Advanced Metal Scavenging Technology for High-Purity Pharmaceutical Intermediates and APIs
Introduction to Advanced Metal Scavenging in Fine Chemical Synthesis
The presence of residual heavy metals in pharmaceutical intermediates and active pharmaceutical ingredients (APIs) represents a critical quality control challenge that directly impacts patient safety and regulatory compliance. Patent CN101638353B introduces a groundbreaking liquid-phase methodology utilizing Disodium Calcium Edetate (EDTA-Ca-Na2) as a superior chelating agent to address this persistent issue. Traditional purification methods often struggle to reduce metal contaminants below the stringent threshold of 20 ppm without compromising the yield or stability of the target molecule. This innovative approach leverages the unique chemical properties of calcium-disodium EDTA to facilitate an efficient ion-exchange mechanism, effectively sequestering toxic metals such as lead, nickel, copper, and tin into the aqueous phase. By integrating this technology, manufacturers can achieve exceptional purity profiles while maintaining the structural integrity of complex organic scaffolds, positioning this method as a vital tool for any reliable pharmaceutical intermediate supplier aiming to meet global GMP standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the removal of trace metals from organic synthesis streams has relied on a variety of techniques, each carrying significant operational drawbacks that hinder cost reduction in API manufacturing. Conventional direct methods, such as filtration with activated carbon or flocculation, are often insufficient for reducing metal levels to the single-digit ppm range required for modern drug substances. Indirect methods involving pH adjustment frequently fail to precipitate all metal species, leaving hundreds of ppm of contamination. Furthermore, advanced techniques like ion-exchange chromatography are limited by low batch throughput and high resin costs, rendering them economically unviable for large-scale production. Functionalized silica gels, while effective, pose risks of damaging glass-lined reactors due to their hardness and require extensive processing times ranging from overnight to several days. Molecular distillation, another alternative, demands high energy input and is restricted to compounds with specific volatility profiles, often leading to thermal degradation of heat-sensitive intermediates.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by employing Disodium Calcium Edetate, a reagent that combines high water solubility with remarkable thermal and chemical stability. Unlike free EDTA, which suffers from poor solubility and requires excessive water volumes that precipitate organic products, or Sodium EDTA, which creates a strongly alkaline environment detrimental to ester and alkali-sensitive groups, the calcium-disodium salt operates at a neutral pH of approximately 7.0. This neutrality allows for the treatment of a vast array of organic compounds without inducing hydrolysis or decomposition. The process involves dissolving the metal-contaminated organic compound and the chelating agent in a biphasic or monophasic solvent system, where the chelator selectively binds heavy metal ions. As demonstrated in the synthesis of complex heterocycles, this method successfully purifies products from reactions involving catalysts like Raney nickel or copper cyanide, achieving residual metal levels well below 20 ppm through simple recrystallization.
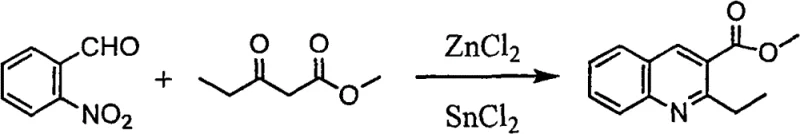
Mechanistic Insights into EDTA-Ca-Na2 Chelation Dynamics
The core efficacy of this purification strategy lies in the thermodynamic stability constants of metal-EDTA complexes relative to the calcium-EDTA complex. In the reaction medium, the Disodium Calcium Edetate acts as a pre-formed chelate where the calcium ion is loosely held compared to transition metals and heavy metals like lead, mercury, or copper. When introduced to the organic phase containing residual metal salts, a ligand exchange reaction occurs wherein the contaminant metal ion (M) displaces the calcium ion (Ca) due to the formation of a more stable M-EDTA complex. This displacement is driven by the higher formation constants of heavy metal-EDTA complexes compared to that of calcium. The resulting metal-EDTA species possesses high hydrophilicity due to its ionic carboxylate groups, causing it to partition preferentially into the aqueous phase of the solvent system. Consequently, the organic product, now stripped of its metallic impurities, remains in the organic phase or precipitates upon cooling, while the toxic metals are safely sequestered in the water layer for disposal.
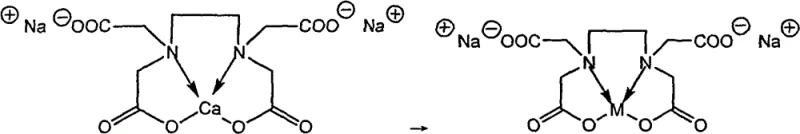
Furthermore, the mechanism ensures robust impurity control by preventing the re-deposition of metals onto the crystal lattice of the product during recrystallization. Since the metal ions are locked in a stable, water-soluble coordination sphere, they are physically separated from the growing organic crystals. This is particularly crucial for catalytic residues from hydrogenation (e.g., Ni, Pd) or coupling reactions (e.g., Cu, Zn), which can otherwise act as pro-oxidants or catalysts for degradation in the final drug product. The use of a biphasic solvent system, typically involving water and a Class III solvent like ethyl acetate or toluene, maximizes the interfacial area for this ion exchange, ensuring rapid kinetics even at moderate temperatures between 20°C and 60°C. This mechanistic elegance translates directly to process reliability, as the chelation is stoichiometric and predictable, allowing for precise calculation of reagent requirements to ensure complete metal scavenging without excess waste.
How to Synthesize High-Purity Organic Compounds Efficiently
Implementing this metal removal technology requires a straightforward adaptation of existing workup procedures, focusing on the strategic introduction of the chelating agent during the isolation phase. The process begins by selecting an appropriate solvent system that dissolves both the crude organic product and the Disodium Calcium Edetate, often utilizing mixtures of water with alcohols, ketones, or esters depending on the solubility profile of the target molecule. Once the system is established, the mixture is agitated at controlled temperatures to allow the chelation equilibrium to shift fully towards the metal-complexed state. Following the reaction period, the system is cooled to induce crystallization of the purified organic compound, leveraging the decreased solubility of the product in the cold solvent mixture while the metal-chelates remain dissolved in the mother liquor. For a comprehensive understanding of the specific operational parameters, including solvent ratios and temperature gradients tailored to your specific molecule, the detailed standardized synthesis steps are provided in the guide below.
- Dissolve the organic compound containing residual metal salts and the EDTA calcium disodium salt chelating agent in a mixed solvent system comprising water and an organic solvent.
- Maintain the reaction mixture at a temperature between 10°C and 100°C for a duration ranging from 5 minutes to 10 hours to facilitate the ion exchange process.
- Cool the reaction mixture to induce recrystallization of the purified organic compound, followed by filtration to separate the solid product from the aqueous phase containing the metal-EDTA complexes.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this EDTA-Ca-Na2 based purification method offers substantial strategic benefits that extend beyond mere technical compliance. The primary advantage lies in the drastic simplification of the downstream processing workflow, which eliminates the need for capital-intensive equipment such as molecular distillation units or columns packed with expensive functionalized silica. By replacing these complex unit operations with a simple liquid-liquid extraction and crystallization step, manufacturers can significantly reduce the overall cycle time for batch production. This streamlining of the process directly contributes to cost reduction in API manufacturing by lowering utility consumption, minimizing solvent usage, and reducing the labor hours associated with monitoring and operating specialized purification equipment. Additionally, the reagent itself, Disodium Calcium Edetate, is a commodity chemical with a stable global supply chain, ensuring that there are no bottlenecks related to the sourcing of exotic scavengers or catalysts.
- Cost Reduction in Manufacturing: The economic impact of switching to this chelation method is profound, primarily driven by the elimination of high-cost consumables and energy-intensive separation techniques. Traditional methods often rely on precious metal scavengers or large quantities of specialized resins that must be disposed of as hazardous waste, incurring significant disposal fees. In contrast, the aqueous waste stream generated by this process contains the metals in a stable, non-leachable form, potentially simplifying wastewater treatment protocols. Furthermore, the high recovery yields associated with gentle recrystallization conditions mean that less starting material is lost to degradation or hold-up in filtration media, thereby improving the overall mass balance and reducing the cost of goods sold (COGS) for high-value intermediates.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemistry enhances the continuity of supply for critical pharmaceutical ingredients. Because the method is tolerant of a wide range of solvents and operates under mild conditions, it is less prone to batch failures caused by minor fluctuations in raw material quality or environmental conditions. The ability to consistently achieve metal levels below 20 ppm reduces the risk of out-of-specification (OOS) results during quality control testing, which is a common cause of shipment delays. This reliability allows supply chain planners to maintain leaner inventory buffers, confident that the purification step will not become a bottleneck. Moreover, the scalability of the process from laboratory benchtop to multi-ton reactors ensures that technology transfer between sites is seamless, supporting a agile and responsive global manufacturing network.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional alternatives, aligning with the increasing regulatory pressure for green chemistry practices. The avoidance of chlorinated solvents often required for silica gel chromatography and the reduction in energy demand compared to distillation contribute to a lower carbon footprint per kilogram of product. The use of Class III solvents, which are recognized by the FDA as having low toxic potential, further simplifies the regulatory dossier for the final drug product. On a commercial scale, the ease of handling aqueous solutions of the chelating agent facilitates automation and continuous processing opportunities, paving the way for future manufacturing efficiencies. This combination of operational simplicity and environmental stewardship makes the technology an attractive option for long-term commercial partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of Disodium Calcium Edetate for metal scavenging, derived directly from the experimental data and scope of the patented technology. These insights are intended to clarify the operational boundaries and potential applications of the method for R&D and production teams evaluating its fit for their specific synthetic routes. Understanding these nuances is essential for optimizing the purification protocol and maximizing the yield and purity of the final active pharmaceutical ingredient.
Q: Why is Disodium Calcium Edetate superior to Sodium EDTA for metal removal in sensitive APIs?
A: Unlike Sodium EDTA, which creates a highly alkaline environment (pH ~11.3) that can hydrolyze esters or degrade alkali-sensitive functional groups, Disodium Calcium Edetate maintains a neutral pH of approximately 7.0. This ensures the structural integrity of delicate pharmaceutical intermediates while effectively chelating heavy metals.
Q: What levels of residual metal reduction can be achieved with this chelation technology?
A: The patented method is capable of reducing residual metal content, such as lead, nickel, copper, and tin, to levels below 20 ppm, meeting stringent international quality specifications for bulk drugs and fine chemical intermediates.
Q: Is this metal removal process scalable for industrial manufacturing?
A: Yes, the process utilizes simple liquid-phase extraction and recrystallization techniques that avoid expensive solid-phase resins or complex molecular distillation, making it highly suitable for large-scale commercial production from 100 kgs to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disodium Calcium Edetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of your intermediates is the foundation of your drug's safety and efficacy. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale chelation to industrial manufacturing is smooth and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of our fine chemicals meets the <20 ppm metal residue standard mandated by global pharmacopoeias. Our commitment to quality assurance means that when you partner with us, you are securing a supply chain that is resilient, compliant, and technically superior.
We invite you to engage with our technical procurement team to discuss how this advanced metal removal technology can be integrated into your current manufacturing processes. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in operational expenditure and waste management costs specific to your facility. We encourage you to contact us today to obtain specific COA data for our high-purity intermediates and to receive detailed route feasibility assessments tailored to your project's unique chemical challenges.
