Advanced Catalytic Strategy for High-Purity Propargyl-Substituted Chiral Oxindole Intermediates
Introduction to Novel Chiral Oxindole Synthesis
The pharmaceutical industry continuously seeks efficient routes to access complex chiral scaffolds that serve as the backbone for potent bioactive molecules. Patent CN115215783A introduces a groundbreaking methodology for the synthesis of propargyl-substituted chiral 3-amino-3,3-disubstituted oxindole compounds, a structural motif prevalent in numerous natural products and therapeutic agents. This innovation addresses a critical gap in organic synthesis by providing a direct, asymmetric route to install alkynyl functionalities onto the oxindole core with exceptional stereocontrol. The disclosed technology leverages a copper-catalyzed decarboxylative propargylation strategy, enabling the construction of two consecutive quaternary carbon chiral centers in a single operation. For R&D directors and process chemists, this represents a significant leap forward in accessing diverse chemical space for antitumor drug discovery, particularly given the demonstrated activity against leukemia cell lines.
Furthermore, the versatility of the terminal alkyne group introduced by this method cannot be overstated. It acts as a privileged functional handle for downstream diversification, allowing medicinal chemists to rapidly generate libraries of derivatives through click chemistry, oxidation, or coupling reactions. This capability drastically accelerates the structure-activity relationship (SAR) studies essential for lead optimization. By integrating this synthetic approach into early-stage drug development pipelines, organizations can streamline the production of high-value intermediates that were previously difficult to obtain with high optical purity. The robustness of the reaction conditions and the broad substrate scope detailed in the patent suggest a high potential for adoption in both academic research and industrial manufacturing settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-amino-3,3-disubstituted oxindoles bearing alkynyl groups has been fraught with challenges regarding regioselectivity and stereocontrol. Traditional alkylation methods often rely on pre-functionalized halides or require harsh basic conditions that can compromise sensitive functional groups elsewhere in the molecule. Moreover, achieving high enantiomeric excess in the formation of quaternary centers at the C3 position of the oxindole ring typically demands expensive chiral auxiliaries or multi-step resolution processes, which inherently lower the overall atom economy and increase waste generation. These limitations not only inflate the cost of goods but also extend the timeline for producing sufficient quantities of material for biological testing. Consequently, the lack of a direct, catalytic asymmetric method has hindered the widespread exploration of this specific chemical space in medicinal chemistry programs.
The Novel Approach
In stark contrast, the methodology described in CN115215783A utilizes a transition metal-catalyzed decarboxylative coupling that operates under remarkably mild conditions. By employing 4-ethynyl cyclic carbonates as electrophilic partners, the reaction proceeds with the extrusion of carbon dioxide, driving the equilibrium forward and minimizing byproduct formation. The use of a chiral copper complex allows for precise facial discrimination during the nucleophilic attack, resulting in products with diastereomeric ratios exceeding 20:1 and enantiomeric excesses up to 97%. 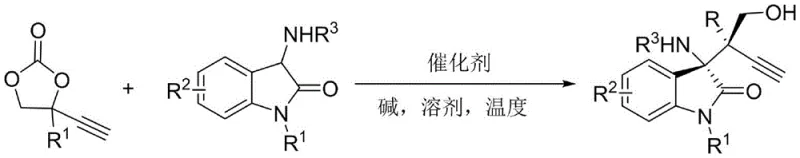 This streamlined approach eliminates the need for stoichiometric chiral reagents and simplifies the purification workflow. The ability to tolerate a wide range of substituents on both the oxindole and the carbonate components underscores the generality of this protocol, making it a powerful tool for the rapid assembly of complex molecular architectures relevant to modern drug discovery.
This streamlined approach eliminates the need for stoichiometric chiral reagents and simplifies the purification workflow. The ability to tolerate a wide range of substituents on both the oxindole and the carbonate components underscores the generality of this protocol, making it a powerful tool for the rapid assembly of complex molecular architectures relevant to modern drug discovery.
Mechanistic Insights into Copper-Catalyzed Decarboxylative Propargylation
The success of this transformation hinges on the intricate interplay between the copper catalyst and the chiral bis-oxazoline ligand. Mechanistically, the copper center coordinates with the terminal alkyne of the cyclic carbonate, facilitating the decarboxylation process to generate a reactive copper-allenyl or copper-propargyl species. This organometallic intermediate is then positioned within the chiral pocket created by the ligand, ensuring that the subsequent nucleophilic attack by the 3-aminooxindole occurs from a specific trajectory. The rigorous control over the transition state geometry is what enables the formation of the challenging all-carbon quaternary stereocenter with such high fidelity. Understanding this mechanism is crucial for process optimization, as factors such as solvent polarity and temperature can subtly influence the coordination sphere of the metal and, consequently, the stereochemical outcome.
Beyond the primary coupling event, the patent highlights the synthetic utility of the resulting propargyl oxindoles through a secondary cyclization cascade.  Under gold or silver catalysis, the internal alkyne can undergo intramolecular hydroalkoxylation or similar cyclization events to forge bridged spirocyclic systems. This tandem capability effectively doubles the value of the initial synthetic effort, providing access to even more rigid and structurally complex scaffolds like compound IV. For supply chain planners, this implies that a single intermediate inventory can serve as a precursor for multiple distinct final active pharmaceutical ingredients (APIs), thereby enhancing supply chain resilience and reducing the need for maintaining separate stockpiles of diverse starting materials.
Under gold or silver catalysis, the internal alkyne can undergo intramolecular hydroalkoxylation or similar cyclization events to forge bridged spirocyclic systems. This tandem capability effectively doubles the value of the initial synthetic effort, providing access to even more rigid and structurally complex scaffolds like compound IV. For supply chain planners, this implies that a single intermediate inventory can serve as a precursor for multiple distinct final active pharmaceutical ingredients (APIs), thereby enhancing supply chain resilience and reducing the need for maintaining separate stockpiles of diverse starting materials.
How to Synthesize Propargyl-Substituted Chiral Oxindoles Efficiently
The experimental protocols outlined in the patent provide a clear roadmap for implementing this chemistry in a laboratory or pilot plant setting. The procedure emphasizes the importance of maintaining an inert atmosphere and controlling the addition sequence of reagents to maximize catalyst turnover and minimize side reactions. Detailed guidance on workup and purification ensures that the final products meet the stringent purity requirements necessary for biological evaluation. While the specific molar ratios and temperatures are optimized for the examples provided, the underlying principles are adaptable to various substrates.
- Prepare the catalytic system by dissolving copper salt (e.g., Cu(acac)2) and chiral bis-oxazoline ligand in an organic solvent like 2-MeTHF at room temperature.
- Cool the mixture to 0°C and sequentially add 4-ethynyl cyclic carbonate, 3-aminooxindole substrate, and an organic base such as triethylamine.
- Stir the reaction at 0°C until completion, then purify the crude product via column chromatography to isolate the high-purity chiral intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this catalytic methodology offers substantial strategic benefits regarding cost efficiency and supply security. The reliance on earth-abundant copper salts rather than precious metals like palladium or rhodium significantly reduces the raw material costs associated with catalyst loading. Furthermore, the commercial availability of the requisite chiral ligands and cyclic carbonate building blocks ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized reagents. This accessibility translates directly into lower procurement overheads and shorter lead times for acquiring the necessary inputs for large-scale production campaigns.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the avoidance of resolution steps drastically simplify the manufacturing process. By achieving high stereoselectivity directly in the bond-forming step, the need for costly chromatographic separations or recrystallizations to upgrade optical purity is minimized. This reduction in downstream processing units leads to significant savings in solvent consumption, energy usage, and labor hours. Additionally, the high atom economy of the decarboxylative reaction means that a larger proportion of the starting mass is incorporated into the final product, reducing waste disposal costs and improving the overall environmental footprint of the synthesis.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a variety of functional groups and proceed at near-ambient temperatures, enhances the reliability of the manufacturing process. Mild conditions reduce the stress on reactor equipment and lower the safety risks associated with high-pressure or high-temperature operations. This operational stability ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and meeting delivery schedules. Moreover, the versatility of the intermediate allows for flexible production planning, where a single stock of the propargyl oxindole can be diverted to different downstream pathways based on real-time market demand.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving standard solvent evaporation and column chromatography, facilitates easy scale-up from gram to kilogram scales without requiring specialized equipment. The use of greener solvents like 2-methyltetrahydrofuran, as exemplified in the optimization studies, aligns with modern sustainability goals and regulatory pressures to reduce the use of chlorinated hydrocarbons. This alignment with green chemistry principles not only improves the corporate social responsibility profile but also future-proofs the manufacturing process against increasingly stringent environmental regulations regarding volatile organic compound emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this synthetic technology. These insights are derived directly from the experimental data and scope limitations defined within the patent documentation, providing a realistic overview of what process teams can expect during technology transfer.
Q: What are the key advantages of this copper-catalyzed method over traditional synthesis?
A: This method offers exceptional stereoselectivity (up to 97% ee and >20:1 dr) under mild conditions (0°C), avoiding the harsh reagents often required in conventional alkylation processes.
Q: Can these intermediates be further functionalized for drug discovery?
A: Yes, the terminal alkyne group serves as a versatile handle for post-synthetic modifications, including click chemistry and cyclization reactions to form complex spiro-oxindole scaffolds.
Q: Is this process scalable for commercial production?
A: The protocol utilizes commercially available catalysts and simple workup procedures, making it highly suitable for scale-up from laboratory to industrial manufacturing levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propargyl-Substituted Chiral Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies in accelerating drug discovery timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable supply chains. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in asymmetric synthesis allows us to navigate the complexities of chiral manufacturing, guaranteeing the optical integrity of every batch delivered to our global partners.
We invite prospective clients to engage with our technical procurement team to discuss how this specific copper-catalyzed route can be integrated into your existing project portfolios. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next generation of antitumor candidates is built on a foundation of chemical excellence and supply chain certainty.
