Advanced Low-VOC Hydroxyalkyl Aryl Ketone Photoinitiators for Commercial Scale-Up
The global shift towards environmentally sustainable manufacturing has placed immense pressure on the chemical industry to eliminate volatile organic compound (VOC) emissions, particularly in the UV curing sector. Patent CN102417553B introduces a groundbreaking class of hydroxyalkyl aryl ketone photoinitiators designed specifically to address this critical challenge by modifying the molecular architecture to prevent the release of volatile fragments. Unlike conventional initiators that degrade into low-boiling point substances, this innovation ensures that photolytic by-products possess atmospheric boiling points significantly exceeding the 250°C threshold, effectively neutralizing VOC discharge at the source. This technological leap not only complies with stringent international environmental standards but also enhances the safety profile of end-products used in sensitive applications like food packaging and automotive coatings. 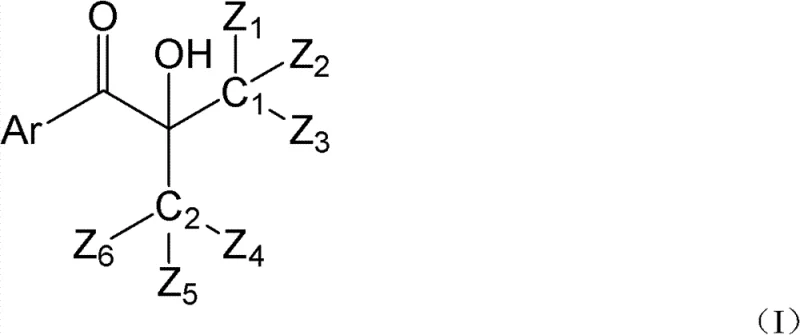 For procurement and supply chain leaders, adopting this technology represents a strategic move towards future-proofing operations against tightening regulatory frameworks while maintaining high performance. The structural versatility allows for customization of the aryl and hydroxyalkyl groups, enabling manufacturers to tailor properties such as solubility and reactivity without compromising on environmental compliance. As a reliable photoinitiator supplier, understanding the depth of this chemical innovation is essential for securing a competitive edge in the green chemistry marketplace.
For procurement and supply chain leaders, adopting this technology represents a strategic move towards future-proofing operations against tightening regulatory frameworks while maintaining high performance. The structural versatility allows for customization of the aryl and hydroxyalkyl groups, enabling manufacturers to tailor properties such as solubility and reactivity without compromising on environmental compliance. As a reliable photoinitiator supplier, understanding the depth of this chemical innovation is essential for securing a competitive edge in the green chemistry marketplace.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional hydroxyalkyl aryl ketone photoinitiators, such as the widely used Darocur 1173 and Irgacure 184, have long dominated the market due to their high initiation efficiency and rapid curing speeds. However, their fundamental chemical limitation lies in their photolysis mechanism, which inevitably generates volatile by-products like benzaldehyde and acetone upon exposure to ultraviolet radiation. These fragments have atmospheric boiling points well below the 250°C safety threshold, classifying them as VOCs that contribute to air pollution and pose potential health risks in enclosed working environments. Furthermore, the high volatility of these degradation products often leads to significant odor issues and migration problems in cured films, which is unacceptable for high-end applications such as food contact materials or medical device coatings. The inability of these legacy molecules to meet increasingly rigorous global emission standards restricts their long-term viability in modern, eco-conscious supply chains. Consequently, manufacturers relying on these conventional chemistries face growing compliance costs and the risk of product obsolescence as regulations tighten.
The Novel Approach
The novel approach detailed in the patent data fundamentally re-engineers the photoinitiator molecule to ensure that all photolytic fragments remain non-volatile under normal atmospheric conditions. By strategically selecting aryl groups and hydroxyalkyl substituents that result in by-products with boiling points greater than 179°C for aromatic aldehydes and 155°C for alkyl ketones, the technology effectively eliminates the root cause of VOC emissions. This structural modification does not merely mask the problem but solves it at the molecular level, ensuring that even after the initiator performs its function and splits into radicals, the resulting debris remains trapped within the cured matrix or possesses negligible vapor pressure. 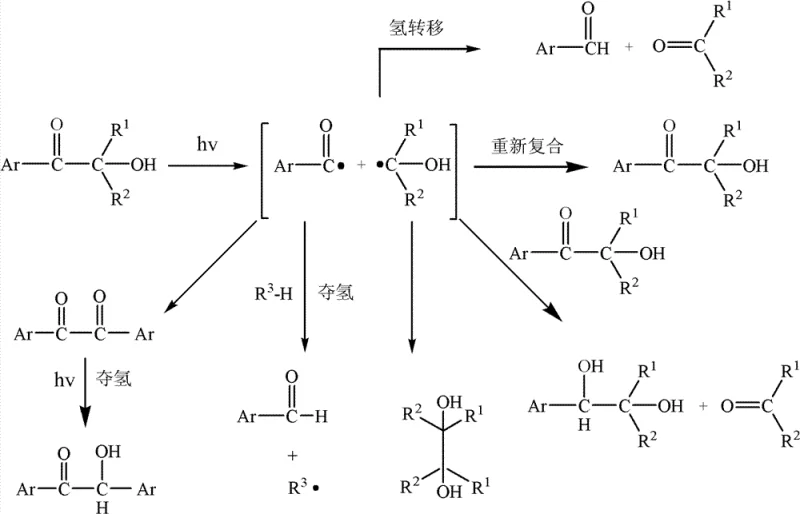 This advancement allows for cost reduction in UV curing manufacturing by removing the need for complex post-curing ventilation systems or additional scavenging agents required to capture volatile fragments. The result is a cleaner production process that aligns perfectly with the demands of a reliable agrochemical intermediate supplier or specialty chemical partner seeking to minimize environmental liability while maximizing product performance.
This advancement allows for cost reduction in UV curing manufacturing by removing the need for complex post-curing ventilation systems or additional scavenging agents required to capture volatile fragments. The result is a cleaner production process that aligns perfectly with the demands of a reliable agrochemical intermediate supplier or specialty chemical partner seeking to minimize environmental liability while maximizing product performance.
Mechanistic Insights into Low-VOC Photoinitiator Design
The core mechanism driving this innovation involves the precise control of bond dissociation energies and the thermodynamic properties of the resulting radical fragments during the Norrish Type I cleavage process. When the hydroxyalkyl aryl ketone absorbs UV energy, the bond between the carbonyl carbon and the alpha-carbon breaks, generating an aroyl radical and a hydroxyalkyl radical. In conventional systems, these radicals abstract hydrogen to form small, volatile molecules; however, in this new design, the steric bulk and molecular weight of the substituents ensure that the final stable by-products are heavy, non-volatile compounds. 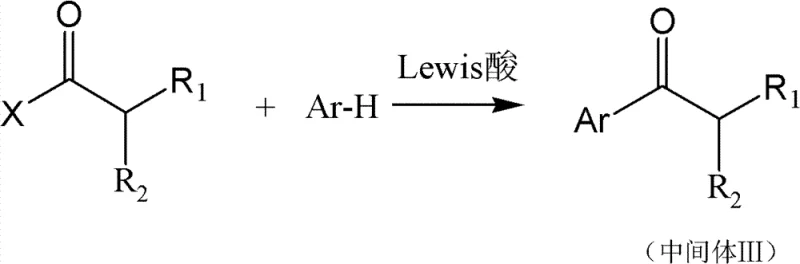 The patent specifies that the sum of carbon and heteroatoms in the substituent groups must be equal to or greater than five, a critical constraint that guarantees the resulting alkyl ketone fragment exceeds the volatility limits of cyclohexane or acetone. This mechanistic precision ensures that the initiation efficiency remains high while the environmental footprint is drastically reduced, offering a dual benefit of performance and sustainability. For R&D directors, this implies a robust impurity profile where the 'impurities' are essentially non-migratory and non-volatile, simplifying the purification and quality control processes significantly.
The patent specifies that the sum of carbon and heteroatoms in the substituent groups must be equal to or greater than five, a critical constraint that guarantees the resulting alkyl ketone fragment exceeds the volatility limits of cyclohexane or acetone. This mechanistic precision ensures that the initiation efficiency remains high while the environmental footprint is drastically reduced, offering a dual benefit of performance and sustainability. For R&D directors, this implies a robust impurity profile where the 'impurities' are essentially non-migratory and non-volatile, simplifying the purification and quality control processes significantly.
Furthermore, the introduction of electron-donating substituent groups on the aromatic ring enhances the light absorption efficiency of the initiator, allowing for effective curing even at lower concentrations or with less intense UV sources. The lengthening of the carbon chain in the molecular structure increases compatibility with various monomer systems, reducing phase separation issues that can plague high-molecular-weight additives. This compatibility is crucial for maintaining the optical clarity and mechanical integrity of the final cured coating, ensuring that the pursuit of low VOCs does not come at the expense of application performance. The ability to tune these electronic and steric properties provides a versatile platform for developing specialized grades tailored to specific resin systems, from rigid epoxy formulations to flexible acrylic coatings.
How to Synthesize Hydroxyalkyl Aryl Ketone Efficiently
The synthesis of these advanced photoinitiators follows a robust, multi-step organic pathway that leverages well-established industrial reactions, ensuring scalability and reproducibility. The process begins with the alkylation of diethyl malonate to create substituted acetic acid intermediates, followed by conversion to acyl halides and subsequent Friedel-Crafts acylation to build the core ketone structure. Detailed standardized synthesis steps see the guide below, which outlines the critical control points for temperature, stoichiometry, and purification to achieve the high purity specifications required for commercial applications. This modular synthetic strategy allows for the easy substitution of different aryl and alkyl groups, facilitating the rapid development of a diverse portfolio of low-VOC initiators to meet varying customer requirements without necessitating entirely new process infrastructure.
- Synthesize substituted acetic acid intermediates via alkylation of diethyl malonate followed by hydrolysis and decarboxylation.
- Convert the acid to an acyl halide and perform a Friedel-Crafts acylation with specific aryl groups to form the alkyl aromatic ketone.
- Execute alpha-halogenation followed by alkaline hydrolysis to yield the final hydroxyalkyl aryl ketone structure with high boiling point by-products.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this low-VOC photoinitiator technology offers substantial strategic advantages for procurement and supply chain teams focused on risk mitigation and operational efficiency. By eliminating the generation of volatile by-products, manufacturers can significantly simplify their environmental compliance protocols, reducing the administrative and capital burden associated with VOC monitoring and abatement systems. This translates directly into cost reduction in electronic chemical manufacturing or coating production, where regulatory fines and remediation costs can erode profit margins. The qualitative improvement in product safety also opens up access to premium market segments, such as food packaging and medical devices, which were previously difficult to serve with traditional high-emission chemistries.
- Cost Reduction in Manufacturing: The elimination of volatile fragments removes the necessity for expensive post-curing thermal treatments or extensive ventilation infrastructure designed to capture solvent vapors. By engineering the molecule to be inherently non-volatile upon degradation, the process avoids the need for additional scavengers or additives that increase raw material costs and complicate formulation stability. This streamlined approach leads to substantial cost savings over the product lifecycle, as the simplified processing requirements reduce energy consumption and equipment maintenance needs associated with air handling systems.
- Enhanced Supply Chain Reliability: The synthesis route utilizes readily available starting materials such as diethyl malonate and common aryl hydrocarbons, ensuring a stable and resilient supply chain不受 geopolitical disruptions affecting exotic catalysts. The robustness of the chemical steps, including standard Friedel-Crafts and hydrolysis reactions, means that production can be scaled across multiple facilities with consistent quality, reducing the risk of supply interruptions. This reliability is critical for reducing lead time for high-purity photoinitiators, allowing downstream customers to maintain lean inventory levels without fear of stockouts.
- Scalability and Environmental Compliance: The process is designed for commercial scale-up of complex photoinitiators, utilizing reaction conditions that are easily managed in standard stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. The inherent environmental benefits of the final product facilitate easier regulatory approval in key markets like Europe and North America, where REACH and TSCA compliance are major hurdles. This proactive alignment with global sustainability goals future-proofs the supply chain against tightening legislation, ensuring long-term business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this low-VOC photoinitiator technology. These answers are derived directly from the patented data to provide accurate guidance for technical teams evaluating the material for integration into their formulations. Understanding these specifics helps stakeholders make informed decisions about transitioning from legacy systems to this advanced, environmentally responsible alternative.
Q: How does this photoinitiator reduce VOC emissions compared to traditional types?
A: Traditional photoinitiators like Darocur 1173 release volatile fragments like benzaldehyde and acetone upon UV exposure. This novel structure ensures that both aromatic aldehyde and alkyl ketone by-products have atmospheric boiling points significantly higher than 250°C, effectively eliminating volatility.
Q: What are the primary industrial applications for these low-VOC photoinitiators?
A: These compounds are specifically designed for UV-curable coatings and inks where environmental compliance is critical, such as food packaging printing and automotive finishes, ensuring minimal odor and migration.
Q: Is the synthesis process scalable for commercial production?
A: Yes, the patented method utilizes standard organic synthesis steps like Friedel-Crafts acylation and alkaline hydrolysis, which are well-established and easily adaptable for large-scale manufacturing from 100 kgs to 100 MT.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxyalkyl Aryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of transitioning to sustainable chemistries without compromising on performance or supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to low-VOC photoinitiators is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for high-performance UV curing applications, providing you with a partner who understands both the chemistry and the commerce of fine chemicals.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific formulation needs. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to our low-VOC solutions, and ask for specific COA data and route feasibility assessments to validate the fit for your production lines. Let us help you engineer a cleaner, more compliant, and cost-effective future for your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
