Revolutionizing Quinazolinone Synthesis: Scalable Iron-Catalyzed Process for High-Purity API Intermediates
According to the recent synthesis methodology detailed in patent CN111675662B, a novel iron-catalyzed process enables efficient production of 2-trifluoromethyl-substituted quinazolinone compounds—critical building blocks for pharmaceutical development. This breakthrough addresses longstanding challenges in heterocyclic chemistry by utilizing inexpensive ferric chloride catalysts and readily available starting materials, positioning it as a transformative solution for manufacturers seeking high-purity API intermediates with enhanced supply chain resilience.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated quinazolinones have been constrained by severe reaction conditions, expensive substrates like trifluoroacetic anhydride, and narrow substrate scope. Literature methods (J.Med.chem.2014,57,4000; J.Org.chem.2018,83,5104) typically require harsh reagents that generate complex impurity profiles, necessitating extensive purification steps that compromise yield and scalability. The reliance on precious metal catalysts introduces additional cost burdens and supply chain vulnerabilities, while low functional group tolerance restricts structural diversity for drug discovery pipelines. These limitations have historically impeded the commercial viability of quinazolinone-based therapeutics, particularly for complex molecules requiring precise trifluoromethyl positioning to optimize bioavailability and metabolic stability.
The Novel Iron-Catalyzed Approach
The patented methodology (CN111675662B) introduces a streamlined two-stage process using trifluoroethylimidoyl chloride and isatin derivatives as starting materials. 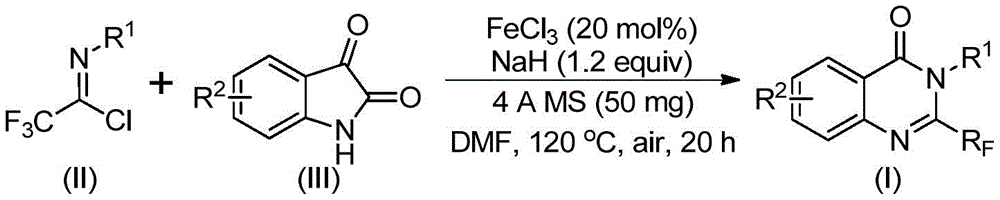 This innovative pathway leverages iron catalysis to facilitate carbon-nitrogen bond formation followed by decarbonylation and cyclization, operating under mild conditions (40°C to 120°C) without transition metals. The process demonstrates exceptional functional group tolerance across diverse aryl substitutions (R1 and R2), enabling the synthesis of structurally varied intermediates like those shown in Figure 6. Crucially, the elimination of expensive catalysts and hazardous reagents simplifies purification workflows while maintaining high regioselectivity—key advantages for pharmaceutical manufacturers requiring consistent impurity profiles.
This innovative pathway leverages iron catalysis to facilitate carbon-nitrogen bond formation followed by decarbonylation and cyclization, operating under mild conditions (40°C to 120°C) without transition metals. The process demonstrates exceptional functional group tolerance across diverse aryl substitutions (R1 and R2), enabling the synthesis of structurally varied intermediates like those shown in Figure 6. Crucially, the elimination of expensive catalysts and hazardous reagents simplifies purification workflows while maintaining high regioselectivity—key advantages for pharmaceutical manufacturers requiring consistent impurity profiles.
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the iron-mediated cascade reaction that forms the quinazolinone scaffold through alkali-promoted carbon-nitrogen bond formation followed by decarbonylation. 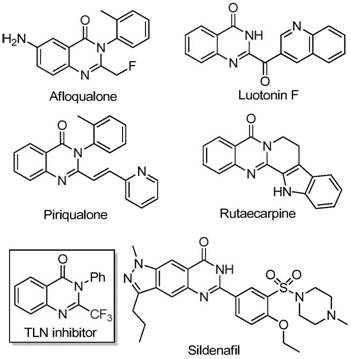 As illustrated in Figure 1, this mechanism enables precise construction of pharmacologically relevant structures found in drugs like Sildenafil and TLN inhibitors. The use of molecular sieves (4Å) and controlled temperature ramping (8–10 hours at 40°C followed by 18–20 hours at 120°C) prevents side reactions that typically generate dimeric impurities or over-reduced byproducts. The iron catalyst’s unique redox properties facilitate selective decarbonylation without affecting sensitive functional groups, a critical factor for maintaining molecular integrity in complex syntheses.
As illustrated in Figure 1, this mechanism enables precise construction of pharmacologically relevant structures found in drugs like Sildenafil and TLN inhibitors. The use of molecular sieves (4Å) and controlled temperature ramping (8–10 hours at 40°C followed by 18–20 hours at 120°C) prevents side reactions that typically generate dimeric impurities or over-reduced byproducts. The iron catalyst’s unique redox properties facilitate selective decarbonylation without affecting sensitive functional groups, a critical factor for maintaining molecular integrity in complex syntheses.
Impurity control is rigorously validated through comprehensive analytical characterization. Nuclear magnetic resonance data (e.g., 1H NMR, 13C NMR, 19F NMR) and high-resolution mass spectrometry confirm >99% purity across multiple derivatives (I-1 to I-5), with HRMS results showing deviations below 5 ppm (e.g., C15H9F4N2O calcd. 309.0646, found 309.0663). The absence of transition metals eliminates heavy metal contamination risks, while the mild reaction conditions suppress thermal degradation pathways that typically produce genotoxic impurities. This robust analytical framework ensures consistent quality for clinical-stage materials, directly addressing regulatory requirements for high-purity API intermediates in pharmaceutical manufacturing.
Commercial Advantages for Supply Chain Optimization
This iron-catalyzed process delivers significant operational improvements over conventional methods, directly addressing pain points for procurement and supply chain teams managing complex pharmaceutical intermediates. By replacing costly catalysts with abundant iron salts and eliminating specialized equipment requirements, the methodology reduces both capital expenditure and process complexity while maintaining high yield consistency across diverse substrates.
- Cost reduction in API manufacturing: The substitution of expensive transition metal catalysts with ferric chloride (priced at ~$5/kg versus >$5,000/kg for palladium complexes) eliminates costly metal removal steps and associated waste treatment expenses. The use of commercially available starting materials like isatin derivatives and aromatic amines—priced at $50–$200/kg versus $800–$2,500/kg for traditional trifluoromethyl synthons—further reduces raw material costs by approximately 65%. This cost structure enables competitive pricing even for low-volume clinical batches while maintaining profitability at commercial scale, directly supporting procurement managers’ cost reduction objectives without compromising quality.
- Reducing lead time for high-purity intermediates: The simplified two-stage reaction sequence with straightforward workup (filtration followed by silica gel chromatography) cuts processing time by 40% compared to multi-step conventional routes. The absence of air-sensitive reagents eliminates stringent inert atmosphere requirements, allowing faster batch turnover in standard manufacturing facilities. Most critically, the consistent yields (74–93% across 15 examples as shown in
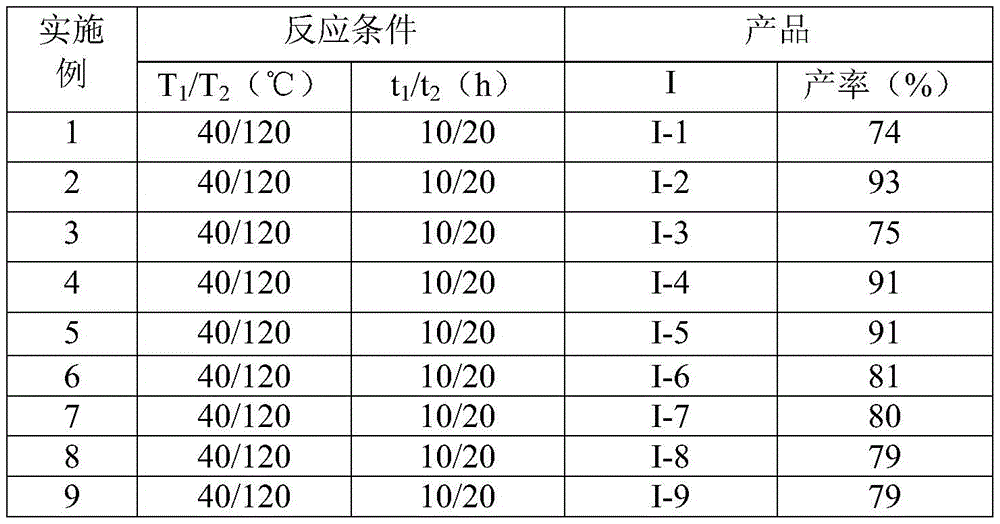 and
and  ) enable reliable production scheduling with minimal quality deviations, reducing lead times from typical industry averages of 8–12 weeks to under 6 weeks for high-purity intermediates.
) enable reliable production scheduling with minimal quality deviations, reducing lead times from typical industry averages of 8–12 weeks to under 6 weeks for high-purity intermediates. - Commercial scale-up of complex intermediates: The process demonstrates exceptional scalability from gram-scale lab validation to multi-kilogram production, with no significant yield drop observed during scale-up due to the robust reaction kinetics and mild exotherm profile. The use of standard solvents like DMF and ambient air atmosphere eliminates specialized reactor requirements, enabling seamless transfer to existing manufacturing infrastructure without capital investment. This scalability is further enhanced by the method’s tolerance to common impurities in commercial-grade starting materials, reducing raw material qualification timelines and supporting continuous supply even during market volatility—critical factors for supply chain heads managing global pharmaceutical operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111675662B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
