Revolutionizing S-Triazine Synthesis: A Single-Solvent Strategy for High-Purity Agrochemical Intermediates
Revolutionizing S-Triazine Synthesis: A Single-Solvent Strategy for High-Purity Agrochemical Intermediates
The global demand for high-performance herbicides and algae control agents necessitates a paradigm shift in the manufacturing of their core intermediates, specifically 2,4-di(alkylamino)-6-alkylthio-s-triazines. Patent CN1057761C introduces a groundbreaking improved process that fundamentally alters the synthetic landscape by replacing traditional multi-solvent, multi-isolation protocols with a streamlined, single-solvent continuous reaction system. This technological leap addresses critical pain points in fine chemical manufacturing, including environmental compliance, yield optimization, and supply chain efficiency. By leveraging a water-immiscible solvent system coupled with precise phase transfer catalysis, this method achieves exceptional purity levels exceeding 98% while drastically reducing the operational complexity associated with intermediate handling. For R&D directors and procurement specialists alike, understanding the mechanistic nuances of this patent is essential for securing a competitive edge in the agrochemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted s-triazines has been plagued by inefficient operational sequences that inflate costs and compromise environmental sustainability. Traditional methodologies, as referenced in prior art such as U.S. Patents 3,830,810 and 3,766,182, typically require a discontinuous workflow where the reaction solvent must be switched between steps—for instance, transitioning from an aromatic hydrocarbon like toluene in the amination steps to a polar solvent like acetone for the mercaptan substitution. This solvent exchange is not merely a logistical inconvenience; it introduces significant opportunities for product loss, increases the load on solvent recovery systems, and complicates the removal of inorganic salts like sodium chloride. Furthermore, conventional protocols often mandate the isolation and purification of the 2,4-diamino-6-chloro-s-triazine intermediate through filtration or recrystallization. These mechanical separation steps are inherently prone to yield degradation, generate substantial solid waste, and extend the overall production cycle time, thereby reducing the throughput capacity of manufacturing facilities.
The Novel Approach
In stark contrast, the improved process detailed in CN1057761C orchestrates the entire three-step successive reaction within a single water-immiscible organic solvent or a mixture thereof, such as o-xylene, toluene, or methyl isobutyl ketone (MIBK). This continuity eliminates the need for intermediate isolation, allowing the reaction mixture to proceed directly from the first amination to the second, and finally to the thiolation without interruption. The strategic integration of a phase transfer catalyst during the final mercaptan addition step is the linchpin of this innovation, enabling the efficient displacement of the remaining chlorine atom under homogeneous-like conditions despite the heterogeneous nature of the reaction mixture. By maintaining the reaction in a single solvent environment, the process facilitates the easy separation of salt byproducts and allows for the direct recovery of the solvent via azeotropic distillation, resulting in a cleaner product profile and a significantly reduced environmental footprint compared to legacy methods.
Mechanistic Insights into Phase Transfer Catalyzed Nucleophilic Substitution
The chemical elegance of this process lies in the controlled nucleophilic aromatic substitution on the s-triazine ring, where the reactivity of the chlorine atoms is sequentially modulated by temperature and pH. The first two steps involve the reaction of cyanuric chloride with primary amines, where the exothermic nature of the reaction is carefully managed to prevent hydrolysis and ensure regioselectivity. The pH is meticulously buffered between 6.5 and 8.0 for the first addition and 8 to 10 for the second, utilizing acid binding agents like sodium hydroxide to neutralize the generated hydrochloric acid in situ. This precise control prevents the formation of hydrolyzed byproducts which are notoriously difficult to separate from the final active ingredient. The retention of the intermediate in solution preserves its kinetic energy and prevents the crystal lattice formation that would otherwise trap impurities, setting the stage for a highly efficient final transformation.
The critical third step involves the displacement of the final chlorine atom by an alkyl mercaptan, a reaction that is kinetically sluggish without assistance due to the decreased electrophilicity of the triazine ring after two electron-donating amino groups have been attached. 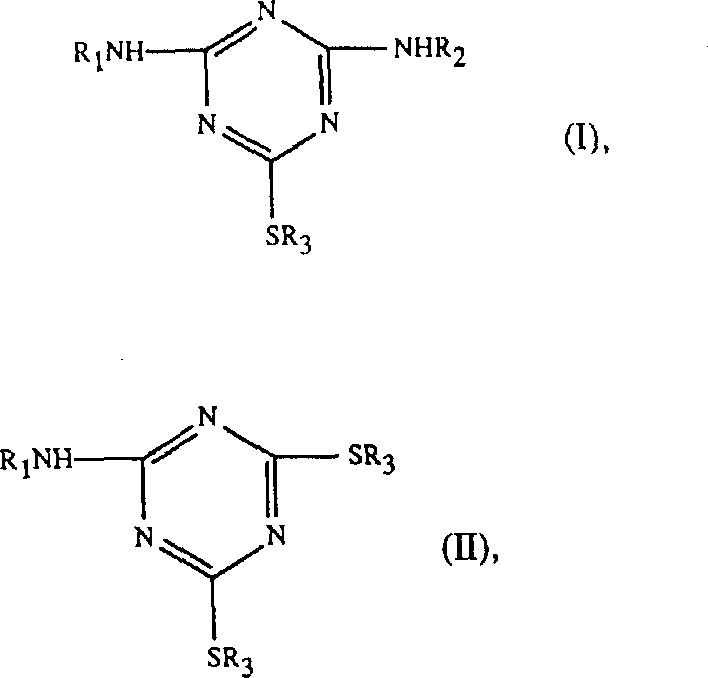 As illustrated in the general structures, the introduction of the phase transfer catalyst, such as tetrabutylammonium bromide or tributylmethylphosphonium chloride, bridges the interface between the organic phase containing the triazine intermediate and the aqueous phase containing the base and mercaptan species. Experimental data from the patent confirms the necessity of this catalyst; in its absence, conversion rates plummet, leaving up to 30% of the undesired chloro-intermediate. With the catalyst, the reaction proceeds to near-completion at temperatures between 90-130°C, yielding products with purity levels consistently above 96%, demonstrating a robust mechanism that translates effectively from laboratory bench to industrial reactor.
As illustrated in the general structures, the introduction of the phase transfer catalyst, such as tetrabutylammonium bromide or tributylmethylphosphonium chloride, bridges the interface between the organic phase containing the triazine intermediate and the aqueous phase containing the base and mercaptan species. Experimental data from the patent confirms the necessity of this catalyst; in its absence, conversion rates plummet, leaving up to 30% of the undesired chloro-intermediate. With the catalyst, the reaction proceeds to near-completion at temperatures between 90-130°C, yielding products with purity levels consistently above 96%, demonstrating a robust mechanism that translates effectively from laboratory bench to industrial reactor.
How to Synthesize 2,4-di(alkylamino)-6-alkylthio-s-triazines Efficiently
The implementation of this synthesis route requires strict adherence to the sequential addition of reagents and precise thermal management to maximize yield and safety. The process begins with the suspension of cyanuric chloride in a selected water-immiscible solvent, followed by the controlled addition of the first amine at low temperatures to manage exotherms. Subsequent steps involve ramping the temperature to facilitate the second amination and finally the thiolation under pressure if gaseous mercaptans are used. The elimination of intermediate work-ups simplifies the operational protocol significantly, allowing for a 'telescoped' manufacturing approach. For detailed standard operating procedures and specific stoichiometric ratios validated by pilot plant data, please refer to the technical guide below.
- React cyanuric chloride with the first alkylamine in a water-immiscible solvent (e.g., o-xylene) at 0-75°C with an acid binding agent to maintain pH 6.5-8.0.
- Without isolating the intermediate, add the second alkylamine to the reaction mixture at 20-95°C, maintaining pH 8-10 to complete the second substitution.
- Add alkyl mercaptan and a phase transfer catalyst (e.g., tetrabutylammonium bromide) to the mixture and heat to 60-150°C to displace the final chlorine atom.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this single-solvent methodology represents a substantial opportunity for cost optimization and risk mitigation. The elimination of solvent switching removes the capital and operational expenditure associated with managing multiple solvent recovery streams and the energy-intensive distillation processes required to swap solvents mid-batch. Furthermore, by avoiding the isolation of intermediates, the process removes several unit operations such as filtration, drying, and re-slurrying, which are often bottlenecks in batch processing. This streamlining directly correlates to a reduction in labor hours, equipment occupancy time, and utility consumption, driving down the overall cost of goods sold (COGS) without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the intensification of the reaction sequence. By telescoping three reaction steps into a single vessel without intermediate isolation, manufacturers can significantly reduce the consumption of raw materials that are typically lost during mechanical transfer and purification stages. The ability to use a single solvent throughout the entire process also simplifies solvent recycling, lowering the net consumption of expensive organic diluents like o-xylene or MIBK. Additionally, the high conversion efficiency ensured by the phase transfer catalyst minimizes the formation of hard-to-remove impurities, reducing the need for costly downstream purification treatments such as extensive recrystallization or chromatography.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the use of commodity chemicals. The process relies on widely available starting materials like cyanuric chloride and common alkylamines, reducing the risk of raw material shortages. The simplified workflow, which eliminates complex isolation steps, reduces the likelihood of batch failures due to handling errors or equipment malfunctions associated with solid-liquid separations. This reliability ensures more consistent lead times and allows suppliers to maintain steadier inventory levels of critical agrochemical intermediates, safeguarding the production schedules of downstream formulators.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the reduction in waste generation is a decisive advantage. The single-solvent approach drastically cuts down on the volume of wastewater and hazardous waste produced, as there is no need to wash away residual solvents from previous steps or dispose of filter cakes from intermediate isolations. The ease of removing sodium chloride byproducts from the organic phase further simplifies effluent treatment. This cleaner profile not only aligns with increasingly stringent global environmental regulations but also facilitates easier scale-up, as heat and mass transfer challenges associated with slurry handling and filtration are minimized in the continuous liquid-phase process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved s-triazine synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, catalyst selection, and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary advantages of the single-solvent method over conventional processes?
A: The single-solvent method eliminates the need for solvent switching (e.g., from toluene to acetone) and intermediate isolation. This significantly reduces cycle time, minimizes waste generation, and simplifies the removal of sodium chloride byproducts, leading to higher overall purity.
Q: Why is a phase transfer catalyst critical in the mercaptan addition step?
A: Experimental data indicates that without a phase transfer catalyst, the displacement of the final chlorine atom by the mercaptan is incomplete, resulting in significant impurities (up to 30% chloro-byproduct). The catalyst ensures near-quantitative conversion under milder conditions.
Q: Can this process be scaled for industrial herbicide production?
A: Yes, the process utilizes common industrial solvents like o-xylene and toluene and avoids complex purification steps like recrystallization of intermediates. The robustness of the reaction conditions (pH and temperature control) makes it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-di(alkylamino)-6-alkylthio-s-triazines Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the single-solvent s-triazine process is optimized for your specific volume requirements. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the high standards required for agrochemical applications, delivering intermediates with minimal impurity profiles and consistent physicochemical properties.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current sourcing model. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and reduce total landed costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
