Revolutionizing Methylthio Heterocycle Production: A Green Catalytic Breakthrough for Global Supply Chains
Revolutionizing Methylthio Heterocycle Production: A Green Catalytic Breakthrough for Global Supply Chains
The global demand for high-value heterocyclic intermediates continues to surge, driven by the relentless expansion of the pharmaceutical and agrochemical sectors. In this context, Patent CN101514190A introduces a transformative methodology for the preparation of methylthio heterocyclic compounds, addressing critical bottlenecks in traditional synthetic routes. This innovation leverages the unique physicochemical properties of room-temperature ionic liquids to facilitate the methylation of mercapto-containing heterocycles using dimethyl carbonate (DMC) as a green methylating agent. By shifting away from hazardous reagents like methyl iodide and dimethyl sulfate, this technology offers a pathway to significantly safer and more sustainable manufacturing processes. For R&D directors and procurement strategists, understanding the implications of this ionic liquid-catalyzed system is paramount for optimizing supply chain resilience and reducing regulatory burdens associated with toxic waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of methylthio heterocycles has relied heavily on aggressive methylating agents such as methyl iodide and dimethyl sulfate. While these reagents are chemically effective, they present severe operational and environmental challenges that modern manufacturing facilities can no longer ignore. Methyl iodide is not only prohibitively expensive but also classified as a highly toxic substance with significant volatility, posing acute health risks to operators and requiring complex containment systems. Similarly, dimethyl sulfate is a known carcinogen with extreme toxicity, necessitating rigorous safety protocols and expensive waste treatment infrastructure to neutralize residual hazards. Furthermore, conventional methods often suffer from moderate yields and poor selectivity, leading to complex purification steps that erode profit margins and extend production lead times. The reliance on volatile organic solvents in these traditional processes further exacerbates the environmental footprint, creating a compounding liability for companies striving to meet increasingly stringent global sustainability standards.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing dimethyl carbonate (DMC) in conjunction with ionic liquids, effectively mitigating the risks associated with legacy chemistries. Dimethyl carbonate is recognized as an environmentally benign reagent that decomposes into non-toxic byproducts, thereby eliminating the generation of hazardous halogenated waste streams. The integration of ionic liquids, specifically imidazolium-based salts like 1-butyl-3-methylimidazole chloride, serves a dual purpose as both the reaction medium and the catalyst. This dual functionality simplifies the reaction setup by removing the need for additional catalytic additives and allows for the operation under relatively mild thermal conditions ranging from 50°C to 150°C. Crucially, the ionic liquid phase can be easily separated from the organic product and recycled for subsequent batches, demonstrating exceptional stability over multiple cycles. This approach not only enhances the safety profile of the manufacturing process but also drives substantial operational efficiencies by streamlining workup procedures and minimizing solvent consumption.
Mechanistic Insights into Ionic Liquid-Catalyzed S-Methylation
The efficacy of this synthetic route lies in the unique interaction between the ionic liquid cation and the nucleophilic sulfur center of the mercapto-heterocycle. The ionic liquid creates a highly polar environment that stabilizes the transition state of the methylation reaction, facilitating the attack of the thiolate anion on the methyl group of dimethyl carbonate. Unlike traditional polar aprotic solvents, the ionic liquid does not merely solvate the reactants but actively participates in the catalytic cycle, likely through hydrogen bonding or electrostatic interactions that lower the activation energy barrier. This mechanistic advantage results in markedly improved substrate conversion rates, often reaching completion within 0.5 to 15 hours depending on the specific heterocyclic scaffold. The versatility of this system is evident in its ability to accommodate a wide range of substrates, including pyrimidines, benzothiazoles, and benzoxazoles, without compromising selectivity. By maintaining a precise molar ratio of substrate to DMC between 1:1.2 and 1:2, the process ensures that side reactions are minimized, yielding products with high purity profiles suitable for downstream pharmaceutical applications.
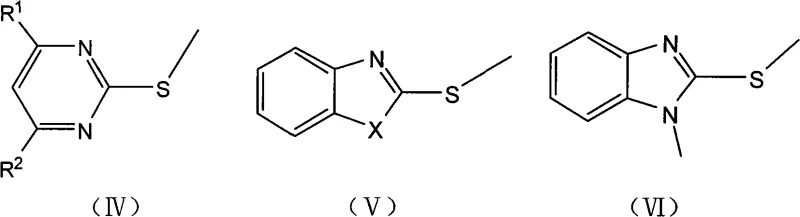
Impurity control is another critical aspect where this ionic liquid system outperforms conventional methodologies. In traditional S-methylation processes, the formation of N-methylated byproducts or over-methylated species is a common occurrence that complicates purification. However, the specific solvation environment provided by the ionic liquid appears to favor S-alkylation over N-alkylation, particularly for substrates like 2-mercaptobenzimidazole where tautomerism can lead to mixed products. The patent data indicates that even for challenging substrates requiring N-methylation alongside S-methylation, such as the conversion of 2-mercaptobenzimidazole to N-methyl-2-methylthiobenzimidazole, the reaction can be tuned by adjusting temperature and reaction time. This level of control is essential for producing high-purity intermediates where trace impurities could impact the efficacy or safety of the final active pharmaceutical ingredient (API). The ability to monitor reaction progress via TLC or HPLC allows for real-time optimization, ensuring that the process remains robust and reproducible across different scales of operation.
How to Synthesize Methylthio Heterocycles Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to both laboratory-scale optimization and industrial-scale production. The process begins with the dissolution of the mercapto-containing heterocyclic starting material directly into the ionic liquid, eliminating the need for preliminary solvent exchanges. Following the addition of dimethyl carbonate, the mixture is heated under stirring to the target temperature range, typically between 90°C and 130°C for optimal kinetics. Reaction monitoring is conducted using standard analytical techniques to determine the endpoint, after which the mixture is cooled to room temperature. The product is then extracted using a standard organic solvent such as ethyl acetate, leaving the ionic liquid phase intact for recovery and reuse. This simplified workflow reduces the number of unit operations required, thereby lowering capital expenditure and operational complexity. For detailed standardized synthesis steps and specific parameter optimization, please refer to the technical guide below.
- Dissolve the mercapto-containing heterocyclic compound in an ionic liquid such as 1-butyl-3-methylimidazole chloride.
- Add dimethyl carbonate in a molar ratio of 1: 1.2 to 1:2 relative to the substrate and heat to 50-150°C.
- Monitor reaction progress via TLC or HPLC, then extract the product with ethyl acetate and recover the ionic liquid solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-catalyzed process offers compelling strategic advantages that extend beyond simple yield improvements. The most significant benefit is the drastic reduction in regulatory and compliance costs associated with handling hazardous materials. By replacing methyl iodide and dimethyl sulfate with dimethyl carbonate, manufacturers can avoid the高昂 costs of specialized storage, transport, and disposal of toxic chemicals. This shift not only lowers the direct cost of goods sold but also mitigates the risk of supply chain disruptions caused by regulatory crackdowns on hazardous substances. Furthermore, the recyclability of the ionic liquid solvent creates a closed-loop system that significantly reduces raw material consumption over the long term. Although the initial investment in ionic liquids may be higher than traditional solvents, the ability to reuse the catalyst-solvent system for multiple cycles amortizes this cost, leading to substantial cumulative savings in large-scale production campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic methylating agents like methyl iodide directly impacts the bill of materials, while the removal of heavy metal catalysts or corrosive acids simplifies the purification train. This streamlined downstream processing reduces the consumption of auxiliary chemicals and energy, resulting in a leaner and more cost-effective manufacturing process. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which further lowers the cost associated with chromatography or recrystallization steps. These efficiencies collectively contribute to a more competitive pricing structure for the final intermediates, allowing suppliers to offer better value to their customers without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on green chemistry principles future-proofs the supply chain against evolving environmental regulations that frequently restrict the use of traditional reagents. Dimethyl carbonate is widely available and produced on a massive scale for other industries, ensuring a stable and secure supply source that is less prone to geopolitical volatility compared to specialized halogenated reagents. The robustness of the ionic liquid system also means that production schedules are less likely to be interrupted by safety incidents or equipment corrosion issues, which are common when handling aggressive acids or alkylating agents. This reliability is crucial for maintaining just-in-time delivery commitments to major pharmaceutical and agrochemical clients who demand consistent quality and uninterrupted supply.
- Scalability and Environmental Compliance: The process is inherently scalable due to the thermal stability of ionic liquids and the manageable exothermicity of the DMC methylation reaction. Unlike microwave-assisted methods or reactions requiring cryogenic conditions, this protocol operates under standard heating conditions that are easily replicated in multi-ton reactors. From an environmental perspective, the near-zero emission of volatile organic compounds (VOCs) and the absence of halogenated waste streams align perfectly with corporate sustainability goals and ISO 14001 standards. This alignment facilitates smoother permitting processes for new manufacturing lines and enhances the brand reputation of companies adopting these green technologies, making them preferred partners for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalysis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production portfolios. The answers highlight the balance between chemical performance and operational practicality that defines the value proposition of this method.
Q: What are the primary advantages of using ionic liquids over traditional solvents for methylation?
A: Ionic liquids serve a dual function as both catalyst and solvent, eliminating the need for volatile organic compounds (VOCs) and allowing for easy recovery and recycling, which significantly reduces waste generation.
Q: Is dimethyl carbonate a safe alternative to methyl iodide for industrial scale-up?
A: Yes, dimethyl carbonate is non-toxic and environmentally benign compared to methyl iodide and dimethyl sulfate, making it a much safer choice for large-scale manufacturing operations.
Q: Can this process be applied to benzothiazole and pyrimidine derivatives?
A: The patent explicitly demonstrates high conversion rates and selectivity for various substrates including 2-mercaptobenzothiazole, 2-mercaptobenzoxazole, and substituted pyrimidines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylthio Heterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory necessity but a strategic imperative for maintaining competitiveness in the global fine chemicals market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ionic liquid-catalyzed methylation described in CN101514190A can be seamlessly translated from the bench to the plant. We are committed to delivering high-purity pharmaceutical intermediates and agrochemical building blocks that meet the most stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our deep expertise in heterogeneous catalysis and green solvent systems, we help our partners navigate the complexities of process optimization to achieve maximum yield and minimal environmental impact.
We invite you to collaborate with us to explore how this advanced methylation technology can enhance your specific product portfolio and drive down your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Whether you are looking to secure a long-term supply of 2-methylthiopyrimidine derivatives or need assistance in developing a novel heterocyclic scaffold, we encourage you to reach out for specific COA data and route feasibility assessments. Let us partner with you to build a more resilient, sustainable, and profitable supply chain for the next generation of life-saving medicines and crop protection solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
