Scalable Enzymatic Resolution of N-Protected Allylglycinate for High-Purity Chiral Intermediates
Scalable Enzymatic Resolution of N-Protected Allylglycinate for High-Purity Chiral Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methods for producing optically pure amino acid derivatives, which serve as critical building blocks for complex drug molecules. Patent CN101284797A introduces a groundbreaking decomposition method for N-protected allyl glycinate that addresses long-standing challenges in chiral synthesis. This technology leverages the unique properties of Bacillus subtilis proteinase cross-linked enzyme crystals to achieve high-yield kinetic resolution of racemic mixtures. By utilizing this biocatalytic approach, manufacturers can access N-protected L-allylglycine and its esters with superior optical purity, overcoming the limitations of traditional chemical synthesis or less efficient enzymatic methods. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented processes is essential for securing a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure N-protected allylglycine has been fraught with significant technical and economic hurdles. Conventional chemical routes often involve multi-step sequences that suffer from low overall yields and poor stereocontrol, necessitating costly purification steps to remove unwanted enantiomers. Furthermore, earlier enzymatic approaches relied heavily on alpha-chymotrypsin, an enzyme derived from animal sources. The reliance on animal-derived biologics introduces severe supply chain vulnerabilities, including batch-to-batch variability, high procurement costs, and strict regulatory restrictions in pharmaceutical manufacturing due to the risk of viral contamination. These factors collectively render traditional methods unsuitable for the cost reduction in pharmaceutical intermediates manufacturing required by modern high-volume production standards.
The Novel Approach
The innovative strategy outlined in the patent utilizes a microbial-derived biocatalyst that offers a sustainable and economically viable alternative. By employing Bacillus subtilis proteinase cross-linked enzyme crystals, the process achieves highly selective hydrolysis of the racemic N-protected allyl glycinate. This method not only eliminates the dependency on animal-derived enzymes but also significantly enhances the stability and reusability of the catalyst. The reaction proceeds under mild conditions in a phosphate buffer system, ensuring that sensitive functional groups remain intact while delivering high conversion rates. This shift towards microbial biocatalysis represents a paradigm shift in how we approach the commercial scale-up of complex chiral intermediates, offering a pathway that is both environmentally benign and industrially robust.
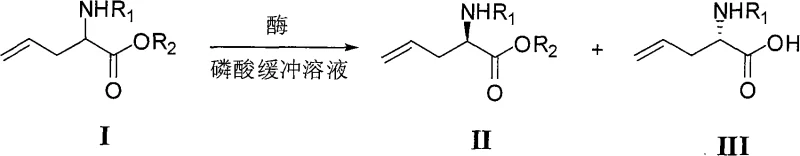
Mechanistic Insights into Subtilisin-Catalyzed Kinetic Resolution
The core of this technology lies in the stereoselective hydrolysis mechanism facilitated by the cross-linked enzyme crystals. In this kinetic resolution process, the enzyme selectively recognizes and hydrolyzes the ester bond of one specific enantiomer of the racemic substrate (Compound I), converting it into the corresponding carboxylic acid (Compound III), while leaving the other enantiomer (Compound II) untouched as the ester. The cross-linking of the enzyme crystals enhances their structural rigidity, allowing them to withstand organic solvents and mechanical stirring without denaturing, which is crucial for maintaining catalytic activity over extended reaction times. This stability ensures that the enzyme maintains its chiral pocket integrity, strictly discriminating between the L- and D-forms of the allylglycine derivative to achieve the reported high enantiomeric excess.
Impurity control is inherently managed through the specificity of the enzymatic reaction. Unlike harsh chemical hydrolysis which might degrade the allyl group or the N-protecting group (such as acetyl, propionyl, or Boc), the enzymatic conditions are mild and highly specific to the ester linkage. This selectivity minimizes the formation of side products such as de-protected amines or polymerized species, resulting in a cleaner crude reaction mixture. Consequently, the downstream purification process is simplified, requiring fewer chromatographic steps to isolate the final high-purity N-protected L-allylglycine products. This mechanistic precision is vital for meeting the stringent quality specifications demanded by regulatory bodies for active pharmaceutical ingredients.
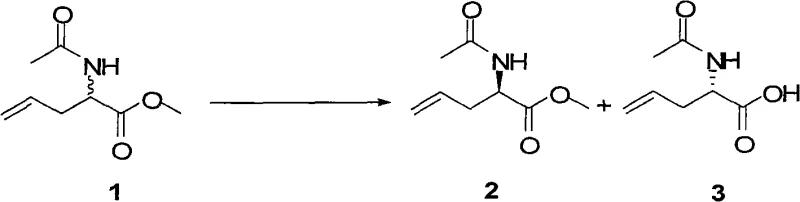
How to Synthesize N-Protected Allylglycinate Efficiently
The synthesis protocol described in the patent provides a clear roadmap for implementing this resolution technology on a pilot or production scale. The process begins with the preparation of a biphasic or homogeneous system containing the racemic substrate, a phosphate buffer to maintain optimal pH, and a co-solvent to enhance substrate solubility. The addition of the cross-linked enzyme crystals initiates the hydrolysis, which is monitored closely to ensure the reaction is stopped at the point of maximum enantiomeric enrichment. Detailed standard operating procedures regarding temperature control, stirring speeds, and work-up protocols are critical for reproducing the high yields and purity levels reported in the experimental examples.
- Dissolve racemic Compound I in a phosphate buffer and organic solvent mixture, then add subtilisin cross-linked enzyme crystals (0.1-20% weight) to initiate hydrolysis.
- Maintain the reaction system between 10-50°C with mechanical stirring for 3 to 24 hours, monitoring progress via HPLC until completion.
- Filter to recover the reusable enzyme, adjust filtrate pH to 8-10 to extract the unhydrolyzed ester (Compound II), then acidify the aqueous phase to pH 2-4 to extract the hydrolyzed acid (Compound III).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic resolution technology offers transformative benefits that extend beyond simple yield improvements. The transition from animal-derived enzymes to microbial cross-linked crystals fundamentally alters the cost structure and risk profile of the supply chain. By removing the reliance on scarce biological resources, manufacturers can secure a more stable and predictable supply of critical catalysts, thereby reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the ability to recycle the enzyme catalyst multiple times without significant loss of activity translates directly into substantial cost savings on raw materials and waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive animal-derived enzymes like alpha-chymotrypsin drastically lowers the direct material costs associated with the resolution process. Additionally, the recyclability of the cross-linked enzyme crystals means that the catalyst cost is amortized over multiple batches, further driving down the cost per kilogram of the final product. The simplified downstream processing, resulting from high reaction specificity, also reduces the consumption of solvents and chromatography media, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing enzymes from microbial fermentation ensures a consistent and scalable supply that is not subject to the fluctuations of the agricultural or slaughterhouse markets. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. The robustness of the cross-linked enzyme crystals also allows for easier storage and transportation, reducing the logistical complexities and cold-chain requirements often associated with liquid enzyme preparations.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial application, utilizing standard reactor equipment and mild reaction conditions that are easily scalable from liters to cubic meters. From an environmental perspective, the use of a biodegradable biocatalyst and the reduction in hazardous waste generation align with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations and enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the process capabilities. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing production workflows.
Q: What are the advantages of using subtilisin cross-linked enzyme crystals over traditional animal-derived enzymes?
A: Unlike alpha-chymotrypsin which is animal-derived, expensive, and restricted in drug manufacture, subtilisin cross-linked enzyme crystals are microbial in origin, cost-effective, recyclable, and suitable for large-scale industrial application without regulatory hurdles associated with animal products.
Q: What level of optical purity can be achieved with this enzymatic resolution method?
A: The patented process demonstrates exceptional stereoselectivity, consistently achieving optical purity greater than 98% for the ester product and greater than 99% for the acid product, meeting the stringent requirements for chiral pharmaceutical intermediates.
Q: Is the enzyme catalyst reusable in this continuous production process?
A: Yes, a key feature of using cross-linked enzyme crystals (CLEC) is their robustness; the patent explicitly states that the enzyme can be filtered and reclaimed after the reaction, significantly reducing catalyst consumption and waste generation compared to free enzyme systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Protected Allylglycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and chemical purity. Our expertise in biocatalysis allows us to optimize processes like the one described in CN101284797A to meet your specific volume and quality requirements.
We invite you to collaborate with us to optimize your supply chain for chiral amino acid derivatives. By leveraging our technical capabilities, you can achieve significant efficiencies in your production processes. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing solutions can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
