Engineered Carbonyl Reductase Mutants Enable Scalable Production of High-Purity Chiral Intermediates
Engineered Carbonyl Reductase Mutants Enable Scalable Production of High-Purity Chiral Intermediates
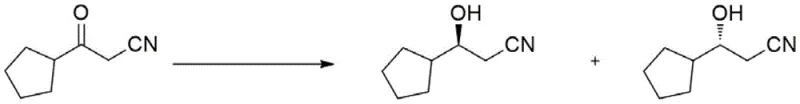
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards biocatalytic solutions for constructing complex chiral molecules, driven by the urgent need for greener and more efficient manufacturing processes. A pivotal development in this domain is documented in patent CN112852768B, which discloses novel carbonyl reductase mutants derived from Burkholderia sp. specifically engineered to synthesize S-3-cyclopentyl-3-hydroxypropionitrile with unprecedented stereoselectivity. This technology addresses a critical bottleneck in the production of chiral beta-hydroxy nitriles, which serve as indispensable building blocks for various bioactive substances and natural products. By leveraging protein engineering to modify the active site of wild-type carbonyl reductase (BuADH), the inventors have created unnatural proteins that exhibit remarkably improved catalytic performance, transforming a challenging chemical reduction into a highly precise biological transformation. For R&D directors and procurement strategists, this patent represents a tangible pathway to securing a reliable pharmaceutical intermediate supplier capable of delivering high-value chiral synthons without the environmental baggage of traditional heavy metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral beta-hydroxy nitriles has relied heavily on chemical asymmetric synthesis methods, which, despite decades of refinement, suffer from inherent structural and economic inefficiencies. Conventional routes often involve asymmetric catalytic hydrogenation or oxo-Michael addition reactions that require expensive chiral ligands, precious metal catalysts like ruthenium or rhodium, and strictly anhydrous conditions to prevent catalyst deactivation. These chemical processes frequently operate under high pressure and temperature, posing significant safety risks and requiring specialized reactor infrastructure that drives up capital expenditure. Furthermore, achieving high enantiomeric excess (ee) chemically often necessitates multiple recrystallization steps or chromatographic separations, leading to substantial material loss and generating large volumes of organic solvent waste. The presence of trace metal residues in the final product is another persistent concern for pharmaceutical manufacturers, as it mandates rigorous and costly purification protocols to meet stringent regulatory limits for elemental impurities in Active Pharmaceutical Ingredients (APIs).
The Novel Approach
In stark contrast, the biocatalytic approach detailed in the patent utilizes engineered carbonyl reductase mutants to perform the asymmetric reduction of 3-cyclopentyl-3-oxopropanenitrile under exceptionally mild and environmentally benign conditions. This novel methodology replaces hazardous chemical reductants with a renewable cofactor regeneration system, typically employing glucose dehydrogenase (GDH) or formate dehydrogenase (FDH) to recycle NADPH in situ. The reaction proceeds in an aqueous buffer system at near-neutral pH and moderate temperatures ranging from 25°C to 50°C, drastically reducing energy consumption and operational hazards. Most critically, the engineered enzymes demonstrate exquisite stereocontrol, directly yielding the desired S-enantiomer with an ee value exceeding 99%, thereby virtually eliminating the need for downstream chiral resolution. This shift from chemocatalysis to biocatalysis not only simplifies the process workflow but also aligns perfectly with the principles of green chemistry, offering a sustainable route for cost reduction in API manufacturing by minimizing waste treatment costs and raw material usage.
Mechanistic Insights into Carbonyl Reductase Mutant Catalysis
The core innovation lies in the rational design and directed evolution of the BuADH enzyme, where specific amino acid residues within the substrate-binding pocket were mutated to optimize steric fit and electronic interactions with the bulky cyclopentyl group of the substrate. The patent identifies four critical positions—H93, V138, A139, and L144—as hotspots for mutation, with specific substitutions such as Histidine to Phenylalanine (H93F) and Alanine to Valine (A139V) proving particularly effective. These mutations alter the hydrophobicity and spatial volume of the active site, forcing the substrate to bind in a specific orientation that favors hydride transfer to the Si-face of the carbonyl group, thus ensuring the formation of the S-configured alcohol. For instance, Mutant 5, which combines the H93F and A139V mutations, exhibits a relative activity of 16.0 compared to the wild type while maintaining an ee value of greater than 99%. This synergistic effect demonstrates how precise protein engineering can overcome the natural limitations of wild-type enzymes, which often lack the necessary rigidity or specificity for non-natural substrates like 3-cyclopentyl-3-oxopropanenitrile.
From an impurity control perspective, the high fidelity of the mutant enzyme ensures that side reactions such as over-reduction or non-specific reduction of the nitrile group are effectively suppressed. The biocatalytic system operates with high chemoselectivity, targeting only the ketone functionality while leaving the sensitive nitrile group intact, a feat that is difficult to achieve with strong chemical reducing agents like lithium aluminum hydride or sodium borohydride without careful protection-deprotection strategies. The use of whole-cell biocatalysts or lyophilized powder further enhances stability, allowing the reaction to tolerate higher substrate concentrations up to 200 g/L without significant loss of activity. This robustness is crucial for industrial applications, as it minimizes the formation of by-products and simplifies the isolation of the target molecule, resulting in a crude product purity of over 95% before any extensive purification, which significantly streamlines the overall manufacturing process for high-purity OLED material precursors or pharmaceutical intermediates.
How to Synthesize S-3-Cyclopentyl-3-Hydroxypropionitrile Efficiently
The implementation of this biocatalytic route involves a streamlined workflow that begins with the construction of recombinant E. coli strains expressing the optimized mutant genes, followed by high-density fermentation to produce the biocatalyst. The process is designed for operational simplicity, utilizing standard fermentation equipment and avoiding the need for exotic reagents or extreme conditions. Detailed below is the standardized protocol for executing this transformation, which serves as a blueprint for scaling this technology from laboratory benchtop to commercial production vessels. The following steps outline the critical parameters for achieving maximum conversion and stereoselectivity.
- Construct recombinant plasmids encoding specific BuADH mutants (e.g., H93F, A139V) and transform into E. coli host cells for expression.
- Cultivate engineered strains in fermentation media, induce protein expression with IPTG, and harvest wet cells via centrifugation.
- Perform biocatalytic reduction in phosphate buffer (pH 6.5) with glucose cofactor regeneration at 30°C to achieve >99% ee and yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this engineered biocatalytic process offers compelling strategic advantages that extend far beyond simple technical feasibility. The transition from metal-catalyzed chemistry to enzyme-catalyzed biology fundamentally alters the cost structure and risk profile of the supply chain, creating a more resilient and sustainable sourcing model. By eliminating the dependency on volatile precious metal markets and complex chiral ligand synthesis, manufacturers can stabilize their raw material costs and reduce exposure to geopolitical supply disruptions. Furthermore, the simplified downstream processing required due to the high purity of the biocatalytic product translates directly into reduced manufacturing cycle times and lower utility costs, providing a significant competitive edge in the fast-paced pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and chiral ligands removes a major cost driver from the bill of materials, while the mild reaction conditions significantly lower energy consumption for heating and cooling. Additionally, the high stereoselectivity (>99% ee) negates the need for costly chiral resolution steps such as preparative HPLC or multiple recrystallizations, which typically result in a 50% theoretical yield loss in racemic processes. The ability to use inexpensive cofactor regeneration systems (glucose or formate) instead of stoichiometric amounts of expensive reducing agents further drives down the variable cost per kilogram, enabling substantial cost savings in commercial scale-up of complex polymer additives or drug intermediates.
- Enhanced Supply Chain Reliability: Biocatalytic processes are inherently more robust against supply chain shocks because the primary catalyst (the enzyme) is produced via fermentation using renewable feedstocks, rather than extracted from finite mineral resources. The use of standard aqueous buffers and common organic cosolvents like methanol or ethanol ensures that raw material sourcing is not constrained by specialized chemical suppliers. Moreover, the stability of the engineered mutants allows for the storage of biocatalyst batches for extended periods, providing manufacturers with the flexibility to build inventory buffers and ensure continuous production even during upstream logistical delays, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process operates in water-rich systems with minimal organic solvent usage, drastically reducing the volume of hazardous waste generated and simplifying compliance with increasingly strict environmental regulations. The absence of heavy metals in the reaction mixture eliminates the need for complex metal scavenging steps and reduces the burden on wastewater treatment facilities. This green profile not only lowers disposal costs but also enhances the corporate sustainability image, which is becoming a critical factor in vendor selection for multinational corporations committed to carbon neutrality and green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonyl reductase technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this biocatalytic route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What are the key advantages of using carbonyl reductase mutants over traditional chemical reduction?
A: The engineered mutants provide exceptional stereoselectivity (>99% ee) under mild aqueous conditions, eliminating the need for expensive chiral metal catalysts and harsh reducing agents typically required in chemical synthesis.
Q: Which specific amino acid mutations improve the catalytic activity for this substrate?
A: Mutations at positions H93, V138, A139, and L144, particularly the combination found in Mutant 5 (H93F/A139V), significantly enhance both relative activity and optical purity compared to the wild-type enzyme.
Q: Is this biocatalytic process suitable for large-scale industrial production?
A: Yes, the process utilizes robust E. coli expression systems and operates at moderate temperatures (25-50°C) with high substrate loading (up to 200 g/L), making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-3-Cyclopentyl-3-Hydroxypropionitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis route described in patent CN112852768B and are fully equipped to bring this advanced technology to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. Our state-of-the-art facilities include dedicated fermentation suites for biocatalyst production and multipurpose reactors capable of handling the specific aqueous-organic biphasic systems required for this synthesis. We adhere to stringent purity specifications and operate rigorous QC labs equipped with chiral HPLC and GC-MS to guarantee that every batch of S-3-cyclopentyl-3-hydroxypropionitrile meets the highest standards of optical purity and chemical integrity required by global regulatory bodies.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains using this cutting-edge biocatalytic technology. By partnering with our technical team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching from your current chemical route to our enzymatic process. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and discuss route feasibility assessments tailored to your specific project timelines and volume requirements. Let us help you secure a sustainable, cost-effective, and high-quality supply of this critical chiral intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
