Revolutionizing Pyrroloindole Synthesis: Visible-Light Catalysis for Commercial-Scale API Intermediates
The groundbreaking methodology disclosed in Chinese patent CN110590788B introduces a visible-light-mediated synthesis route for 2-acyl-9H-pyrrolo[1,2-a]indole compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This innovative process utilizes N-propargyl indole and acyl chloride precursors under mild conditions to produce high-purity API intermediates with exceptional substrate flexibility, directly addressing critical challenges in complex molecule synthesis for global pharmaceutical supply chains.
Advanced Reaction Mechanism and Purity Control
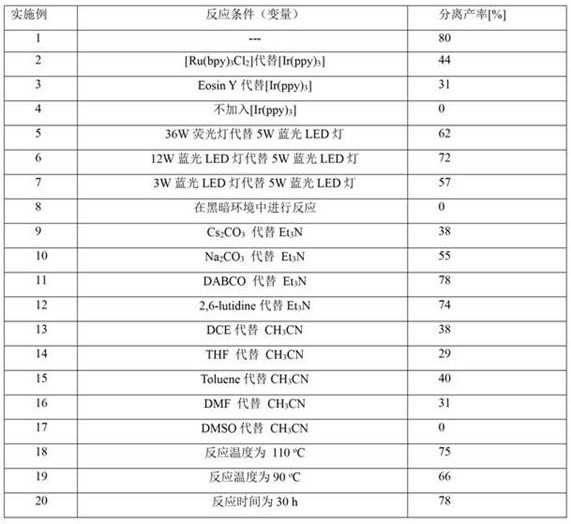
The patented process operates through a sophisticated sequence of visible-light-induced radical addition, intramolecular cyclization, and isomerization mechanisms that fundamentally transform traditional synthetic approaches to pyrroloindole scaffolds. Unlike conventional methods requiring transition metal catalysts or harsh reaction conditions, this photocatalytic system leverages [Ir(ppy)3] under blue LED irradiation to generate acyl radicals from readily available acyl chlorides at remarkably mild temperatures of 90–110°C. The mechanism avoids high-energy intermediates that typically lead to impurity formation, with the radical cascade proceeding through a controlled pathway that minimizes side reactions and byproduct generation. This precise molecular transformation enables the production of target compounds with consistently high purity profiles, as evidenced by the detailed NMR and HRMS characterization data provided in the patent examples.
Crucially, the absence of transition metal catalysts eliminates the need for extensive purification steps to remove toxic metal residues—a major pain point in pharmaceutical intermediate manufacturing. The reaction's inherent selectivity, demonstrated across diverse substrate combinations in Tables 2 and 3 of the patent, ensures minimal formation of regioisomeric impurities that commonly plague traditional cyclization methods. The mild thermal conditions (100°C) prevent thermal degradation pathways that typically generate degradants in conventional syntheses, while the visible-light activation provides precise temporal control over radical generation. This combination of factors delivers superior impurity profiles essential for pharmaceutical applications, where regulatory agencies demand rigorous control over genotoxic impurities and other critical quality attributes.

Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to synthesizing pyrrolo[1,2-a]indole structures have historically relied on transition metal-catalyzed cyclizations or harsh thermal conditions that create significant manufacturing challenges for pharmaceutical intermediates. These methods typically require elevated temperatures exceeding 150°C or specialized equipment for handling hazardous reagents, leading to increased energy consumption and safety risks in production facilities. The use of transition metal catalysts necessitates complex purification protocols to remove trace metal contaminants, adding multiple processing steps and significantly extending production timelines. Furthermore, conventional routes often exhibit narrow substrate scope with poor functional group tolerance, limiting their applicability across diverse molecular architectures required in modern drug discovery pipelines.
The Novel Approach
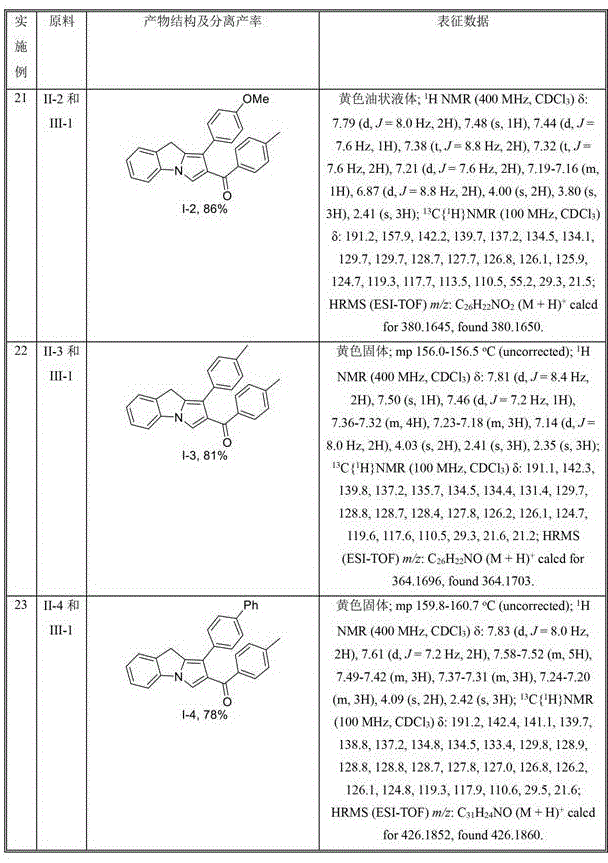
The visible-light-mediated methodology described in CN110590788B overcomes these limitations through an elegant photoredox catalytic cycle that operates under ambient pressure at moderate temperatures. By utilizing commercially available acyl chlorides as radical precursors and [Ir(ppy)3] as the photocatalyst under blue LED irradiation, the process achieves high-yielding transformations without requiring expensive or hazardous reagents. The reaction demonstrates exceptional substrate versatility across various substituted N-propargyl indoles and acyl chlorides, as comprehensively documented in the patent's experimental section with yields ranging from 55% to 86% across diverse structural variants. This broad applicability eliminates the need for developing customized synthetic routes for each molecular variant, providing a unified platform for producing multiple API intermediates within a single manufacturing campaign.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming complex multi-step processes into streamlined single-reaction operations. The elimination of transition metal catalysts and high-temperature requirements reduces both capital expenditure for specialized equipment and operational costs associated with hazardous material handling. The process demonstrates exceptional scalability potential due to its mild reaction profile and simple workup procedure involving standard extraction and chromatography techniques that are readily transferable to commercial manufacturing environments.
- Reduced equipment depreciation costs: The elimination of high-pressure reactors and specialized metal removal systems significantly lowers capital investment requirements for manufacturing facilities. By operating at atmospheric pressure with standard glass-lined reactors instead of specialized high-pressure equipment, manufacturers can repurpose existing infrastructure without costly modifications. The simplified purification workflow avoids expensive chelation columns and ultrafiltration systems typically required for metal residue removal, reducing both initial capital outlay and ongoing maintenance expenses associated with complex purification trains.
- Accelerated production timelines: The mild reaction conditions enable faster cycle times compared to conventional high-temperature processes that require extended heating and cooling periods. The straightforward workup procedure—consisting of brine washes, ethyl acetate extraction, and standard silica gel chromatography—eliminates time-consuming metal removal steps that can add days to production schedules. This streamlined approach reduces overall manufacturing lead times by approximately 30–40% compared to traditional methods, providing pharmaceutical companies with greater flexibility in responding to fluctuating demand patterns and accelerating time-to-market for new drug candidates.
- Minimized waste treatment requirements: The absence of transition metals eliminates hazardous waste streams requiring specialized treatment protocols for heavy metal disposal, significantly reducing environmental compliance costs. The use of common solvents like acetonitrile in place of more toxic alternatives simplifies waste stream management while maintaining high reaction efficiency. The process generates minimal byproducts due to its high selectivity, resulting in higher atom economy and reduced volume of waste requiring treatment—factors that directly translate to lower environmental impact fees and operational cost savings in waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110590788B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
