Advanced Isocyanate Silane Synthesis: Scalable Technology for High-Performance Polyurethane Applications
The chemical landscape for high-performance polyurethane sealants and modified resins is undergoing a significant transformation driven by the need for safer, more efficient synthesis routes. Patent CN100349902C introduces a groundbreaking methodology for preparing isocyanate group-containing silanes, a critical class of coupling agents and crosslinkers. Unlike conventional pathways that rely on hazardous chlorosilanes or expensive noble metal catalysts, this innovation leverages a carbamate intermediate strategy that fundamentally alters the economic and safety profile of production. By utilizing aminosilanes and carbonates under alkaline catalysis, the process achieves reaction selectivity exceeding 97% and product purity greater than 98%. This technical breakthrough offers a robust foundation for manufacturers seeking to optimize their supply chains while adhering to increasingly stringent environmental regulations. The implications for the global fine chemical market are profound, offering a viable alternative for the reliable isocyanate silane supplier seeking to differentiate through process excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isocyanate-functionalized silanes has been plagued by significant technical and economic hurdles that hinder scalable manufacturing. Traditional methods often involve the reaction of chlorosilanes with sodium isocyanate or potassium isocyanate in alcoholic solvents, a process that generates substantial amounts of hazardous salt waste and requires rigorous handling of toxic reagents. Alternatively, hydrosilylation of unsaturated isocyanates using platinum or palladium catalysts presents a different set of challenges, primarily driven by the exorbitant cost of noble metals and the complexity of catalyst removal to meet purity standards. These legacy processes not only inflate the cost reduction in polyurethane sealant manufacturing but also introduce supply chain vulnerabilities due to the reliance on rare or highly regulated raw materials. Furthermore, the toxicity associated with sodium isocyanate poses severe occupational health risks, necessitating expensive containment infrastructure and waste treatment protocols that erode profit margins. For procurement managers and supply chain heads, these factors translate into higher lead times and increased operational overhead, making the search for alternative synthetic routes a strategic priority.
The Novel Approach
The patented process described in CN100349902C offers a transformative solution by replacing hazardous reagents with benign, commercially available starting materials. By reacting aminosilanes with carbonic esters in the presence of an alkaline catalyst, the method bypasses the need for toxic isocyanate salts and expensive transition metals entirely. This novel approach simplifies the reaction pathway into a manageable sequence of carbamate formation, neutralization, and thermal cracking, which significantly reduces the complexity of downstream processing. The elimination of noble metal catalysts not only lowers the direct material cost but also removes the need for complex metal scavenging steps, thereby streamlining the production workflow. Additionally, the formation of only small amounts of solid salt waste minimizes environmental impact and simplifies waste disposal compliance, aligning with modern green chemistry principles. This shift represents a paradigm change in how high-purity isocyanate silane is produced, offering a scalable and economically superior alternative for the commercial scale-up of complex polymer additives.
Mechanistic Insights into Alkaline-Catalyzed Carbamate Cracking
The core of this innovation lies in the precise control of the carbamate intermediate formation and its subsequent thermal decomposition. The reaction initiates with the nucleophilic attack of the aminosilane on the carbonyl carbon of the carbonate ester, facilitated by an alkaline catalyst such as sodium methylate or magnesium ethylate. This step generates a silicon-based organic carbamate mixture, which serves as a stable precursor to the final isocyanate product. The choice of catalyst is critical, as it must promote the initial addition without causing premature decomposition or side reactions that could compromise the integrity of the silane functionality. The molar ratio of aminosilane to carbonate is carefully balanced, typically ranging from 1:10 to 10:1, to ensure complete conversion while minimizing excess reagent recovery costs. Understanding this mechanistic pathway is essential for R&D directors focused on impurity谱 control, as the stability of the carbamate intermediate dictates the overall yield and selectivity of the process.
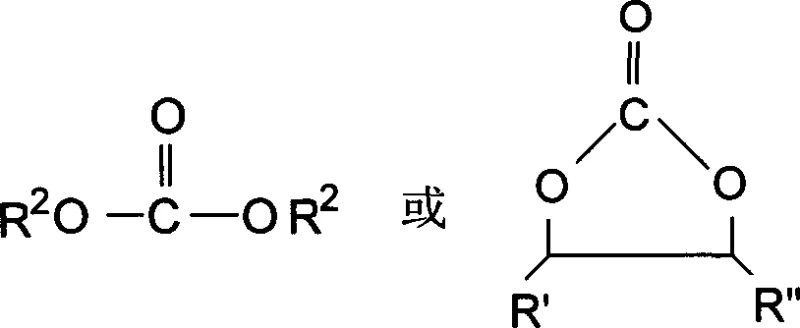
Following the formation of the carbamate intermediate, the process employs a neutralization step to quench the alkaline catalyst, preventing further unwanted reactions during the workup phase. The mixture is then filtered to remove the resulting solid salts, yielding a clear liquid that is remarkably free from particulate contamination. This filtration step is a key differentiator, as it ensures that the subsequent distillation column remains free from fouling, which is a common issue in continuous chemical processing. The clear liquid is then subjected to vacuum distillation cracking at temperatures between 130°C and 140°C under reduced pressure (1-10 kPa). This thermal treatment induces the elimination of the alkoxy group from the carbamate, regenerating the isocyanate functionality attached to the silane backbone. The infrared spectrum of the final product confirms the presence of the characteristic N=C=O absorption peak at 2210-2276 cm-1, validating the successful transformation. This mechanistic clarity allows for precise process optimization, ensuring consistent quality for high-purity OLED material or polymer additive applications.
How to Synthesize Isocyanate Silane Efficiently
Implementing this synthesis route requires a systematic approach to reaction control and purification to maximize yield and safety. The process begins with the slow addition of aminosilane to the carbonate under stirring, followed by a controlled neutralization and filtration sequence to isolate the intermediate. The final cracking step via vacuum distillation is critical for achieving the specified purity levels required for high-performance applications. Detailed standard operating procedures regarding temperature gradients, pressure settings, and catalyst loading are essential for reproducibility on an industrial scale. For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters derived from the patent examples.
- React aminosilane with carbonate using an alkaline catalyst to form a silicon-based organic carbamate mixture.
- Neutralize the reaction mixture with a neutralizing agent such as acetic acid to stop the catalytic activity.
- Filter the mixture to remove solid salts and obtain a clear liquid containing the carbamate intermediate.
- Perform vacuum distillation cracking on the clear liquid at 130-140°C to isolate the high-purity isocyanate silane.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process delivers substantial value by addressing the primary cost drivers and supply chain bottlenecks associated with silane manufacturing. The substitution of expensive noble metal catalysts with inexpensive alkaline earth or alkali metal catalysts results in a drastic reduction in raw material expenditure. Furthermore, the use of readily available carbonates and aminosilanes ensures a stable supply chain, reducing the risk of production delays caused by raw material shortages. The simplicity of the workup procedure, involving basic filtration and distillation, lowers the capital expenditure required for specialized equipment, making it accessible for a wider range of manufacturing facilities. For procurement managers, these factors combine to offer a compelling value proposition that enhances margin potential without compromising on product quality or performance specifications.
- Cost Reduction in Manufacturing: The elimination of platinum or palladium catalysts removes a significant variable cost from the production budget, allowing for more competitive pricing strategies. Additionally, the high reaction selectivity minimizes the formation of by-products, reducing the loss of valuable raw materials and lowering waste disposal costs. The process operates under relatively mild conditions compared to high-pressure hydrosilylation, resulting in lower energy consumption per unit of product. These cumulative efficiencies translate into significant cost savings that can be passed down the supply chain or retained as improved gross margin. By optimizing the catalyst loading and recycling unreacted carbonates, manufacturers can further enhance the economic viability of this route.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like carbonates and aminosilanes ensures that raw material sourcing is not dependent on single-source suppliers or geopolitically sensitive regions. This diversification of the supply base reduces the risk of disruptions and allows for more flexible inventory management strategies. The robustness of the chemical process means that production can be scaled up or down rapidly in response to market demand without requiring complex re-validation of catalyst systems. For supply chain heads, this reliability is crucial for maintaining consistent delivery schedules to downstream formulators of polyurethane sealants. The reduced toxicity of the reagents also simplifies logistics and storage requirements, further streamlining the supply chain operations.
- Scalability and Environmental Compliance: The generation of minimal solid salt waste simplifies the environmental compliance burden, reducing the need for extensive wastewater treatment infrastructure. This green chemistry profile aligns with the sustainability goals of major multinational corporations, making the product more attractive for eco-conscious procurement programs. The straightforward scale-up from laboratory to commercial production is facilitated by the use of standard unit operations like filtration and distillation, which are well-understood by engineering teams. This ease of scale-up reduces the time-to-market for new products derived from this silane, providing a competitive edge in fast-moving industries. The process inherently supports the commercial scale-up of complex polymer additives by providing a consistent and high-quality feedstock.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the patent data and are intended to clarify the operational advantages for potential partners. Understanding these details is crucial for making informed decisions about adopting this route for industrial production. The answers provided reflect the specific technical parameters and benefits outlined in the intellectual property documentation.
Q: How does this process improve safety compared to traditional chlorosilane methods?
A: Traditional methods often utilize toxic sodium isocyanate or chlorosilanes, posing significant handling risks. This patented route uses aminosilanes and carbonates, which are less toxic and generate minimal solid salt waste, drastically improving operational safety and environmental compliance.
Q: What purity levels can be achieved with this vacuum distillation cracking method?
A: The process is designed to achieve exceptional purity levels exceeding 98%, with reaction selectivity reaching over 97%. This high purity is critical for high-grade polyurethane sealants where impurity profiles can affect curing and mechanical properties.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes cheap and easily obtainable raw materials and avoids expensive noble metal catalysts like platinum or palladium. The simple operational path and high yield (over 90%) make it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isocyanate Silane Supplier
The technical potential of this carbamate cracking route represents a significant opportunity for manufacturers seeking to optimize their polyurethane supply chains. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of isocyanate silane meets the exacting standards of the global market. We understand the critical nature of impurity control in high-performance applications and have the analytical capabilities to verify the structural integrity of the final product. Partnering with us ensures access to a supply chain that is both resilient and technically sophisticated.
We invite you to engage with our technical procurement team to discuss how this process can be integrated into your manufacturing strategy. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your operation. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this approach for your specific needs. Our team is ready to support your transition to more efficient and sustainable chemical manufacturing processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
