Scalable Manufacturing of 4-Chloro-2H-Chromene Derivatives via Advanced BTC Catalysis
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic intermediates that balance efficiency with environmental sustainability. Patent CN101225079B introduces a significant advancement in the preparation of 4-chloro-2H-chromene derivatives, a critical scaffold found in numerous bioactive natural products and therapeutic agents. This technology replaces the traditional, hazardous phosphorus oxychloride (POCl3) methodology with a cleaner bis(trichloromethyl)carbonate (BTC) and N,N-dimethylformamide (DMF) system. By generating the Vilsmeier reagent in situ under controlled low-temperature conditions, this process achieves superior selectivity and yield while drastically reducing toxic waste streams. For R&D directors and procurement specialists, this represents a pivotal shift towards greener chemistry without compromising the structural integrity or purity required for downstream drug synthesis.
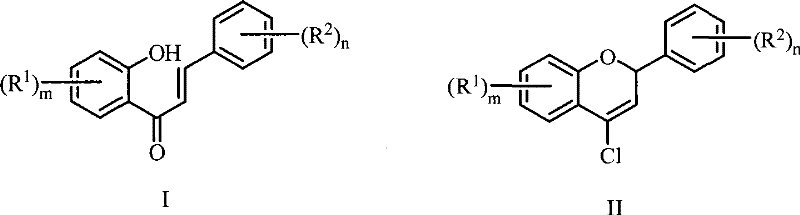
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-chloro-2H-chromene derivatives relied heavily on the Vilsmeier-Haack reaction utilizing POCl3 and excess DMF. While effective, this legacy approach presents severe operational and environmental challenges that modern supply chains can no longer ignore. The primary issue lies in the aggressive nature of POCl3, which generates substantial amounts of phosphorus-containing acidic wastewater, necessitating complex and costly neutralization and treatment protocols before discharge. Furthermore, the conventional method often requires temperatures exceeding 90°C to drive the reaction to completion. At these elevated thermal levels, DMF becomes unstable and prone to decomposition, leading to the formation of unidentified side products that complicate purification and lower the overall isolated yield. This lack of selectivity not only increases raw material consumption but also creates bottlenecks in the final purification stages, impacting the cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The innovative methodology described in the patent data utilizes bis(trichloromethyl)carbonate (BTC), commonly known as triphosgene, as a safer and more controllable chlorinating agent. In this refined process, BTC reacts with DMF at a mild 0-5°C to generate the active Vilsmeier reagent precisely when needed, minimizing reagent degradation. The subsequent addition of the substituted o-hydroxychalcone substrate allows the cyclization to proceed efficiently upon warming to 60-90°C, preferably between 75-85°C. This temperature window is critical; it is high enough to ensure rapid kinetics but low enough to prevent the thermal decomposition of the DMF solvent observed in older methods. As illustrated in the reaction scheme below, this approach streamlines the transformation from the open-chain chalcone to the fused chromene ring system with remarkable efficiency.
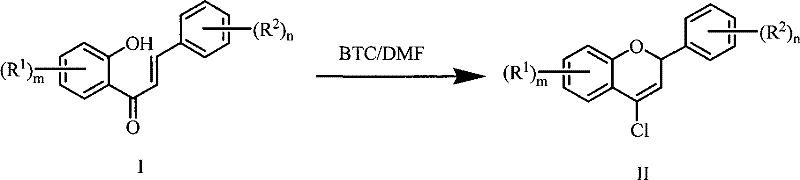
Mechanistic Insights into BTC-Mediated Vilsmeier-Haack Cyclization
The core of this synthetic breakthrough lies in the controlled generation of the electrophilic species. When BTC is introduced to DMF at 0-5°C, it undergoes a stoichiometric reaction to form the chloroiminium salt, the active Vilsmeier reagent, along with the release of carbon dioxide and chloride ions. This in-situ generation ensures that the concentration of the active electrophile is optimized throughout the reaction timeline, preventing the localized hotspots of reactivity that often lead to polymerization or tar formation in batch processes. The mild initial temperature of 0-5°C is essential for stabilizing this intermediate, ensuring that it reacts selectively with the electron-rich enone system of the o-hydroxychalcone rather than degrading the solvent matrix. This precise control over the reaction environment is what allows for the broad substrate tolerance observed in the patent examples, accommodating various electron-donating and electron-withdrawing groups on the aromatic rings.
Following the initial addition, the cyclization mechanism proceeds through an electrophilic aromatic substitution followed by intramolecular ring closure. The presence of the ortho-hydroxyl group is crucial, acting as an internal nucleophile that attacks the activated beta-carbon of the enone system. The use of BTC ensures that the chlorine atom is incorporated at the 4-position of the chromene ring with high regioselectivity. Impurity control is inherently built into this mechanism; by avoiding the harsh conditions of POCl3, the process minimizes the formation of poly-chlorinated byproducts or hydrolysis products that typically plague the crude reaction mixtures of conventional methods. The result is a cleaner crude profile, which significantly reduces the burden on downstream chromatography or crystallization steps, directly contributing to higher overall process mass intensity (PMI) metrics.
How to Synthesize 4-Chloro-2H-Chromene Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety in a pilot or production setting. The process begins with the dissolution of DMF in a suitable organic solvent such as toluene or 1,2-dichloroethane, followed by the careful dropwise addition of the BTC solution while maintaining strict temperature control at 0-5°C. Once the Vilsmeier reagent is formed, the substituted o-hydroxychalcone is introduced, and the mixture is allowed to stir at low temperature before being heated to the optimal cyclization range of 75-85°C. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and workup techniques are critical for maximizing yield and ensuring batch-to-batch consistency.
- Prepare the Vilsmeier reagent by reacting bis(trichloromethyl)carbonate (BTC) with N,N-dimethylformamide (DMF) in an organic solvent at 0-5°C.
- Add the substituted o-hydroxychalcone compound to the reaction mixture at 0-5°C and maintain this temperature for 0.5 to 2 hours.
- Raise the reaction temperature to 60-90°C (preferably 75-85°C) and stir for 1 to 15 hours to complete the cyclization, followed by aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this BTC-based methodology offers tangible strategic benefits beyond simple chemical efficacy. The elimination of phosphorus pollutants translates directly into reduced environmental compliance costs and simpler waste disposal logistics, which are increasingly significant factors in the total cost of ownership for chemical manufacturing. Furthermore, the use of BTC, a stable solid reagent, simplifies storage and handling compared to the corrosive liquids required in traditional methods, enhancing workplace safety and reducing the risk of supply disruptions due to hazardous material transport regulations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive phosphorus waste treatment infrastructure. By operating at milder temperatures that prevent solvent decomposition, the consumption of DMF is drastically reduced, lowering raw material expenses. The higher selectivity of the reaction means less product is lost to side reactions, effectively increasing the throughput per batch without requiring additional capital investment in larger reactors. This efficiency gain allows for a more competitive pricing structure for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: BTC and the required organic solvents like toluene and 1,2-dichloroethane are commodity chemicals with robust global supply chains, ensuring consistent availability. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process are widely accessible. The simplicity of the operation, which does not require exotic equipment or extreme pressure conditions, means that multiple qualified manufacturers can potentially adopt this route, diversifying the supply base and reducing the risk of shortages for critical chromene derivatives.
- Scalability and Environmental Compliance: The reaction profile is exceptionally well-suited for commercial scale-up of complex pharmaceutical intermediates. The exothermic nature of the initial reagent formation is easily managed at 0-5°C, and the subsequent heating phase operates within standard jacketed reactor capabilities. From an environmental standpoint, the removal of phosphorus sources aligns with stringent global green chemistry initiatives, future-proofing the supply chain against tightening regulatory frameworks. This compliance advantage ensures long-term continuity of supply without the risk of production halts due to environmental violations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for making informed sourcing decisions. The following questions address common inquiries regarding the practical implementation and quality attributes of this specific manufacturing route. These insights are derived directly from the experimental data and technical specifications outlined in the relevant intellectual property, providing a transparent view of the process capabilities.
Q: What are the primary advantages of using BTC over POCl3 for chromene synthesis?
A: Using bis(trichloromethyl)carbonate (BTC) eliminates phosphorus-containing wastewater, significantly reducing environmental compliance costs. Additionally, the BTC/DMF system offers milder reaction conditions and higher selectivity compared to the traditional POCl3 method, which often suffers from DMF decomposition at elevated temperatures.
Q: What is the typical yield range for this BTC-mediated cyclization process?
A: According to patent data CN101225079B, the process demonstrates robust yields ranging from approximately 70% to over 90% depending on the specific substituents on the chalcone backbone and the solvent system employed, such as toluene or 1,2-dichloroethane.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly scalable due to the use of readily available solid reagents (BTC) and common organic solvents. The mild temperature profile (0-5°C initiation followed by 60-90°C cyclization) allows for safe heat management in large reactors, facilitating commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2H-Chromene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of novel therapeutics depends on the reliability and quality of the starting materials. Our technical team has extensively evaluated the BTC-mediated cyclization pathway described in CN101225079B and confirmed its potential for delivering high-purity intermediates with consistent impurity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to full-scale market launch. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 4-chloro-2H-chromene derivative meets the exacting standards required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
