Advanced 3-Step Synthesis of Tris-Aryl-S-Triazines for Commercial UV Stabilization
The chemical industry continuously seeks more efficient pathways for producing high-performance stabilizers, and patent CN1232823A presents a significant breakthrough in the synthesis of tris-aryl-o-hydroxyphenyl-s-triazines. This intellectual property outlines a novel three-step process starting from cyanuryl chloride or corresponding phenoxy substituted triazines, which fundamentally alters the traditional manufacturing landscape for UV absorbers. Unlike conventional methods that often struggle with selectivity issues and excessive waste generation, this invention leverages specific nucleophilic displacement reactions followed by controlled Friedel-Crafts alkylation. The result is a robust methodology capable of producing asymmetric three-aryl-s-triazines with higher precision and reduced environmental impact. For R&D directors and procurement specialists, understanding this patented route is critical as it offers a tangible pathway to cost reduction in polymer additives manufacturing while ensuring the supply of high-purity UV absorber materials. The process is designed to overcome the non-selective nature of direct Friedel-Crafts reactions on cyanuryl chloride, which has long been a bottleneck in scaling these complex molecules.
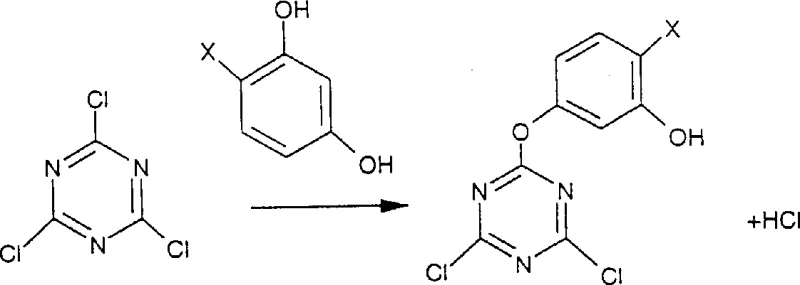
Furthermore, the versatility of this synthesis allows for the creation of various derivatives, including those based on resorcinol, which are essential for protecting natural or synthetic materials from actinic radiation. The ability to control the substitution pattern on the s-triazine ring means that manufacturers can tailor the UV absorption profile to specific polymer matrices, enhancing the longevity and performance of end products. This level of control is achieved through the strategic use of protecting groups and leaving groups that respond differently to nucleophilic and electrophilic conditions. By integrating this technology, supply chain heads can anticipate a more reliable plastic additives supplier network, as the simplified step count reduces the probability of batch failures and production delays. The patent explicitly details conditions that minimize the use of hazardous Lewis acids in favor of protic acids in the final stages, aligning with modern environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of asymmetric three-aryl-s-triazines has been plagued by the inherent lack of selectivity in Friedel-Crafts reactions involving cyanuryl chloride. When attempting to introduce different aryl groups sequentially, the reaction often proceeds non-selectively, leading to a mixture of symmetric and asymmetric products that are difficult and costly to separate. Conventional four-step processes were developed to mitigate this, involving the installation and removal of protecting groups such as methyl mercaptan, which adds significant complexity and waste to the operation. These traditional routes typically require stoichiometric amounts of Lewis acids like aluminum chloride, generating substantial quantities of aluminum waste that are environmentally intractable and expensive to dispose of. The need for multiple isolation and purification steps between each reaction stage further drives up the cost of goods sold and extends the lead time for high-purity UV absorbers. Additionally, the harsh conditions required for these classical methods can degrade sensitive functional groups, limiting the scope of substrates that can be utilized. For procurement managers, these inefficiencies translate into volatile pricing and supply chain vulnerabilities, as the reliance on hazardous reagents introduces regulatory risks.
The Novel Approach
The novel approach disclosed in CN1232823A circumvents these obstacles by utilizing a three-step synthesis that capitalizes on the differential reactivity of leaving groups on the triazine ring. By initially replacing one or two chlorine atoms with a phenolic moiety via nucleophilic substitution, the process creates an intermediate where the remaining chlorine atoms are activated for subsequent Friedel-Crafts reactions without the need for external protecting groups. This strategic maneuver eliminates the need for the fourth step found in prior art, thereby streamlining the entire production workflow. The invention further innovates by demonstrating that protic acids, such as hydrogen chloride or methylsulfonic acid, can effectively catalyze the final substitution of phenoxy groups with phenols like resorcinol. This shift from stoichiometric Lewis acids to catalytic protic acids drastically reduces the generation of metal waste and simplifies the workup procedure. The ability to perform these reactions in a pot or with minimal isolation steps enhances the overall yield and throughput of the manufacturing plant. For technical teams, this represents a significant advancement in process chemistry, offering a cleaner, faster, and more economical route to commercial scale-up of complex polymer additives.
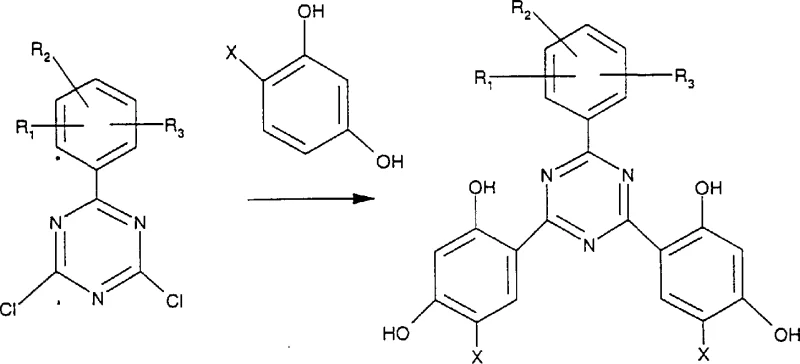
Mechanistic Insights into Protic Acid-Catalyzed Friedel-Crafts Reaction
The core mechanistic advantage of this patent lies in the nuanced understanding of leaving group ability and catalyst selection during the Friedel-Crafts alkylation steps. In the second step of the novel process, a Lewis acid catalyst is employed to replace the remaining chlorine atoms on the triazine ring with aryl groups, utilizing the phenoxy moiety installed in the first step as a temporary blocking group. This phenoxy group is sufficiently stable under Lewis acid conditions to prevent unwanted substitution, yet it acts as an effective leaving group in the presence of strong protic acids during the third step. This differential reactivity is the key to achieving asymmetry without complex protection-deprotection sequences. The patent highlights that the phenoxy group is a poorer leaving group than chlorine under Friedel-Crafts conditions, which allows for the selective introduction of the second aryl group. Subsequently, the use of a strong protic acid activates the phenoxy group for displacement by a strong Friedel-Crafts substrate like resorcinol. This mechanism ensures that the final product is formed with high regioselectivity, minimizing the formation of isomeric impurities that could compromise the UV absorption efficiency. For R&D directors, this mechanistic clarity provides confidence in the reproducibility and robustness of the process when transferring from lab to pilot scale.
Impurity control is inherently built into this synthetic design through the selective nature of the nucleophilic and electrophilic steps. By avoiding the non-selective direct reaction of cyanuryl chloride with multiple aryl sources, the process prevents the formation of symmetric by-products that are notoriously difficult to remove. The use of specific temperature ranges, such as -20°C to 50°C for the nucleophilic step and 70°C to 150°C for the Friedel-Crafts step, further optimizes the reaction kinetics to favor the desired pathway. The patent also notes that the hydrogen chloride gas released during the initial nucleophilic reaction can sometimes act as a catalyst for subsequent steps, reducing the need for additional reagents. This self-catalyzing aspect not only lowers material costs but also simplifies the reaction mixture, making downstream purification more straightforward. The rigorous control over reaction parameters ensures that the final s-triazine product meets stringent purity specifications required for high-performance applications in polymers and coatings. This level of quality control is essential for maintaining the integrity of the supply chain and ensuring consistent performance for end-users.
How to Synthesize Tris-Aryl-S-Triazines Efficiently
The synthesis of these advanced UV absorbers begins with the careful selection of starting materials, specifically cyanuryl chloride and the appropriate phenolic compounds, to ensure the desired substitution pattern is achieved. The process requires precise control over reaction temperatures and stoichiometry, particularly during the nucleophilic displacement phase where alkaline conditions are maintained to facilitate the replacement of chlorine atoms. Following the formation of the phenoxy-triazine intermediate, the reaction mixture is subjected to Lewis acid catalysis to introduce the aryl groups, often utilizing the aromatic substrate itself as the solvent to drive the equilibrium forward. The final transformation involves the displacement of the phenoxy group with resorcinol or similar phenols in the presence of a protic acid catalyst, completing the three-step sequence. Detailed standardized synthesis steps see the guide below.
- Perform nucleophilic displacement of one or two leaving groups in cyanuryl chloride with a phenolic moiety under alkaline conditions.
- Execute a Friedel-Crafts reaction using a Lewis acid catalyst to replace remaining leaving groups with aryl groups.
- Replace the phenolic moiety with a phenol such as resorcinol using a protic acid catalyst or a combination of Lewis and protic acid.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from conventional four-step methods to this novel three-step process offers profound commercial advantages that directly impact the bottom line and supply chain resilience. By eliminating a full synthetic step, manufacturers can significantly reduce the consumption of raw materials, solvents, and energy, leading to substantial cost savings in plastic additives manufacturing. The reduction in process complexity also means fewer unit operations are required, which lowers capital expenditure for new production lines and increases the throughput of existing facilities. For procurement managers, this efficiency translates into a more competitive pricing structure for high-purity UV absorbers, allowing for better margin management in downstream polymer production. Furthermore, the shift away from stoichiometric Lewis acids towards catalytic protic acids reduces the burden of hazardous waste disposal, aligning with increasingly strict environmental regulations and reducing compliance costs. This environmental benefit also enhances the sustainability profile of the supply chain, which is a growing priority for multinational corporations.
- Cost Reduction in Manufacturing: The elimination of the protecting group installation and removal steps inherent in prior art methods removes the need for expensive reagents like methyl mercaptan and the associated handling costs. By reducing the total number of reaction steps from four to three, the overall yield of the process is inherently improved, as fewer isolation and purification stages mean less product loss. The ability to use protic acids instead of large quantities of aluminum chloride in the final step drastically cuts down on the cost of catalysts and the expense of treating aluminum-containing waste streams. These cumulative efficiencies result in a lower cost of goods sold, providing a strategic advantage in a competitive market. Additionally, the potential to use the aromatic substrate as a solvent in the second step further reduces solvent procurement and recovery costs.
- Enhanced Supply Chain Reliability: Simplifying the synthesis route reduces the number of potential failure points in the manufacturing process, leading to more consistent batch quality and higher on-time delivery rates. The use of readily available starting materials like cyanuryl chloride and resorcinol ensures that raw material sourcing is stable and less susceptible to market fluctuations compared to specialized protecting group reagents. The robustness of the reaction conditions, which tolerate a wide temperature range and various solvent systems, allows for greater flexibility in production scheduling and facility utilization. This reliability is crucial for supply chain heads who need to guarantee continuous availability of critical additives to polymer manufacturers. The reduced dependency on hazardous Lewis acids also mitigates regulatory risks that could otherwise disrupt supply.
- Scalability and Environmental Compliance: The three-step process is inherently more scalable due to its reduced complexity and lower waste generation, making it easier to transition from pilot plant to commercial production volumes. The significant reduction in aluminum waste addresses a major environmental pain point associated with traditional Friedel-Crafts chemistry, facilitating easier permitting and compliance with green chemistry initiatives. The ability to perform reactions in fewer pots and with less stringent isolation requirements simplifies the engineering design of large-scale reactors and downstream processing equipment. This scalability ensures that the supply of high-purity UV absorbers can grow in tandem with market demand without requiring disproportionate increases in infrastructure. The cleaner process profile also enhances the safety of the manufacturing environment, reducing occupational health risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for tris-aryl-s-triazines. These answers are derived directly from the technical specifications and beneficial effects outlined in patent CN1232823A, providing clarity on process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for their supply chains. The responses cover aspects of catalyst selection, reaction conditions, and the comparative advantages over existing methods.
Q: How does this novel method improve upon conventional Friedel-Crafts synthesis?
A: Conventional methods often require four steps and suffer from non-selective reactions when preparing asymmetric triazines. This novel method utilizes a specific nucleophilic substitution step that allows for a streamlined 3-step process, significantly reducing complexity and waste.
Q: What catalysts are used in the final step of this synthesis?
A: The final step can be catalyzed by strong protic acids such as hydrogen chloride or methylsulfonic acid, optionally in combination with a Lewis acid synergist. This avoids the need for stoichiometric amounts of Lewis acids like aluminum chloride in certain variations.
Q: What are the primary applications of the resulting s-triazines?
A: The prepared s-triazines function as highly effective UV absorbers, used for stabilizing organic substrates, including polymers and coatings, against the adverse effects of actinic light and radiation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tris-Aryl-S-Triazine Supplier
The technical potential of this three-step synthesis route represents a significant opportunity for optimizing the production of high-performance UV stabilizers. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this patented process to industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of s-triazine meets the exacting standards demanded by the global polymer industry. We understand the critical nature of supply continuity and quality consistency, and our technical team is prepared to navigate the nuances of protic acid catalysis and nucleophilic substitution to deliver superior results. Partnering with us means gaining access to a supply chain that is both robust and innovative.
We invite you to initiate a dialogue regarding your specific requirements for UV absorbers and stabilizers. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential benefits of switching to this novel synthesis method for your operations. Please contact us to request specific COA data and route feasibility assessments tailored to your product portfolio. By collaborating with NINGBO INNO PHARMCHEM, you secure a partner dedicated to driving efficiency and quality in your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
