Advanced Synthesis of 9-Alkyl-10-Hydroxycamptothecin Derivatives for Commercial API Production
Advanced Synthesis of 9-Alkyl-10-Hydroxycamptothecin Derivatives for Commercial API Production
Introduction to Novel Camptothecin Derivatives and Patent CN1616460A
The pharmaceutical industry continuously seeks enhanced topoisomerase I inhibitors to combat malignant tumors, and patent CN1616460A introduces a significant breakthrough in the structural modification of camptothecin alkaloids. This intellectual property details the synthesis of new 9-alkyl-10-hydroxycamptothecin derivatives that exhibit superior antitumor activity compared to the parent compound while addressing historical solubility and toxicity challenges. The core innovation lies in the strategic functionalization of the camptothecin scaffold, specifically targeting the 9 and 10 positions to optimize pharmacological profiles for clinical applications. By leveraging a Claisen rearrangement strategy, the patent enables the introduction of diverse alkyl chains that were previously inaccessible through conventional Mannich reactions, thereby opening new avenues for drug development. This technical advancement is critical for R&D directors seeking high-purity intermediates with well-defined impurity profiles for next-generation oncology therapeutics. The structural integrity of the E-ring lactone, which is essential for biological activity, is meticulously preserved throughout the synthetic sequence described in the documentation. 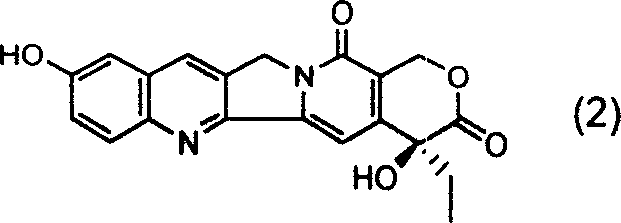
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of camptothecin at the 9-position has been predominantly achieved through the Mannich reaction, which imposes severe structural limitations on the resulting derivatives. This traditional methodology restricts the substituent at the 9-position to a simple aminomethyl group, effectively capping the chemical diversity available for optimizing drug potency and pharmacokinetic properties. Furthermore, the Mannich reaction often requires harsh conditions that can compromise the stability of the sensitive lactone ring, leading to complex impurity profiles that are difficult to purge during downstream processing. The inability to introduce longer alkyl chains or functionalized side chains limits the ability of medicinal chemists to fine-tune the lipophilicity and water solubility balance of the final drug substance. Consequently, many potential candidates with superior therapeutic indices remain inaccessible using these legacy synthetic routes, creating a bottleneck in the pipeline for new anticancer agents. Supply chain managers also face challenges with the reproducibility of these older methods when attempting to scale up for commercial manufacturing due to inconsistent yields and purification difficulties.
The Novel Approach
The methodology outlined in patent CN1616460A overcomes these historical constraints by employing a Claisen rearrangement mechanism to install alkyl groups at the 9-position with remarkable versatility. This novel approach begins with the etherification of 10-hydroxycamptothecin with substituted allyl bromides, creating a stable intermediate that undergoes a thermal [3,3]-sigmatropic rearrangement to shift the allyl moiety to the carbon framework. This strategic shift allows for the introduction of a wide range of substituents, including straight-chain alkyls, branched alkyls, and even functionalized groups containing hydroxyl or amino functionalities. The process avoids the use of heavy metal catalysts in the key bond-forming step, relying instead on thermal energy in high-boiling polar aprotic solvents like DMF or DMSO. This results in a cleaner reaction profile with fewer metal-related impurities, significantly simplifying the purification workflow and reducing the overall cost of goods sold. For procurement teams, this translates to a more robust supply chain with reduced reliance on expensive transition metal catalysts and complex removal steps.
Mechanistic Insights into Claisen Rearrangement and Hydrogenation
The core chemical transformation driving this synthesis is the Claisen rearrangement, a pericyclic reaction that proceeds through a concerted cyclic transition state to form carbon-carbon bonds with high regioselectivity. In this specific application, the allyl ether formed at the 10-position oxygen atom rearranges upon heating to place the allyl group at the adjacent 9-position on the aromatic ring. This mechanism is highly advantageous because it does not generate ionic intermediates that could lead to polymerization or side reactions common in electrophilic aromatic substitutions. The reaction conditions are carefully controlled using solvents with high boiling points to ensure sufficient thermal energy is available to overcome the activation barrier of the rearrangement without degrading the sensitive camptothecin core. Following the rearrangement, the resulting 9-allyl intermediate possesses a double bond that can be further manipulated to introduce saturation or additional functional groups. 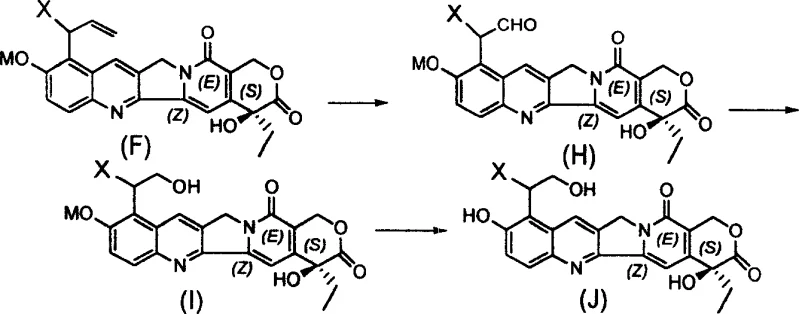
Impurity control is a critical aspect of this mechanism, particularly regarding the stability of the lactone ring under the reaction conditions. The use of mild bases during the initial etherification step, such as potassium carbonate, ensures that the phenolic hydroxyl is deprotonated without attacking the lactone carbonyl. During the subsequent hydrogenation step, catalysts like palladium on carbon are selected for their chemoselectivity, reducing the alkene double bond while leaving the lactone and other sensitive functional groups intact. This selectivity is paramount for maintaining the optical purity of the S-configuration at the 20-position, which is essential for topoisomerase I binding affinity. Any racemization at this center would render the compound inactive, so the process parameters are tightly defined to prevent epimerization. For quality control laboratories, this means that the critical quality attributes of the final API intermediate can be consistently met with minimal batch-to-batch variation.
How to Synthesize 9-Alkyl-10-Hydroxycamptothecin Efficiently
The synthesis of these high-value intermediates follows a logical three-step sequence that balances reaction efficiency with operational simplicity for industrial scale-up. The process begins with the alkylation of the starting material, followed by the thermal rearrangement, and concludes with a catalytic reduction to finalize the side chain structure. Each step has been optimized in the patent examples to demonstrate feasibility on a gram scale, providing a clear roadmap for process chemists to adapt the conditions for larger reactor volumes. The detailed standardized synthesis steps see the guide below for specific operational parameters and stoichiometry.
- Perform etherification of 10-hydroxycamptothecin with substituted allyl bromide in the presence of a base like potassium carbonate to form the allyl ether intermediate.
- Execute a thermal Claisen rearrangement in high-boiling solvents such as DMF to shift the allyl group from the 10-oxygen to the 9-carbon position.
- Conduct catalytic hydrogenation using palladium on carbon or Raney nickel to saturate the allyl double bond, yielding the final 9-alkyl-10-hydroxycamptothecin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages in terms of cost structure and supply chain resilience for pharmaceutical manufacturers. The elimination of complex transition metal catalysis in the key carbon-carbon bond forming step reduces the dependency on scarce and expensive precious metals, which are subject to significant market volatility. This shift towards thermal rearrangement chemistry simplifies the waste stream profile, as there are no heavy metal residues that require specialized and costly disposal procedures, aligning with increasingly stringent environmental regulations. For supply chain heads, the use of commodity chemicals like allyl bromide and common solvents ensures that raw material availability is not a bottleneck, even during periods of global supply disruption. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in temperature or mixing, leading to higher batch success rates and more predictable delivery schedules.
- Cost Reduction in Manufacturing: The process significantly lowers manufacturing costs by removing the need for expensive transition metal catalysts and the associated downstream purification steps required to meet residual metal specifications. By utilizing thermal energy rather than catalytic reagents for the key structural modification, the overall reagent cost per kilogram of product is drastically reduced. Furthermore, the high selectivity of the Claisen rearrangement minimizes the formation of byproducts, which increases the overall yield and reduces the volume of solvents and materials needed for chromatographic purification. This efficiency translates directly into a more competitive pricing structure for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 10-hydroxycamptothecin and substituted allyl bromides ensures a stable and continuous supply of raw materials from multiple global sources. Unlike processes that depend on specialized ligands or custom-synthesized catalysts, this route uses commodity chemicals that are produced in high volumes by the bulk chemical industry. This diversification of the supply base mitigates the risk of single-source failures and allows procurement managers to negotiate better terms with vendors. Additionally, the simplicity of the unit operations facilitates technology transfer between manufacturing sites, ensuring that production can be maintained even if one facility faces operational challenges.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable because it avoids hazardous reagents and extreme pressure conditions that often limit batch sizes in fine chemical production. The use of standard solvents like DMF and dioxane allows for the utilization of existing glass-lined or stainless steel reactors without the need for specialized equipment investments. From an environmental standpoint, the absence of heavy metal waste simplifies the effluent treatment process, reducing the environmental footprint of the manufacturing site. This compliance with green chemistry principles not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these camptothecin derivatives based on the patent specifications. These answers are derived from the detailed experimental data and mechanistic descriptions provided in the intellectual property documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is crucial for making informed decisions about process adoption and supply partnerships.
Q: What is the primary advantage of the Claisen rearrangement method over the Mannich reaction for camptothecin modification?
A: The Mannich reaction is limited to introducing only one-carbon methyl groups at the 9-position, whereas the Claisen rearrangement strategy described in patent CN1616460A allows for the introduction of diverse alkyl chains ranging from C1 to C6, including alkenyl, alkynyl, and substituted alkyl groups, significantly expanding the chemical space for structure-activity relationship studies.
Q: How is the 10-position hydroxyl group managed during the synthesis of these derivatives?
A: The 10-position hydroxyl group can be selectively protected as an ether, such as a methoxy group, during the alkylation steps to prevent side reactions, and can subsequently be deprotected using demethylating agents like hydrobromic acid or trimethylsilyl iodide to restore the active phenolic hydroxyl moiety required for topoisomerase I inhibition.
Q: What are the scalability considerations for the hydrogenation step in this process?
A: The hydrogenation step utilizes conventional catalysts such as palladium on carbon or Raney nickel under standard pressure or atmospheric hydrogen conditions, which are well-established unit operations in fine chemical manufacturing, ensuring that the process can be safely scaled from gram-level laboratory synthesis to multi-ton commercial production without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Alkyl-10-Hydroxycamptothecin Supplier
NINGBO INNO PHARMCHEM stands ready to support your development and commercialization needs with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in handling complex heterocyclic chemistry, ensuring that the stringent purity specifications required for oncology intermediates are consistently met through our rigorous QC labs. We understand the critical nature of supply continuity in the pharmaceutical sector and have established robust protocols to maintain quality and delivery performance even under demanding timelines. Our facility is equipped to handle the specific solvent and thermal requirements of the Claisen rearrangement process safely and efficiently.
We invite you to contact our technical procurement team to discuss your specific requirements and request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that can identify further opportunities to optimize your supply chain for this critical intermediate. Let us help you accelerate your drug development timeline with a reliable and high-quality supply of 9-alkyl-10-hydroxycamptothecin derivatives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
