Revolutionizing Bisoprolol Synthesis: A High-Yield Mitsunobu Strategy for Commercial Scale-Up
Revolutionizing Bisoprolol Synthesis: A High-Yield Mitsunobu Strategy for Commercial Scale-Up
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for more efficient and cost-effective manufacturing processes for critical active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in patent CN112778142B, which discloses a novel preparation method for Bisoprolol free base, the key precursor to the widely prescribed beta-1 blocker Bisoprolol fumarate. This technology represents a paradigm shift from traditional, labor-intensive multi-step syntheses to a streamlined, high-efficiency coupling strategy. By leveraging a modified Mitsunobu reaction protocol, this innovation addresses long-standing industry challenges regarding yield optimization, impurity control, and operational complexity. For R&D directors and procurement managers alike, understanding the mechanistic underpinnings and commercial implications of this patent is essential for securing a competitive edge in the global supply chain of hypertension and angina medications.
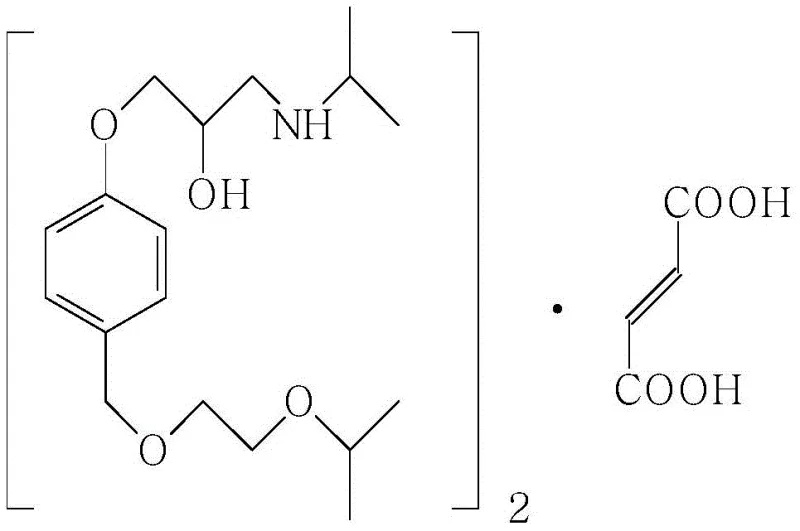
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bisoprolol has been plagued by inefficiencies inherent in linear, multi-step pathways. As illustrated in prior art, traditional routes typically commence with 4-hydroxybenzaldehyde, necessitating a sequence of reduction, alcoholic hydroxyl etherification, phenolic hydroxyl etherification, amination, and finally salification. This convoluted approach not only extends the production cycle but also introduces multiple points of failure where yield attrition occurs. Data from existing literature indicates that such conventional methods often result in dismal total yields ranging merely from 15% to 27%, primarily due to the accumulation of side products and the rigorous purification requirements at each stage. Furthermore, older methodologies frequently rely on high-vacuum distillation for purification, a process that is energy-intensive and poses significant safety risks on a large scale. The inability of these legacy processes to meet modern purity standards, often detecting dozens of related impurities upon sensitive analysis, renders them increasingly obsolete in a regulatory environment that demands exceptional quality control.
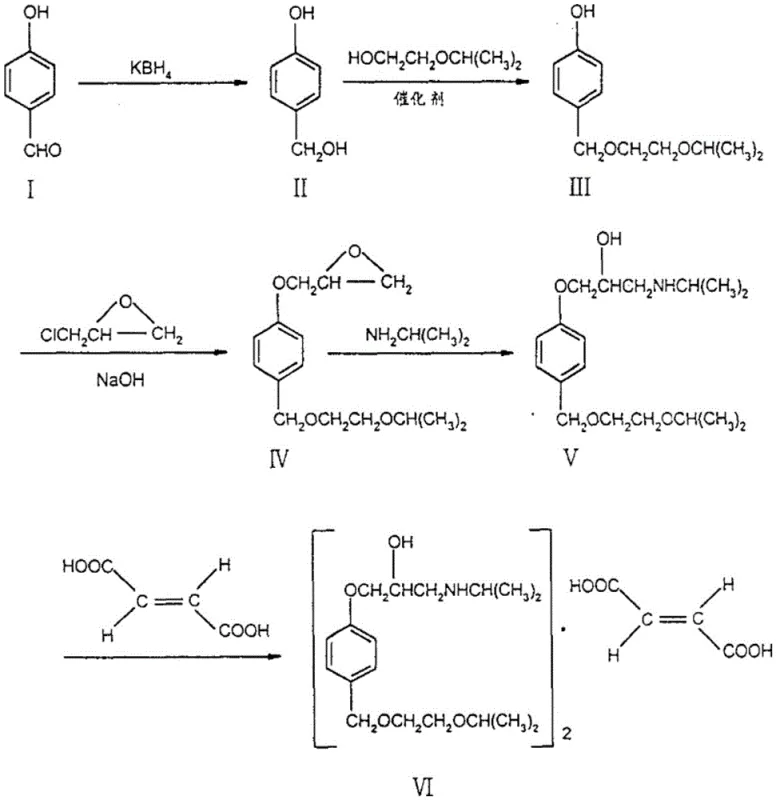
The Novel Approach
In stark contrast to the cumbersome legacy protocols, the methodology outlined in patent CN112778142B introduces a convergent synthetic strategy that dramatically condenses the process timeline. This innovative route utilizes 4-isopropoxy ethoxy methyl phenol and 3-isopropylamino-1, 2-propylene glycol as advanced starting materials, effectively bypassing several intermediate steps required in traditional syntheses. By employing a reaction mediating agent alongside a specific Mitsunobu reagent system, the process achieves a direct etherification that constructs the core carbon-oxygen bond with remarkable precision. This strategic simplification does more than just save time; it fundamentally alters the economic equation of production by minimizing unit operations and reducing the consumption of solvents and reagents. The result is a robust manufacturing process that delivers superior product quality with significantly reduced operational overhead, positioning it as an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolios.
Mechanistic Insights into Polymer-Supported Mitsunobu Etherification
The core of this technological breakthrough lies in the sophisticated application of the Mitsunobu reaction, specifically tailored through the use of polymer-supported reagents to enhance selectivity and simplify workup. In this system, triphenylphosphine-polystyrene resin (ps-TPP) serves as the crucial mediator, functioning not merely as a catalyst but as a solid-phase scavenger that dictates the reaction trajectory. The presence of the bulky polystyrene backbone introduces significant steric hindrance around the phosphorus center, which kinetically favors the reaction of the primary alcohol over the secondary hydroxyl group on the propylene glycol derivative. This regioselectivity is paramount for ensuring the formation of the desired ether linkage without generating unwanted isomeric byproducts that would complicate downstream purification. Furthermore, the use of N, N, N', N'-tetramethyl azodicarbonamide (TMAD) as the oxidant provides a distinct advantage due to its unique solubility profile, which complements the solid-phase nature of the phosphine resin to create a homogeneous yet easily separable reaction environment.
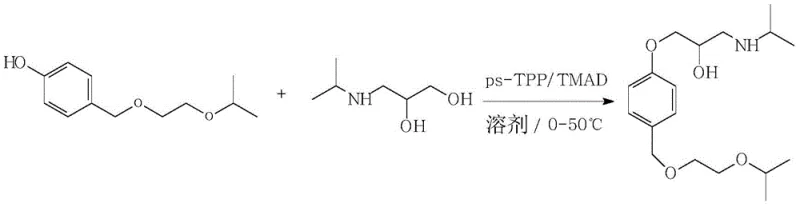
Beyond the reaction kinetics, the post-reaction processing mechanism offers profound implications for impurity control and operational efficiency. In classical Mitsunobu reactions utilizing liquid triphenylphosphine, the removal of the resulting triphenylphosphine oxide byproduct is notoriously difficult, often requiring column chromatography which is impractical for ton-scale production. However, the utilization of ps-TPP in this patented process transforms this challenge into a simple mechanical operation; upon completion of the reaction, the spent resin containing the phosphine oxide can be removed via straightforward filtration. This physical separation method effectively eliminates a major source of contamination and avoids the need for complex solvent exchanges or extensive washing procedures. Additionally, the water solubility of the TMAD byproducts allows for their easy removal during the aqueous extraction phase, ensuring that the final organic phase contains the Bisoprolol free base with exceptional purity levels exceeding 99%, thereby meeting the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Bisoprolol Free Base Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing chemical manufacturing infrastructure, requiring only standard reactor setups and common laboratory techniques adapted for scale. The process begins with the dissolution of the phenolic substrate and the amino-diol in a suitable solvent such as tetrahydrofuran or dichloromethane, followed by the controlled addition of the solid resin and the azodicarbonamide reagent under inert atmosphere. Maintaining the reaction temperature within the mild range of 0-50°C ensures optimal conversion while minimizing thermal degradation of sensitive functional groups. Following the reaction period, the workflow transitions to a simplified workup involving filtration and liquid-liquid extraction, avoiding the need for specialized distillation equipment. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and step-by-step operational parameters, please refer to the standardized guide below.
- React 4-isopropoxy ethoxy methyl phenol with 3-isopropylamino-1,2-propylene glycol in a solvent like THF or DCM, using triphenylphosphine resin and TMAD.
- Upon completion, filter the reaction mixture to remove the solid triphenylphosphine oxide resin byproduct.
- Extract the filtrate with water and an organic solvent, then concentrate the organic phase to isolate the pure Bisoprolol free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible strategic benefits that extend far beyond simple chemical yield. The elimination of high-vacuum distillation steps, which are characteristic of older methods, represents a substantial reduction in energy consumption and capital equipment maintenance costs. By shifting from a multi-step linear process to a convergent one-step coupling, manufacturers can drastically reduce the lead time for high-purity pharmaceutical intermediates, allowing for more responsive inventory management and faster time-to-market for finished drug products. This agility is crucial in a volatile global market where supply continuity is often threatened by logistical bottlenecks and raw material shortages. Furthermore, the simplified purification protocol reduces the dependency on specialized chromatographic resins and excessive solvent usage, aligning production practices with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The transition to a polymer-supported reagent system fundamentally alters the cost structure of Bisoprolol production by removing the most expensive and time-consuming purification stages. Traditional methods relying on distillation or chromatography incur high operational expenditures due to energy usage and solvent loss; in contrast, the filtration-based workup of this new method minimizes utility costs and maximizes solvent recovery rates. Additionally, the dramatic improvement in overall yield from roughly 20% to over 90% means that significantly less raw material is required to produce the same amount of final product, directly lowering the cost of goods sold (COGS) and improving margin potential for generic manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of this synthetic route, which utilizes readily available starting materials and avoids the use of exotic or highly regulated catalysts that might face supply constraints. The simplicity of the reaction conditions, operating at near-ambient temperatures and pressures, reduces the risk of batch failures due to equipment malfunction or operator error, ensuring a consistent and predictable output of critical intermediates. This reliability allows supply chain planners to maintain leaner safety stocks without compromising production schedules, thereby freeing up working capital and reducing warehousing costs associated with holding large inventories of hazardous chemicals.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process offers a clear pathway for commercial scale-up of complex pharmaceutical intermediates without the proportional increase in waste generation seen in traditional routes. The ability to filter off solid byproducts rather than generating liquid waste streams laden with phosphine oxides simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility. This 'green chemistry' aspect not only facilitates easier regulatory approval in jurisdictions with strict emission standards but also future-proofs the supply chain against tightening environmental legislation, ensuring long-term operational viability and corporate social responsibility compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Bisoprolol synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on improved quality and cost metrics.
Q: How does the new Mitsunobu route improve yield compared to traditional methods?
A: Traditional multi-step routes often suffer from cumulative yield losses, resulting in total yields as low as 15-27%. The novel one-step Mitsunobu coupling described in patent CN112778142B achieves yields exceeding 90%, drastically reducing material waste.
Q: What are the purification advantages of using triphenylphosphine resin?
A: Unlike liquid triphenylphosphine which requires complex chromatographic separation, the polymer-supported resin (ps-TPP) allows for simple filtration to remove phosphine oxide byproducts, significantly simplifying post-treatment and lowering production costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process eliminates the need for high-vacuum distillation and uses mild reaction temperatures (0-50°C), making it highly scalable, safer, and more energy-efficient for commercial manufacturing of cardiovascular intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisoprolol Free Base Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN112778142B requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield Mitsunobu process are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific requirements of polymer-supported reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Bisoprolol free base meets the highest international standards for cardiovascular drug manufacturing.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this optimized route for your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
