditions compared to traditional nucleophilic substitutions. This shift in synthetic logic allows for the preservation of sensitive functional groups that would otherwise decompose under the harsh basicity or high temperatures of conventional methods. By utilizing cheap and commercially available cuprous cyanide as the cyanide source, the process achieves high atom economy while avoiding the use of volatile and toxic hydrogen cyanide gas, thereby enhancing workplace safety and reducing the burden on specialized containment infrastructure required for industrial manufacturing.
Mechanistic Insights into Pd-Catalyzed Cyanation of Sulfonium Salts
The success of this transformation hinges on the precise interplay between the palladium catalyst, the bulky phosphine ligand, and the sulfonium leaving group. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-sulfur bond of the alkenyl sulfonium salt, generating a cationic organopalladium intermediate. This step is facilitated by the excellent leaving group ability of the sulfonium moiety, which drives the equilibrium forward even at moderate temperatures. Subsequent transmetallation with the copper-cyanide species introduces the nitrile functionality onto the metal center. The choice of ligand is critical here; bulky electron-rich ligands like X-PHOS stabilize the active catalytic species and prevent the formation of inactive palladium black, ensuring sustained turnover numbers throughout the reaction cycle. Finally, reductive elimination releases the desired multi-substituted alkenyl cyanide product and regenerates the palladium(0) catalyst to close the loop.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy pathways. The concerted nature of the reductive elimination step ensures high stereoselectivity, preserving the geometric integrity of the double bond which is crucial for the biological activity of many downstream drug candidates. Furthermore, the use of a well-defined catalytic cycle minimizes the formation of homocoupling byproducts or polymerization side reactions that often plague free-radical cyanation attempts. The mild alkaline conditions provided by potassium tert-butoxide are sufficient to activate the cyanide source without promoting the hydrolysis of the nitrile group to the corresponding amide or acid, a common degradation pathway in less optimized processes. This level of control over the reaction trajectory translates directly into simpler purification protocols and higher overall yields for the final isolated material.
How to Synthesize Multi-substituted Alkenyl Cyanides Efficiently
To implement this robust synthetic route in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding reagent stoichiometry and atmospheric control to maximize efficiency. The process begins with the preparation of the alkenyl sulfonium salt precursor, followed by the key cyanation step where precise temperature management is essential for optimal conversion. While the general procedure is straightforward, attention to detail regarding the exclusion of moisture and oxygen is paramount to maintain catalyst activity. For a comprehensive guide on the exact operational steps, please refer to the standardized protocol below which details the mixing order, heating profiles, and workup procedures necessary to achieve the high purity standards required for pharmaceutical applications.
- Prepare the reaction vessel under nitrogen atmosphere and add alkenyl sulfonium salt, cuprous cyanide, palladium acetate catalyst, potassium tert-butoxide base, and X-PHOS ligand.
- Add anhydrous DMF solvent to the mixture and heat the reaction system to 80°C while stirring for 12 to 24 hours to ensure complete conversion.
- Filter the cooled reaction mixture through diatomite, remove volatiles under reduced pressure, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology represents a strategic opportunity to optimize sourcing costs and mitigate supply risks associated with complex intermediates. The reliance on stable, shelf-stable sulfonium salts eliminates the need for just-in-time delivery of sensitive reagents that degrade rapidly, thus allowing for larger batch sizes and more flexible inventory management. Moreover, the use of commodity chemicals like cuprous cyanide and palladium acetate ensures that raw material costs remain predictable and insulated from the volatility often seen with exotic organometallic reagents. This stability in the supply chain is further reinforced by the method's tolerance to a wide range of substrates, meaning a single manufacturing line can be adapted to produce a diverse portfolio of cyanide derivatives without extensive retooling or process redevelopment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and hazardous reagents typically required for cyanation. By replacing toxic hydrogen cyanide gas or unstable organometallic cyanides with solid cuprous cyanide, the facility avoids the capital expenditure associated with specialized gas handling systems and scrubbers. Additionally, the high catalytic efficiency means that precious metal loading can be kept low, significantly reducing the cost of goods sold per kilogram of product. The simplified workup procedure, which often requires only filtration and standard chromatography, reduces solvent consumption and labor hours, leading to substantial cost savings in the overall production budget without compromising on quality.
- Enhanced Supply Chain Reliability: Supply continuity is greatly improved due to the commercial availability of the key starting materials. Unlike custom-synthesized precursors that may have long lead times, the olefins and sulfoxides needed to generate the sulfonium salts are widely sourced from global chemical suppliers. This diversification of the raw material base reduces dependency on single-source vendors and mitigates the risk of production stoppages due to upstream shortages. Furthermore, the robustness of the reaction conditions allows for manufacturing in standard stainless steel reactors rather than requiring lined vessels for highly corrosive acids or bases, increasing the number of qualified contract manufacturing organizations capable of executing the process.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the exothermic profile of the reaction, which is manageable under standard cooling conditions, unlike some highly energetic cyanation reactions. The use of DMF as a solvent, while requiring recovery, is well-established in the industry with mature recycling technologies available to minimize waste discharge. The absence of heavy metal co-catalysts like nickel or cobalt simplifies the wastewater treatment process and ensures that the final product meets stringent residual metal specifications required by regulatory bodies. This alignment with green chemistry principles not only reduces environmental fees but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, providing clarity for R&D teams evaluating its potential integration into their existing pipelines. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance to practical application scenarios. Understanding these nuances is essential for assessing the feasibility of adopting this method for specific target molecules within your development portfolio.
Q: What are the critical reaction conditions for this cyanation process?
A: The optimal conditions involve using palladium acetate as the catalyst, X-PHOS as the ligand, and potassium tert-butoxide as the base in DMF solvent at 80°C.
Q: Why are alkenyl sulfonium salts preferred over allyl halides for this synthesis?
A: Alkenyl sulfonium salts offer superior structural diversity, easier preparation, and better stability compared to traditional allyl halides which often suffer from storage issues.
Q: Does this method support large-scale production of diverse substrates?
A: Yes, the method demonstrates broad substrate scope including various aryl and heteroaryl groups, making it highly suitable for commercial scale-up of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-substituted Alkenyl Cyanide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development timelines. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed cyanation route and is fully equipped to translate these laboratory findings into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to market launch. Our state-of-the-art facilities are designed to handle sensitive organometallic chemistry with rigorous QC labs dedicated to maintaining stringent purity specifications, guaranteeing that every batch of multi-substituted alkenyl cyanide meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your needs, obtain specific COA data for our catalog compounds, and receive detailed route feasibility assessments that demonstrate how we can optimize your supply chain for maximum efficiency and reliability.
