Scalable Purification Technology for High-Purity Vitamin A Intermediates
Introduction to Advanced Purification Technologies
The synthesis of high-value pharmaceutical intermediates often faces significant bottlenecks during the purification stage, particularly when dealing with thermally sensitive or high-boiling compounds. Patent CN111269121A introduces a transformative purification method specifically designed for 8-oxo-3,7-dimethyl-octadienyl carboxylate compounds, which serve as critical precursors in the industrial production of Vitamin A, carotenoids, and Coenzyme Q. Traditional methods relying on high-vacuum rectification or column chromatography have historically imposed heavy burdens on manufacturing efficiency and equipment costs. This new technical approach leverages the reversible formation of alkali metal alpha-hydroxysulfonates to achieve superior separation of by-products and raw material impurities under normal pressure conditions. By shifting from physical separation based on boiling points to chemical separation based on solubility and reactivity, this innovation offers a robust pathway for producing high-purity intermediates suitable for stringent pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of 8-oxo-3,7-dimethyl-octadienyl carboxylate compounds has been plagued by inefficiencies inherent to thermal separation techniques. Prior art methods, such as those described in patents JPH078827B2 and EP0234496B2, typically yield crude products contaminated with unreacted hydroxyl-containing compounds, excessive oxidation by-products like carboxylic acids, and residual chlorides. To isolate the target molecule, manufacturers were forced to employ reduced pressure rectification under high vacuum conditions. This process is not only energy-intensive and time-consuming but also requires sophisticated, expensive equipment capable of maintaining extreme vacuum levels. Furthermore, the high boiling point of the target compound makes it exceptionally difficult to separate impurities with similar vapor pressures, resulting in low distillation efficiency and compromised chemical purity. In cases where column chromatography was utilized to achieve necessary purity levels, the process became entirely unsuitable for large-scale industrial production due to prohibitive solvent consumption and low throughput.
The Novel Approach
The methodology disclosed in CN111269121A circumvents these thermal limitations by utilizing a chemical derivatization strategy. The core of this innovation involves reacting the crude 8-oxo-3,7-dimethyl-octadienyl carboxylate with an aqueous solution of alkali metal bisulfite in the presence of a water-miscible lower aliphatic alcohol. This reaction selectively converts the target aldehyde functionality into a water-soluble alpha-hydroxysulfonate salt, effectively pulling the desired product into the aqueous phase while leaving lipophilic impurities and non-reactive by-products in the organic phase or as solids. 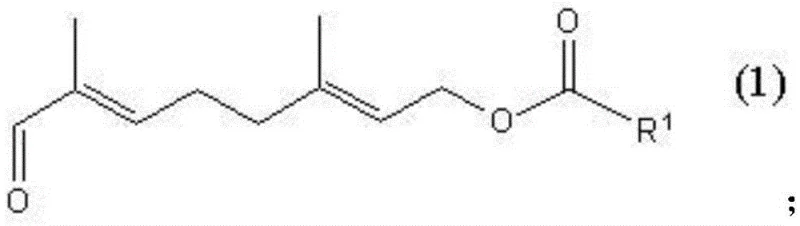 This phase separation allows for the removal of complex impurity profiles without the need for high-energy distillation. The process operates under normal atmospheric pressure, significantly reducing equipment requirements and operational hazards associated with high-vacuum systems. Following the separation of the aqueous layer, the pure target compound is regenerated through a decomposition reaction, yielding a product with markedly improved chemical purity suitable for downstream synthesis of vitamins and fine chemicals.
This phase separation allows for the removal of complex impurity profiles without the need for high-energy distillation. The process operates under normal atmospheric pressure, significantly reducing equipment requirements and operational hazards associated with high-vacuum systems. Following the separation of the aqueous layer, the pure target compound is regenerated through a decomposition reaction, yielding a product with markedly improved chemical purity suitable for downstream synthesis of vitamins and fine chemicals.
Mechanistic Insights into Bisulfite Adduct Formation and Decomposition
The efficacy of this purification route relies on the specific reactivity of the aldehyde group within the 8-oxo-3,7-dimethyl-octadienyl framework. In the first step, the nucleophilic attack of the bisulfite ion on the carbonyl carbon forms a stable addition product. This reaction proceeds rapidly only in water-miscible solvents like methanol or ethanol, which facilitate the interaction between the organic substrate and the aqueous bisulfite species. The resulting alkali metal alpha-hydroxysulfonate, depicted in Formula II, possesses distinct solubility characteristics compared to the starting material. 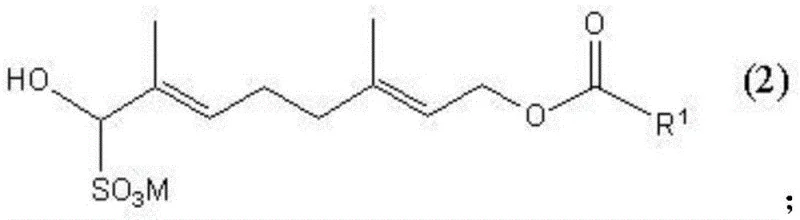 Because the sulfonate salt is highly soluble in water but insoluble in organic solvents, a simple liquid-liquid extraction can effectively partition the target molecule away from neutral organic impurities, unreacted starting materials, and non-polar by-products. This chemical selectivity is far superior to physical separation methods when dealing with structurally similar contaminants that share close boiling points.
Because the sulfonate salt is highly soluble in water but insoluble in organic solvents, a simple liquid-liquid extraction can effectively partition the target molecule away from neutral organic impurities, unreacted starting materials, and non-polar by-products. This chemical selectivity is far superior to physical separation methods when dealing with structurally similar contaminants that share close boiling points.
The regeneration of the purified compound is achieved in the second step by disrupting the sulfonate adduct. This can be accomplished by introducing an acidic substance, a basic substance, or a competing water-soluble aldehyde into the aqueous layer in the presence of a water-immiscible organic solvent. For instance, adding hydrochloric acid or sodium hydroxide shifts the equilibrium, decomposing the sulfonate back into the free aldehyde and bisulfite ions. The liberated 8-oxo-3,7-dimethyl-octadienyl carboxylate, now free from the impurities left behind in the aqueous waste stream, partitions into the organic solvent layer. This mechanism ensures that the final product is not only chemically pure but also retains its stereochemical integrity, as the mild reaction conditions prevent the racemization of optically active isomers often seen in harsh thermal processes.
How to Synthesize 8-Oxo-3,7-Dimethyl-Octadienyl Carboxylate Efficiently
Implementing this purification protocol requires precise control over solvent selection and stoichiometric ratios to maximize recovery and purity. The process begins by dissolving the crude ester in a lower aliphatic alcohol, preferably methanol, and treating it with a concentrated aqueous solution of sodium or potassium bisulfite. The reaction mixture is maintained at moderate temperatures, typically between 25°C and 70°C, to ensure complete conversion to the sulfonate adduct without degrading the sensitive diene system. Once the aqueous layer containing the adduct is isolated, it is subjected to a decomposition step using reagents such as formaldehyde or dilute mineral acids in a biphasic system with dichloromethane or ethyl acetate. Detailed standardized synthesis steps and specific operating parameters for this efficient purification route are provided below.
- React crude 8-oxo-3,7-dimethyl-octadienyl carboxylate with aqueous alkali metal bisulfite in a water-miscible lower aliphatic alcohol solvent to form a water-soluble alpha-hydroxysulfonate intermediate.
- Separate the aqueous layer containing the sulfonate adduct from organic impurities and unreacted starting materials using a water-immiscible organic solvent extraction.
- Decompose the purified aqueous sulfonate layer using an acidic, basic, or aldehyde reagent in the presence of a water-immiscible organic solvent to regenerate and extract the purified target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology represents a significant opportunity to optimize manufacturing costs and secure supply continuity. By eliminating the dependency on high-vacuum distillation columns and chromatography resins, the process drastically simplifies the production infrastructure required for Vitamin A intermediates. This simplification translates directly into reduced capital expenditure for specialized equipment and lower maintenance overheads. Furthermore, the ability to operate under normal pressure enhances plant safety and reduces the risk of production stoppages due to equipment failure, thereby ensuring a more reliable supply of critical pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of energy-intensive high-vacuum distillation steps leads to substantial savings in utility costs and processing time. Additionally, the replacement of expensive chromatographic purification with simple liquid-liquid extraction significantly lowers solvent consumption and waste disposal costs. The use of common, inexpensive reagents like sodium bisulfite and methanol further drives down the raw material cost profile compared to specialized oxidants or catalysts used in alternative routes.
- Enhanced Supply Chain Reliability: The robustness of this chemical purification method allows for consistent batch-to-batch quality, reducing the rejection rates that often plague complex synthetic sequences. Since the process does not rely on scarce or highly specialized separation media, the supply chain is less vulnerable to disruptions in the availability of specific consumables. The scalability of the liquid-liquid extraction process ensures that production volumes can be increased seamlessly to meet market demand without requiring proportional increases in facility footprint.
- Scalability and Environmental Compliance: Operating at normal pressure and moderate temperatures reduces the environmental footprint of the manufacturing process by lowering energy consumption. The aqueous waste streams generated are primarily composed of inorganic salts and alcohols, which are easier to treat and manage compared to the complex organic waste mixtures associated with column chromatography. This alignment with green chemistry principles facilitates easier regulatory compliance and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the practical application of the bisulfite adduct method for industrial stakeholders.
Q: Why is bisulfite adduct formation preferred over vacuum distillation for this intermediate?
A: Vacuum distillation of 8-oxo-3,7-dimethyl-octadienyl carboxylates requires high vacuum conditions due to high boiling points, leading to low efficiency and difficulty removing close-boiling impurities. The bisulfite method operates at normal pressure and chemically separates the target molecule from structural analogs.
Q: What solvents are critical for the success of this purification method?
A: The process requires a water-miscible lower aliphatic alcohol, such as methanol or ethanol, for the initial adduct formation to ensure rapid reaction kinetics. Subsequent extraction utilizes water-immiscible solvents like dichloromethane or ethyl acetate.
Q: Does this method preserve optical activity in chiral isomers?
A: Yes, the purification method is mild and does not involve harsh thermal conditions that typically cause racemization. If the crude starting material contains optically active isomers, the purified product retains its optical activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Oxo-3,7-Dimethyl-Octadienyl Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the global supply chain for vitamins and nutraceuticals. Our technical team has extensively evaluated the purification methodology described in CN111269121A and integrated its principles into our manufacturing protocols to ensure superior product quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 8-oxo-3,7-dimethyl-octadienyl carboxylate meets the exacting standards required for pharmaceutical synthesis.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for Vitamin A precursors. By leveraging our expertise in process optimization and cost-effective manufacturing, we can help your organization achieve significant efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
