Advanced Dibromohydantoin Bromination for High-Purity Methyl 4-Bromomethylbenzoate Derivatives Manufacturing
The pharmaceutical and agrochemical industries constantly demand more efficient pathways for synthesizing critical building blocks, particularly benzylic bromides which serve as versatile electrophiles in coupling reactions. Patent CN112745216A introduces a significant technological advancement in the preparation of methyl 4-bromomethylbenzoate and its derivatives, addressing long-standing inefficiencies in traditional bromination protocols. This innovation centers on the strategic replacement of conventional brominating agents with dibromohydantoin, coupled with a radical initiation system that drastically enhances reaction kinetics and selectivity. For R&D directors and process chemists, this represents a pivotal shift towards greener, higher-yielding synthesis routes that minimize waste and maximize throughput. The method not only streamlines the production of these key intermediates but also ensures a purity profile that meets the stringent requirements of modern drug substance manufacturing, effectively bridging the gap between laboratory discovery and commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-bromomethyl benzoates has relied heavily on elemental bromine or N-bromosuccinimide (NBS), both of which present substantial operational and economic drawbacks in an industrial setting. Elemental bromine, while highly reactive, poses severe safety hazards due to its corrosive nature and volatility, often leading to uncontrollable poly-bromination side reactions that complicate downstream purification and reduce overall yield. Alternatively, NBS has been the standard for controlled radical bromination, yet it suffers from sluggish reactivity, frequently requiring prolonged reflux conditions extending up to 24 hours to achieve partial conversion. Data from prior art indicates that NBS-mediated reactions often stall with 20-40% of the starting material remaining unreacted, necessitating complex recycling loops or resulting in significant material loss. Furthermore, the stoichiometric inefficiency of NBS, which provides only one active bromine atom per molecule despite its molecular weight, inflates raw material costs and generates excessive succinimide waste that must be managed.
The Novel Approach
The disclosed invention overcomes these bottlenecks by employing dibromohydantoin as a superior brominating agent, activated by radical initiators such as azobisisobutyronitrile (AIBN) or benzoyl peroxide. This novel approach leverages the unique reactivity profile of dibromohydantoin, which possesses higher activity than NBS but avoids the aggressive, non-selective behavior of elemental bromine. By operating at moderate temperatures between 60°C and 70°C, the reaction achieves complete conversion within a remarkably short window of 3 to 5 hours, representing a five-fold increase in processing speed compared to traditional NBS methods. The structural integrity of the product is maintained with high fidelity, as evidenced by gas chromatography results showing no residual starting material and purity levels exceeding 99%. This method transforms the bromination step from a rate-limiting bottleneck into a highly efficient unit operation, enabling rapid iteration in process development and robust performance in commercial manufacturing.
Mechanistic Insights into Dibromohydantoin-Mediated Radical Bromination
The core of this technological breakthrough lies in the mechanistic efficiency of the radical chain reaction initiated by AIBN in the presence of dibromohydantoin. Upon thermal decomposition, AIBN generates cyanoisopropyl radicals which abstract hydrogen atoms from the hydantoin ring or directly interact with the substrate to initiate the radical chain. The dibromohydantoin molecule serves as a reservoir of bromine radicals, releasing them in a controlled manner that favors mono-bromination at the benzylic position. Unlike NBS, which can suffer from equilibrium limitations that slow down the propagation step, dibromohydantoin maintains a steady concentration of active bromine species, driving the reaction to completion. This kinetic advantage is crucial for preventing the formation of dibromo or tribromo byproducts, ensuring that the reaction stops selectively at the desired mono-brominated stage. The solvent system, typically utilizing dichloroethane or acetonitrile, further stabilizes the radical intermediates and ensures homogeneous mixing, which is vital for consistent heat transfer and reaction control on a large scale.
Impurity control is inherently built into this mechanism through the precise tuning of reagent stoichiometry and reaction temperature. By maintaining the molar ratio of dibromohydantoin to substrate between 0.5 and 0.6, the process ensures that there is just enough bromine source to convert the substrate without excess that could lead to over-bromination. The absence of inorganic bromides eliminates the formation of salt wastes and avoids the phase-transfer complications associated with heterogeneous reaction systems. Post-reaction workup involves a simple aqueous wash with sodium thiosulfate to quench any residual oxidative species, followed by brine washing to remove polar impurities. This streamlined purification protocol minimizes the risk of product degradation during isolation and ensures that the final crystalline product meets the rigorous specifications required for pharmaceutical intermediates, with H-NMR data confirming the absence of structural anomalies.
How to Synthesize Methyl 4-Bromomethylbenzoate Efficiently
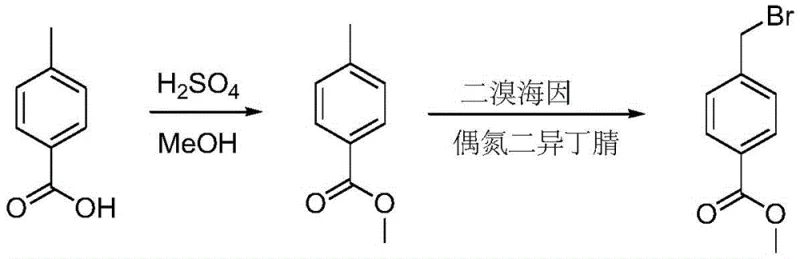
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity methyl 4-bromomethylbenzoate, starting from readily available p-toluic acid. The process begins with a classic acid-catalyzed esterification to generate the methyl ester, followed by the critical radical bromination step using the optimized dibromohydantoin system. Operators should note that the sequential addition of the radical initiator is key to maintaining a steady reaction rate and preventing thermal runaway. The detailed standardized synthetic steps below outline the precise conditions for reagent charging, temperature control, and isolation procedures necessary to replicate the high yields reported in the patent data.
- Perform esterification of p-toluic acid with methanol or ethanol using sulfuric acid catalyst at 60-80°C for 8-12 hours to form the corresponding benzoate ester.
- Conduct radical bromination by reacting the ester with dibromohydantoin (0.5-0.6 molar ratio) and AIBN catalyst in dichloroethane at 60-70°C for 3-5 hours.
- Isolate the product by washing the reaction mixture with sodium thiosulfate and brine, drying the organic layer, concentrating, and recrystallizing from n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this dibromohydantoin-based methodology translates directly into tangible operational efficiencies and cost optimizations. The primary driver of value is the drastic reduction in cycle time; by compressing the reaction window from nearly a full day to merely a few hours, manufacturing facilities can significantly increase their asset utilization rates and throughput capacity without requiring additional capital investment in reactor volume. This acceleration allows for more flexible production scheduling and faster response times to market demand fluctuations, which is critical in the volatile landscape of fine chemical supply. Furthermore, the elimination of hazardous elemental bromine simplifies regulatory compliance and reduces the overhead costs associated with handling dangerous goods, storage, and specialized waste disposal protocols.
- Cost Reduction in Manufacturing: The economic argument for switching to dibromohydantoin is compelling, driven by both raw material efficiency and waste reduction. Since each molecule of dibromohydantoin contains two active bromine atoms capable of brominating two substrate molecules, the theoretical atom economy is double that of NBS, leading to substantial savings in reagent costs. Additionally, the high conversion rate eliminates the need for costly recovery processes for unreacted starting materials, which are often a hidden expense in NBS-mediated processes. The simplified workup procedure, which avoids complex chromatographic separations or extensive distillation, further lowers utility consumption and labor costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of stable, commercially available reagents that do not suffer from the supply chain volatility often seen with elemental halogens. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without compromising yield, ensures consistent batch-to-batch quality. This reliability reduces the risk of production delays caused by failed batches or off-spec material, thereby securing a steady flow of intermediates for downstream API synthesis. The ability to source dibromohydantoin from multiple global suppliers also mitigates the risk of single-source dependency, enhancing the overall resilience of the procurement strategy.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern green chemistry principles. The reduction in reaction time and the use of recyclable solvents like dichloroethane lower the overall energy footprint of the manufacturing process. The absence of heavy metal catalysts or toxic inorganic byproducts simplifies effluent treatment, making it easier to meet increasingly strict environmental discharge standards. As production scales from pilot plants to multi-ton commercial reactors, the exothermic nature of the reaction remains manageable due to the controlled release of bromine radicals, ensuring safe operation even at large volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel bromination technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and process adaptability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is dibromohydantoin preferred over N-bromosuccinimide (NBS) for this bromination?
A: Dibromohydantoin offers superior activity compared to NBS, allowing for complete conversion without residual starting material, whereas NBS often leaves 20-40% unreacted substrate. Additionally, dibromohydantoin is more cost-effective due to its dual bromine atoms per molecule and significantly reduces reaction time from 12-24 hours down to 3-5 hours.
Q: What is the typical yield and purity achievable with this novel method?
A: The process consistently achieves molar yields between 80% and 92%, with specific embodiments demonstrating yields as high as 91.7%. Gas chromatography analysis confirms product purity exceeding 99%, with no detectable raw material peaks, indicating high conversion efficiency.
Q: Can this method be scaled for industrial production of agrochemical intermediates?
A: Yes, the method is highly scalable. It utilizes common solvents like dichloroethane and avoids hazardous elemental bromine, simplifying safety protocols. The short reaction cycle and straightforward workup involving aqueous washing and recrystallization make it ideal for large-scale manufacturing of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-Bromomethylbenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team specializes in the commercialization of complex organic intermediates, leveraging cutting-edge technologies like the dibromohydantoin bromination method to deliver superior value to our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity observed in the lab are faithfully reproduced in our manufacturing facilities. Our state-of-the-art QC labs enforce stringent purity specifications, utilizing advanced analytical techniques to verify that every batch of methyl 4-bromomethylbenzoate meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this efficient synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data, discuss route feasibility assessments, or schedule a technical audit of our production capabilities. Let us help you secure a reliable, cost-effective supply of high-purity intermediates that drive your innovation forward.
