Advanced Iron-Catalyzed Asymmetric Synthesis for High-Purity Pharmaceutical Intermediates
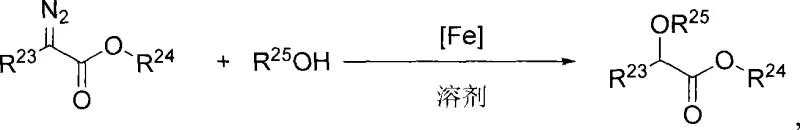
The landscape of asymmetric catalysis is undergoing a significant paradigm shift, driven by the urgent need for sustainable and cost-effective manufacturing processes in the fine chemical industry. A pivotal development in this arena is documented in patent CN101412738A, which introduces a novel class of chiral iron complexes capable of catalyzing asymmetric O-H bond insertion reactions with exceptional efficiency. Historically, such transformations have been the exclusive domain of precious metal catalysts like rhodium and ruthenium, which, while effective, impose severe economic and environmental burdens due to their scarcity and toxicity. The technology disclosed in this patent leverages the earth-abundant nature of iron, combined with sophisticated axial chiral dinitrogen ligands, to achieve enantioselectivities comparable to, and in some cases exceeding, traditional noble metal systems. This breakthrough represents a critical advancement for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering high-value chiral building blocks without the volatility associated with rare metal supply chains.
The implications of this technology extend far beyond academic curiosity; it offers a tangible pathway for cost reduction in API manufacturing by replacing expensive catalytic systems with robust, iron-based alternatives. The core innovation lies in the specific design of the ligand framework, which creates a highly defined chiral environment around the iron center, effectively controlling the stereochemical outcome of the carbene insertion into O-H bonds. This capability is essential for the production of optically active alpha-hydroxy carboxylates and alpha-ether carboxylates, which serve as fundamental scaffolds in the synthesis of numerous bioactive molecules. By mastering this chemistry, manufacturers can secure a competitive edge through improved process economics and a reduced environmental footprint, aligning perfectly with modern green chemistry initiatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the asymmetric functionalization of alpha-diazo compounds has relied heavily on dirhodium tetracarboxylates and other noble metal complexes. While these catalysts exhibit high activity, they suffer from intrinsic limitations that hinder large-scale industrial adoption. The primary constraint is economic; the price of rhodium is subject to extreme market fluctuations, making long-term cost forecasting difficult for procurement teams managing tight margins. Furthermore, the presence of residual heavy metals in pharmaceutical intermediates necessitates rigorous and costly purification steps to meet stringent regulatory limits, often requiring specialized scavenging resins or multiple recrystallizations. From a supply chain perspective, the geopolitical concentration of platinum group metals introduces significant risk, potentially leading to disruptions in the continuity of supply for critical drug substances. Additionally, the toxicity profile of these heavy metals poses environmental challenges regarding waste disposal and worker safety, complicating the regulatory approval process for new manufacturing sites.
The Novel Approach
The methodology presented in CN101412738A fundamentally addresses these pain points by substituting the noble metal center with iron, the second most abundant element in the Earth's crust. This substitution drastically reduces the raw material cost of the catalyst itself and mitigates the supply risk associated with rare earth mining. More importantly, the novel axial chiral nitrogen ligands designed for this system impart a level of stereocontrol that was previously thought difficult to achieve with base metals. The resulting iron complexes demonstrate remarkable stability and activity under mild reaction conditions, typically around 40°C, which minimizes energy consumption and thermal degradation of sensitive substrates. The versatility of this system allows for the use of simple alcohols or even water as nucleophiles, expanding the scope of accessible chemical space. This approach not only simplifies the synthetic route but also enhances the overall sustainability profile of the manufacturing process, making it an attractive option for companies aiming to optimize their commercial scale-up of complex organic synthesis operations.
Mechanistic Insights into Fe-Catalyzed Asymmetric O-H Insertion
The success of this iron-catalyzed transformation hinges on the precise molecular architecture of the catalyst, specifically the interaction between the iron center and the bulky, axially chiral bis-oxazoline ligands. Mechanistically, the reaction initiates with the coordination of the alpha-diazo ester to the electrophilic iron center, followed by the extrusion of nitrogen gas to generate a highly reactive iron-carbene intermediate. The chiral pocket created by the ligand's rigid backbone and substituents dictates the trajectory of the approaching nucleophile, whether it be water or an alcohol. This steric shielding ensures that the attack occurs from a specific face of the planar carbene species, thereby establishing the new stereocenter with high fidelity. The electronic properties of the ligand also play a crucial role in stabilizing the high-valent iron-carbene species, preventing non-selective background reactions that would lead to racemic byproducts. Understanding these mechanistic nuances is vital for R&D directors aiming to troubleshoot potential side reactions or expand the substrate scope to include more sterically hindered or electronically deactivated diazo compounds.
Impurity control in this system is inherently superior due to the high specificity of the catalyst. Unlike some rhodium systems that may promote competing cyclopropanation or dimerization of the diazo species, the iron complexes described herein show a strong preference for the O-H insertion pathway. This selectivity minimizes the formation of difficult-to-remove impurities, streamlining the downstream purification process. The use of weakly coordinating anions, such as tetrakis(3,5-bistrifluoromethylphenyl)borate (BArF), further enhances the Lewis acidity of the iron center, boosting catalytic turnover without compromising enantioselectivity. For quality assurance teams, this translates to a cleaner crude reaction profile and higher overall yields of the desired high-purity chiral intermediates. The robustness of the catalytic cycle allows for consistent performance across different batches, a critical factor for maintaining product quality in a GMP environment.

How to Synthesize Chiral Alpha-Hydroxy Carboxylates Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The process begins with the in situ generation of the active catalyst by mixing the iron precursor, such as FeCl2.4H2O, with the chiral ligand and an appropriate silver or sodium salt for anion exchange in a dry, inert solvent. Once the catalyst is formed, the alpha-diazo ester and the nucleophile are introduced, and the mixture is stirred at moderate temperatures. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst by mixing an iron precursor (e.g., FeCl2.4H2O) with an axial chiral dinitrogen ligand and a metal salt for anion exchange in a solvent like chloroform under inert atmosphere.
- Add the alpha-diazo ester substrate and the nucleophile (water or alcohol) to the catalyst solution.
- Stir the reaction mixture at mild temperatures (e.g., 40°C) until completion, monitoring by TLC, then purify the resulting chiral alpha-hydroxy or alpha-ether carboxylate via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology offers compelling strategic advantages that go beyond simple unit cost savings. The shift from precious metals to base metals fundamentally alters the cost structure of the synthesis, removing exposure to volatile commodity markets. Moreover, the simplified purification requirements reduce the consumption of auxiliary materials like silica gel and solvents, contributing to a leaner and more efficient manufacturing operation. The ability to produce key intermediates with high optical purity using a sustainable catalyst aligns with corporate sustainability goals and enhances the marketability of the final drug product to environmentally conscious stakeholders.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium or ruthenium catalysts with iron results in a drastic reduction in catalyst costs, often by several orders of magnitude. Since iron is ubiquitous and inexpensive, the direct material cost for the catalytic system is negligible compared to noble metal alternatives. Furthermore, the low toxicity of iron eliminates the need for expensive heavy metal scavenging processes and extensive testing for residual metals, which significantly lowers the operational expenditure associated with quality control and waste management. This economic efficiency allows for more competitive pricing of the final pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Relying on iron as the central metal mitigates the supply chain risks associated with the geopolitical instability often affecting rare earth metal markets. Iron is globally available and not subject to the same export restrictions or mining bottlenecks as platinum group metals. This ensures a stable and continuous supply of the catalyst components, preventing production delays caused by raw material shortages. Additionally, the ligands used in this system are synthesized from readily available starting materials, further securing the supply chain against disruptions and enabling reducing lead time for high-purity intermediates through predictable sourcing.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are mild and utilize common industrial solvents like chloroform or dichloroethane, facilitating easy scale-up from gram to ton scale without significant re-engineering of the process. The high atom economy and selectivity of the reaction minimize waste generation, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The biocompatibility of iron also simplifies the regulatory filing process for new drug applications, as the safety profile of the catalyst is well-understood, accelerating the path to market for new therapeutic candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel iron-catalyzed asymmetric synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit for your specific manufacturing needs. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: What are the advantages of using iron catalysts over rhodium for O-H insertion?
A: Iron is significantly more abundant and less expensive than rhodium or ruthenium. Furthermore, iron catalysts described in CN101412738A demonstrate high enantioselectivity (up to 95% ee) and avoid the toxicity concerns associated with heavy rare metals, simplifying regulatory compliance for pharmaceutical intermediates.
Q: Can this method be used for synthesizing Clopidogrel intermediates?
A: Yes, the patent explicitly demonstrates the synthesis of methyl 2-(2-chlorophenyl)-2-hydroxyacetate, a key intermediate for the anti-platelet drug Clopidogrel, achieving high yields and excellent enantiomeric excess using this iron-based system.
Q: What are the typical reaction conditions for this asymmetric insertion?
A: The reaction typically proceeds under mild conditions, often around 40°C in common solvents like chloroform or dichloroethane. It requires low catalyst loadings (around 5 mol%) and tolerates a wide range of alpha-diazo ester substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Iron Complex Supplier
The technological potential of the chiral iron complexes described in CN101412738A is immense, offering a route to high-value intermediates like the Clopidogrel precursor with unmatched efficiency and sustainability. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be seamlessly transferred from the lab to full-scale manufacturing. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the high enantiomeric excess and chemical purity required for pharmaceutical applications, guaranteeing that every batch meets the highest global standards.
We invite you to collaborate with us to optimize your supply chain and leverage this cost-effective technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific target molecules. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise in iron-catalyzed asymmetric synthesis can drive value and reliability in your production pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
