Advanced Synthesis of Azoxystrobin Intermediates: A Technical Breakthrough for Scalable Agrochemical Manufacturing
Advanced Synthesis of Azoxystrobin Intermediates: A Technical Breakthrough for Scalable Agrochemical Manufacturing
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of key agrochemical intermediates, particularly Azoxystrobin, widely known in the industry as ICIA 5504. Patent CN102070538B introduces a transformative methodology for preparing (E)-2-[2-(6-(2-cyanophenoxy)pyrimidin-4-yloxy)phenyl]-3-methoxyacrylate, addressing critical bottlenecks in yield and purity that have long plagued conventional manufacturing routes. This technical insight report analyzes the proprietary use of dimethyl sulfate (DMS) as a catalyst, which replaces traditional strong acids to facilitate the critical elimination step, thereby enhancing the overall efficiency of the supply chain for reliable agrochemical intermediate suppliers. By optimizing the reaction conditions and catalyst selection, this process offers a robust pathway for the commercial scale-up of complex fungicide intermediates.
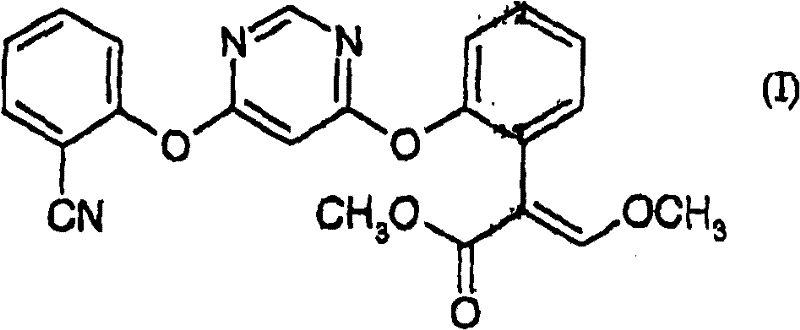
The structural integrity of the final product, as depicted in the associated chemical diagrams, relies heavily on the precise stereochemistry of the acrylate moiety. The patent details a versatile synthetic strategy that allows for flexibility in the reaction sequence, enabling manufacturers to adapt the process based on raw material availability and reactor configuration. This adaptability is crucial for maintaining supply continuity in a volatile market, ensuring that production schedules remain uninterrupted regardless of upstream fluctuations in specific precursor availability. Furthermore, the method emphasizes the use of readily available reagents, positioning it as a cost-effective solution for large-scale industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO92/08703 and PCT/GB97/02015, typically rely on harsh conditions and less efficient catalysts to achieve the desired transformation. For instance, conventional routes often utilize cuprous chloride or potassium carbonate at elevated temperatures ranging from 120°C to 125°C in polar aprotic solvents like DMF. These traditional approaches frequently suffer from significant drawbacks, including the formation of complex impurity profiles that necessitate rigorous and costly purification steps. The use of heavy metal catalysts like copper introduces additional environmental compliance burdens and requires specialized waste treatment protocols, which can drastically increase the operational expenditure for manufacturing facilities. Moreover, the thermal stress imposed on the molecules during these prolonged heating periods can lead to decomposition, resulting in suboptimal yields that fail to meet the stringent purity specifications required by modern pharmaceutical and agrochemical standards.
The Novel Approach
In stark contrast, the novel approach outlined in CN102070538B leverages dimethyl sulfate (DMS) as a highly effective catalyst for the conversion of dimethoxy methyl propionate derivatives into the corresponding methoxy-methyl acrylates. This method operates under milder yet efficient conditions, typically between 90°C and 130°C, utilizing acetic anhydride as both a reagent and a dehydrating agent. The substitution of conventional Bronsted acids like p-toluenesulfonic acid with DMS results in a marked enhancement in total recovery rates, with experimental data indicating yields rising from approximately 71% to over 87%. This significant improvement is attributed to the unique mechanistic pathway facilitated by DMS, which promotes cleaner elimination reactions with minimal side-product formation. The process also offers the flexibility to be conducted in various solvents such as toluene or butyl acetate, or even under solvent-free conditions, providing substantial versatility for process engineers aiming to optimize cost reduction in agrochemical intermediate manufacturing.
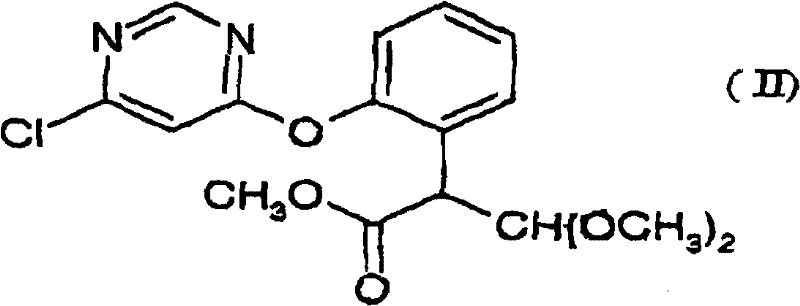
Mechanistic Insights into Dimethyl Sulfate-Catalyzed Elimination
The core innovation of this patent lies in the mechanistic role of dimethyl sulfate during the elimination of methanol from the dimethoxy methyl propionate scaffold. Unlike traditional acid catalysts that may promote non-selective hydrolysis or polymerization, DMS acts as a potent methylating and activating agent that facilitates the formation of the conjugated double bond system essential for the biological activity of Azoxystrobin. The reaction proceeds through a coordinated transition state where the acetic anhydride activates the carbonyl group, while the DMS assists in the departure of the methoxy group, driving the equilibrium towards the formation of the thermodynamically stable (E)-isomer. This precise control over the reaction trajectory ensures that the resulting acrylate intermediate possesses the correct geometric configuration, which is critical for the subsequent coupling reaction with 2-cyanophenol. The avoidance of strong protic acids minimizes the risk of hydrolyzing the sensitive pyrimidine ether linkage, thereby preserving the structural integrity of the molecule throughout the synthesis.
Furthermore, the impurity control mechanism inherent in this DMS-catalyzed route is superior to that of acid-catalyzed alternatives. Comparative examples within the patent demonstrate that reactions employing p-toluenesulfonic acid generate a higher load of tarry by-products and unidentified impurities, which complicate the crystallization process. In the DMS-mediated process, the by-products are primarily volatile esters like methyl acetate, which can be easily removed via simple vacuum distillation at temperatures around 130°C under reduced pressure. This ease of separation significantly reduces the burden on downstream purification units, allowing for a more streamlined workflow that enhances the overall throughput of the manufacturing plant. The ability to distill off low-boiling by-products before the final coupling step ensures that the subsequent reaction with 2-cyanophenol proceeds with high fidelity, free from the interference of residual acidic species that could deactivate the base catalysts used in the second stage.
How to Synthesize ICIA 5504 Efficiently
The synthesis of ICIA 5504 via this patented route involves a strategic sequence of esterification, elimination, and nucleophilic substitution steps that can be adapted for batch or continuous processing. The process begins with the activation of the dimethoxy methyl propionate precursor, followed by a critical elimination step to generate the reactive acrylate species, and concludes with the etherification with 2-cyanophenol to finalize the molecular architecture. Detailed standardized synthetic steps for implementing this high-yield protocol are provided in the guide below, ensuring reproducibility and safety at scale.
- React 2-[2-(6-chloropyrimidin-4-yloxy)phenyl]-3,3-dimethoxy methyl propionate with acetic anhydride using dimethyl sulfate (DMS) as a catalyst at 90-130°C to form the acrylate intermediate.
- Purify the intermediate via vacuum distillation to remove by-products like methyl acetate, followed by solvent exchange if necessary.
- Couple the purified acrylate intermediate with 2-cyanophenol in the presence of potassium carbonate and a base catalyst (e.g., DABCO) at 120-125°C to yield the final ICIA 5504 product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMS-catalyzed synthesis route presents a compelling value proposition centered on cost efficiency and operational reliability. The elimination of expensive and environmentally hazardous heavy metal catalysts, such as cuprous chloride, removes the need for complex metal scavenging processes and specialized waste disposal contracts, leading to significant overhead reductions. Additionally, the use of dimethyl sulfate, a commodity chemical, replaces more costly or less available specialty acids, stabilizing the raw material cost base against market volatility. The simplified workup procedure, characterized by straightforward distillation and crystallization, reduces cycle times and energy consumption, directly contributing to a lower cost of goods sold (COGS) for the final intermediate. This process optimization allows manufacturers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly price-sensitive agrochemical sector.
- Cost Reduction in Manufacturing: The switch to dimethyl sulfate catalysis eliminates the need for expensive transition metal catalysts and the associated purification infrastructure, resulting in substantial cost savings. The higher reaction yields mean that less raw material is wasted per kilogram of product, effectively lowering the material intensity of the process. Furthermore, the ability to recover and recycle solvents like toluene or butyl acetate adds another layer of economic efficiency, making the overall process financially robust.
- Enhanced Supply Chain Reliability: By utilizing widely available commodity chemicals like acetic anhydride and dimethyl sulfate, the supply chain becomes less vulnerable to disruptions caused by the scarcity of niche reagents. The flexibility to operate under solvent-free conditions or with common industrial solvents provides manufacturers with the agility to adapt to local supply constraints. This resilience ensures consistent delivery schedules for downstream formulators, mitigating the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-hundred-gram batches with clear pathways to tonnage production. The reduction in hazardous waste generation, particularly the absence of heavy metal sludge, simplifies regulatory compliance and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only lowers disposal costs but also enhances the corporate sustainability profile of the supplier, a growing requirement for major agrochemical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent's experimental data and claims. These insights are intended to clarify the operational benefits and technical feasibility for potential partners and licensees.
Q: Why is dimethyl sulfate preferred over p-toluenesulfonic acid in this synthesis?
A: Dimethyl sulfate acts as a superior catalyst for the elimination reaction, significantly improving total recovery rates (up to 87.5% vs 71.2% with Tosic acid) and reducing impurity formation, which simplifies downstream purification.
Q: Can the reaction sequence be reversed in this process?
A: Yes, the patent explicitly states that the reaction sequence is flexible. One can first couple the chloropyrimidine derivative with 2-cyanophenol to form the ether linkage, and subsequently perform the acrylate formation, or vice versa, without compromising the final yield.
Q: What are the optimal temperature ranges for the DMS-catalyzed step?
A: The reaction between the dimethoxy methyl propionate derivative and acetic anhydride is optimally conducted between 90°C and 130°C, with a preferred range of 100-125°C to ensure complete conversion while minimizing thermal degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ICIA 5504 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102070538B to maintain competitiveness in the global agrochemical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this DMS-catalyzed process are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of ICIA 5504 meets the exacting standards required for formulation into final fungicide products, providing our clients with peace of mind regarding quality and consistency.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our technical expertise and optimized manufacturing capabilities, we can help you navigate the complexities of scaling this innovative route. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our partnership can drive value and efficiency in your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
