Advanced Synthesis of Organophosphine Photoinitiators for Commercial Scale-Up
Advanced Synthesis of Organophosphine Photoinitiators for Commercial Scale-Up
The chemical industry is witnessing a significant transformation in the production of high-performance organophosphine compounds, driven by the urgent need for safer and more sustainable manufacturing protocols. Patent CN110872320A introduces a groundbreaking one-pot direct condensation reaction that fundamentally alters the synthesis landscape for mesityloyl diphenylphosphine oxide, a critical photoinitiator used extensively in radiation curing systems. This innovation addresses long-standing challenges related to hazardous reagents and complex multi-step procedures that have historically plagued the production of these essential electronic chemicals. By enabling the direct condensation of mesityloyl halide with diphenylphosphine oxide, the technology offers a streamlined pathway that enhances both economic competitiveness and environmental compliance. For global procurement leaders and R&D directors, this represents a pivotal opportunity to secure a reliable organophosphine compound supplier capable of delivering high-purity materials with reduced supply chain risk. The implications for cost reduction in electronic chemical manufacturing are profound, as the elimination of dangerous intermediates simplifies waste treatment and lowers overall operational expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acylphosphine oxide photoinitiators has relied on cumbersome and hazardous methodologies that pose significant risks to both personnel and the environment. Traditional routes often necessitate the use of highly air-sensitive organometallic reagents such as n-butyllithium to generate phosphine lithium salts, which require stringent inert atmosphere conditions and specialized handling equipment. Furthermore, many existing processes involve the use of explosive organic peroxides as oxidizing agents or promoters, creating substantial safety liabilities during large-scale production. The reliance on transition metal catalysts also introduces the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet the stringent purity specifications required for electronic applications. These factors collectively contribute to elevated production costs and extended lead times, making it difficult for manufacturers to respond agilely to market demands. Additionally, the generation of hazardous byproducts like methyl chloride or chloroethane complicates regulatory compliance and increases the burden on environmental governance systems.
The Novel Approach
In stark contrast to these legacy methods, the novel one-pot condensation technology disclosed in the patent provides a robust and efficient alternative that circumvents the aforementioned bottlenecks. This approach utilizes readily available precursors such as mesityloyl halide and diphenylphosphine oxide, reacting them directly under controlled conditions to yield the target organophosphine compounds with high selectivity. The process operates within a moderate temperature range, typically between -20°C and 150°C, allowing for flexible adaptation to various reactor configurations without the need for extreme cryogenic or high-pressure infrastructure. By avoiding the use of pyrophoric reagents and explosive oxidants, the new method significantly enhances workplace safety and reduces the complexity of hazard management protocols. Moreover, the ability to selectively produce either the photoinitiator or the novel flame retardant variant by simply adjusting molar ratios offers unparalleled versatility for manufacturers serving diverse market segments. This flexibility ensures a stable supply of high-purity organophosphine compounds while minimizing waste generation and energy consumption.
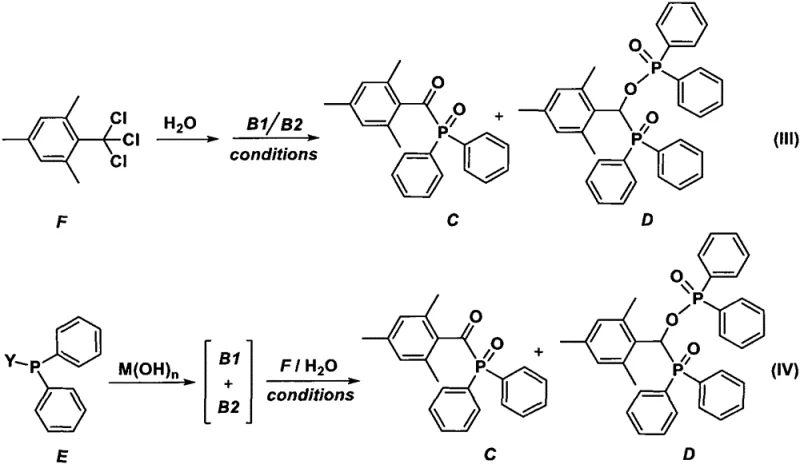
Mechanistic Insights into One-Pot Direct Condensation
The core of this technological breakthrough lies in the unique mechanistic pathway that facilitates the direct coupling of the acyl halide and the phosphine oxide without intermediate isolation. The reaction proceeds through the interaction of mesityloyl halide with diphenylphosphine oxide, which exists in equilibrium between its phosphine oxide and hydroxyphosphine tautomers. Under the influence of specific reaction conditions and additives, these tautomers engage in a nucleophilic attack on the carbonyl carbon of the acyl halide, leading to the formation of the P-C bond essential for the photoinitiator structure. The presence of activators, such as Lewis acids or even the product itself in an autocatalytic manner, further accelerates the reaction kinetics and improves overall yield. This mechanistic understanding allows for precise tuning of the reaction environment to favor the formation of the desired product while suppressing side reactions that could lead to impurities. For R&D teams, this level of control is crucial for optimizing the impurity profile and ensuring consistent batch-to-batch quality in commercial production settings.
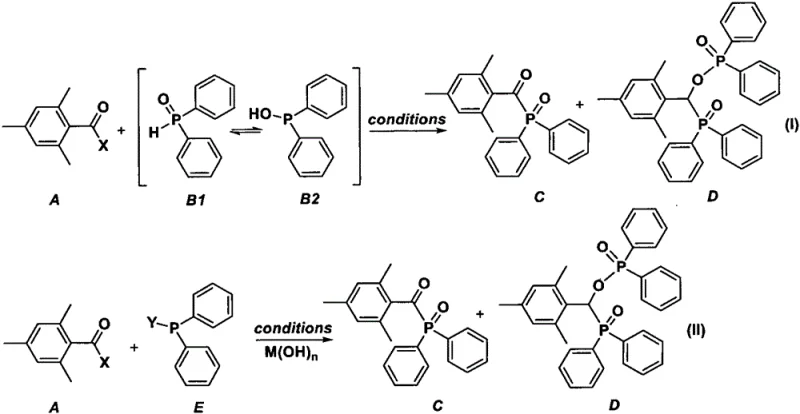
Impurity control is another critical aspect where this new mechanism offers distinct advantages over conventional oxidation-based routes. By eliminating the need for external oxidants like hydrogen peroxide, the process avoids the formation of oxidative byproducts that are often difficult to remove and can compromise the stability of the final photoinitiator. The direct condensation pathway ensures that the phosphorus atom remains in the desired oxidation state throughout the synthesis, reducing the likelihood of over-oxidation or degradation. Furthermore, the use of mild bases and solvents minimizes the risk of hydrolysis or decomposition of sensitive functional groups within the molecule. This results in a cleaner crude product that requires less intensive purification, thereby preserving yield and reducing solvent usage. For quality assurance managers, this translates to a more predictable and manageable production process that consistently meets the rigorous standards demanded by downstream applications in coatings, inks, and electronic displays.
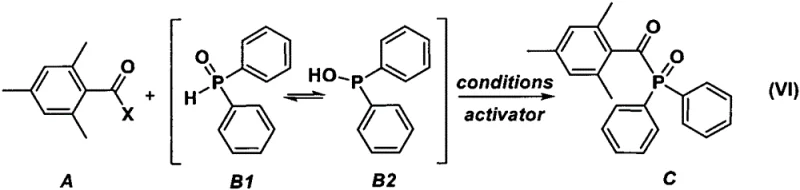
How to Synthesize Mesityloyl Diphenylphosphine Oxide Efficiently
Implementing this synthesis route in a production environment requires careful attention to reaction parameters and material handling to maximize efficiency and safety. The process begins with the preparation of a dry reaction vessel under an inert atmosphere, followed by the addition of the mesityloyl halide precursor and a suitable solvent system. Diphenylphosphine oxide is then introduced slowly, often in the presence of a base or activator to facilitate the condensation reaction. Temperature control is maintained throughout the addition and subsequent stirring period to ensure optimal reaction kinetics and prevent thermal runaway. Once the reaction is complete, the mixture is quenched with acid, and the product is isolated through standard workup procedures such as extraction and crystallization. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Prepare the reaction vessel with dry solvent and add mesityloyl halide precursor under inert gas protection.
- Introduce diphenylphosphine oxide and a suitable base or activator slowly while maintaining low temperature control.
- Quench the reaction with acid, separate the organic phase, and purify via crystallization or chromatography to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis technology offers substantial benefits that extend beyond mere technical feasibility to impact the bottom line directly. The elimination of expensive and hazardous reagents such as organolithiums and peroxides leads to a significant reduction in raw material costs and associated safety infrastructure investments. Simplified workup procedures mean less solvent consumption and lower waste disposal fees, contributing to a more sustainable and cost-effective operation. For procurement managers, this translates into a more competitive pricing structure for high-purity organophosphine compounds without compromising on quality or performance. The robustness of the process also ensures greater supply chain reliability, as it is less susceptible to disruptions caused by the scarcity of specialized reagents or stringent regulatory restrictions on hazardous chemicals. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global customers.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and explosive oxidants drastically simplifies the purification process, eliminating the need for expensive heavy metal scavengers and reducing energy consumption during distillation. This streamlining of the production workflow results in substantial cost savings that can be passed down to customers or reinvested into further R&D initiatives. Additionally, the use of common solvents and readily available precursors minimizes procurement complexity and reduces the risk of price volatility associated with niche chemicals. By optimizing the molar ratios and reaction conditions, manufacturers can achieve high yields with minimal waste, further enhancing the economic viability of the process. These factors collectively contribute to a leaner manufacturing model that is better equipped to withstand market fluctuations and competitive pressures.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available starting materials ensures a consistent supply of inputs, reducing the risk of production delays due to raw material shortages. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, which are widely available and easy to maintain, further bolstering supply chain resilience. This accessibility means that production can be scaled up or shifted between facilities with minimal requalification effort, providing flexibility in response to changing demand patterns. For supply chain heads, this reliability is paramount in ensuring that customer orders are fulfilled on time and that inventory levels are optimized to prevent stockouts or overstock situations. The ability to produce multiple products from the same platform also adds a layer of security against market shifts in specific end-use sectors.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates easy scale-up from laboratory to commercial production without the need for complex process redesign or additional unit operations. This scalability is complemented by the process inherent environmental benefits, such as the absence of ozone-depleting substances and the reduction of hazardous waste streams. Compliance with international environmental regulations is simplified, reducing the administrative burden and potential liability associated with non-compliance. The lower energy footprint of the process aligns with corporate sustainability goals, making it an attractive option for companies seeking to reduce their carbon footprint. These attributes make the technology not only commercially viable but also socially responsible, appealing to stakeholders who prioritize environmental stewardship alongside financial performance.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis technology, providing clarity for stakeholders evaluating its potential integration into their operations. These answers are derived directly from the patent data and reflect the practical realities of implementing this process in an industrial setting. Understanding these details is essential for making informed decisions about procurement strategies and production planning. The information provided here serves as a foundational resource for discussions between technical teams and management regarding the adoption of this innovative methodology.
Q: What are the safety advantages of this new synthesis route?
A: This process eliminates the need for highly air-sensitive organometallic reagents like n-butyllithium and avoids explosive peroxides, significantly enhancing operational safety.
Q: Can this method produce flame retardant variants?
A: Yes, by adjusting molar ratios and conditions, the process selectively yields mesitylene carbinol diphenyl phosphine oxide diphenyl phosphonate, a novel halogen-free flame retardant.
Q: Is the process scalable for industrial production?
A: The one-pot nature, mild temperature range from -20°C to 150°C, and use of common solvents make it highly suitable for commercial scale-up without complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mesityloyl Diphenylphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable source for advanced electronic chemicals that meet the highest standards of quality and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of organophosphine compound performs optimally in your applications. Our expertise in process optimization allows us to adapt quickly to specific customer requirements, providing tailored solutions that enhance your product performance and market competitiveness. Partnering with us means gaining access to a wealth of technical knowledge and a robust supply chain network designed to support your long-term growth objectives.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can benefit your specific operations. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient production method. Our experts are ready to provide specific COA data and route feasibility assessments to help you make data-driven decisions. By collaborating with us, you can leverage our capabilities to reduce lead time for high-purity organophosphine compounds and secure a competitive advantage in the global marketplace. Contact us today to initiate the conversation and explore the possibilities for your business.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
