Scalable Green Synthesis of 2-(N,N-Disubstituted Amino)-4-Thiazolinone Intermediates
Scalable Green Synthesis of 2-(N,N-Disubstituted Amino)-4-Thiazolinone Intermediates
The pharmaceutical and agrochemical industries are increasingly demanding sustainable manufacturing processes that do not compromise on yield or purity. Patent CN102408392A introduces a groundbreaking green preparation method for synthesizing 2-(N,N-disubstituted amino)-4-thiazolinone derivatives directly in an aqueous phase. This technology represents a significant departure from traditional organic solvent-dependent routes, utilizing water as the primary reaction medium and sodium acetate as a mild, cost-effective catalyst. By operating at moderate temperatures between 80-90°C, this method achieves exceptional conversion rates while adhering to strict green chemistry principles. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this aqueous protocol offers a compelling solution for producing high-value heterocyclic building blocks with minimal environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(N,N-disubstituted amino)-4-thiazolinone compounds has relied heavily on volatile organic solvents such as dichloromethane, toluene, or ethanol. These conventional methodologies present substantial drawbacks, including high flammability risks, significant toxicity to operators, and complex waste disposal requirements that drive up operational expenditures. Furthermore, traditional routes often suffer from lower yields and require harsh reaction conditions or expensive transition metal catalysts that necessitate rigorous downstream purification to meet stringent purity specifications. The reliance on organic media also complicates the isolation of the final product, often requiring energy-intensive distillation processes to recover solvents, which contradicts modern sustainability goals in fine chemical manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique properties of water to facilitate the cyclization reaction efficiently and safely. By employing sodium acetate as a catalyst in an aqueous environment, the reaction proceeds smoothly at 80-90°C, eliminating the need for hazardous organic solvents entirely. This shift not only drastically simplifies the workup procedure—allowing for straightforward extraction and neutralization—but also significantly reduces the cost of raw materials and waste treatment. The method demonstrates remarkable versatility across various N,N-disubstituted thiourea substrates, consistently delivering yields between 83% and 93%. This robustness makes it an ideal candidate for cost reduction in agrochemical intermediate manufacturing and pharmaceutical applications where supply chain reliability is paramount.
Mechanistic Insights into Aqueous Phase Cyclization
The core of this synthetic strategy involves a nucleophilic substitution followed by an intramolecular cyclization, facilitated by the polar nature of the aqueous medium. Initially, the sulfur atom of the N,N-disubstituted thiourea acts as a nucleophile, attacking the electrophilic alpha-carbon of the ethyl chloroacetate. The presence of water enhances the solubility of the ionic intermediates and stabilizes the transition states, thereby accelerating the reaction kinetics without the need for aggressive heating. Sodium acetate serves a dual role, acting as a weak base to deprotonate the thiourea nitrogen and buffering the system to prevent hydrolysis of the ester group prematurely. This delicate balance ensures that the cyclization to form the thiazolinone ring occurs selectively, minimizing the formation of unwanted byproducts.
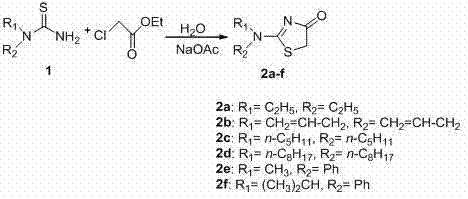
Following the initial substitution, the intermediate undergoes rapid cyclization to form the stable five-membered thiazolinone ring structure. The aqueous environment plays a critical role in impurity control by keeping inorganic salts and polar side products in the water phase, while the organic product can be easily extracted into ethyl acetate. This inherent partitioning capability simplifies the purification process, allowing for high-purity isolation after a simple drying step with anhydrous magnesium sulfate and column chromatography. Understanding this mechanism is vital for scaling up the process, as it confirms that the reaction is not diffusion-limited and can be effectively managed in large-scale reactors with standard agitation systems. The result is a clean reaction profile that supports the production of high-purity OLED material precursors or bioactive pharmaceutical intermediates.
How to Synthesize 2-(N,N-Disubstituted Amino)-4-Thiazolinone Efficiently
Implementing this green synthesis route requires precise control over stoichiometry and temperature to maximize yield and minimize waste. The standard protocol involves dissolving the N,N-disubstituted thiourea and sodium acetate in water, heating the mixture to 80-90°C to ensure complete dissolution before introducing the ethyl chloroacetate. Maintaining the temperature within this narrow window is crucial for driving the reaction to completion within 1 to 2 hours while preventing thermal degradation of the sensitive heterocyclic product. For detailed operational parameters and specific molar ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Dissolve N,N-disubstituted thiourea and sodium acetate in water by heating in an oil bath at 80-90°C.
- Add ethyl chloroacetate to the mixture and react under magnetic stirring at 80-90°C for 1-2 hours.
- Cool to room temperature, neutralize with sodium carbonate, extract with ethyl acetate, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the transition to this aqueous-based synthesis offers profound advantages for supply chain stability and cost management. By replacing expensive and regulated organic solvents with water, manufacturers can achieve substantial cost savings in both raw material procurement and hazardous waste disposal. The simplicity of the workup procedure reduces the overall processing time, allowing for faster turnaround times and improved responsiveness to market demand fluctuations. Additionally, the use of non-toxic reagents enhances workplace safety, potentially lowering insurance premiums and regulatory compliance burdens associated with volatile organic compound (VOC) emissions.
- Cost Reduction in Manufacturing: The elimination of costly organic solvents and transition metal catalysts directly lowers the bill of materials for every batch produced. Since water is the primary solvent, the expense associated with solvent recovery and recycling infrastructure is virtually eliminated, leading to a leaner cost structure. Furthermore, the high yields reported (83-93%) mean that less raw material is wasted, optimizing the atom economy and further driving down the unit cost of the final active ingredient. This economic efficiency is critical for maintaining competitiveness in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The raw materials required for this process, specifically N,N-disubstituted thioureas and ethyl chloroacetate, are commodity chemicals with robust and established global supply chains. Unlike specialized catalysts that may face sourcing bottlenecks, these inputs are readily available from multiple vendors, ensuring continuity of supply even during market disruptions. The simplified process flow also reduces the risk of production delays caused by complex purification steps or equipment failures related to solvent handling, thereby guaranteeing consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: This method is inherently designed for industrial scale-up, as it avoids the safety hazards associated with large volumes of flammable solvents. The aqueous system allows for easier heat management in large reactors, reducing the risk of thermal runaway incidents. Moreover, the process aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions and wastewater treatment, future-proofing the manufacturing facility against evolving compliance standards. This sustainability profile adds significant value for end-users who prioritize green chemistry in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Whether you are evaluating this route for pilot plant trials or full commercial production, these answers highlight the key operational benefits and technical feasibility of the method.
Q: What are the primary advantages of using water as a solvent in this thiazolinone synthesis?
A: Using water eliminates the need for volatile and flammable organic solvents, significantly reducing environmental impact and safety hazards while simplifying the workup procedure through easy phase separation.
Q: What yield range can be expected from this aqueous preparation method?
A: According to patent CN102408392A, the method consistently achieves high yields ranging from 83% to 93%, demonstrating superior efficiency compared to traditional organic solvent-based routes.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes inexpensive raw materials like sodium acetate and water, operates at moderate temperatures (80-90°C), and avoids complex purification steps, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(N,N-Disubstituted Amino)-4-Thiazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic routes to meet the evolving needs of the global pharmaceutical and agrochemical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this aqueous cyclization can be seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-(N,N-disubstituted amino)-4-thiazolinone performs reliably in your downstream applications.
We invite you to collaborate with us to leverage this green technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can optimize your supply chain and reduce your overall manufacturing costs.
