Advanced TADF Host Material Br-DMIC-TRZ: Technical Breakthroughs and Commercial Scalability for OLED Production
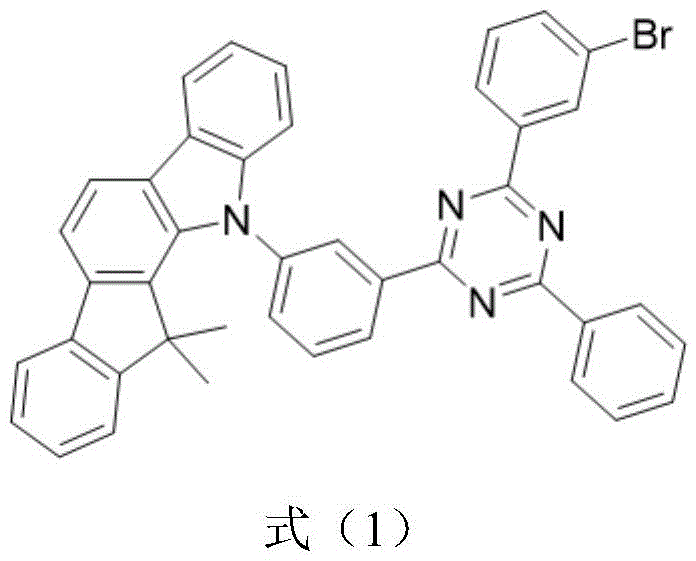
The rapid evolution of the organic light-emitting diode (OLED) industry demands continuous innovation in host materials to maximize internal quantum efficiency and device longevity. Patent CN116041329A introduces a groundbreaking high-efficiency thermally activated delayed fluorescence (TADF) host material, chemically identified as Br-DMIC-TRZ, which represents a significant leap forward in display and optoelectronic materials technology. This novel compound is synthesized by strategically combining 5,7-dihydro-7,7-dimethylindeno[2,1-b]carbazole with 2-(3-bromophenyl)-4,6-diphenyl-1,3,5-triazine, creating a molecular architecture that optimizes exciton utilization. Unlike traditional fluorescent hosts that waste triplet excitons, this TADF material leverages reverse intersystem crossing to theoretically achieve 100% internal quantum efficiency, addressing a critical bottleneck in modern flat panel display and solid-state lighting manufacturing. For R&D directors and procurement specialists seeking a reliable display & optoelectronic materials supplier, understanding the structural nuances and synthetic feasibility of this patent is essential for securing a competitive advantage in the next generation of organic electronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the OLED industry has relied heavily on traditional host materials such as 4,4'-Bis(N-carbazolyl)-1,1'-biphenyl (CBP), which, while stable, suffer from inherent limitations in exciton harvesting efficiency. Conventional hosts often fail to effectively utilize triplet excitons generated during electroluminescence, leading to significant energy loss and reduced device performance metrics such as external quantum efficiency (EQE). Furthermore, many existing host materials require complex synthetic routes involving expensive catalysts or harsh reaction conditions that complicate cost reduction in electronic chemical manufacturing. The inability to finely tune the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels in older generations of materials often results in poor charge balance within the emissive layer, causing efficiency roll-off at high brightness levels. These technical deficiencies necessitate a shift towards advanced molecular designs that can overcome spin statistics restrictions without compromising thermal stability or processability.
The Novel Approach
The innovative approach detailed in patent CN116041329A overcomes these historical constraints by introducing a bromine-substituted triazine segment that actively enhances singlet-triplet exciton conversion through the heavy atom effect. This structural modification facilitates more efficient energy transfer to guest luminescent materials, thereby drastically improving the overall efficiency of the OLED device compared to standard CBP-based systems. The synthesis method described is notably simple and utilizes raw materials that are easy to obtain, which is a critical factor for ensuring supply chain continuity and reducing lead time for high-purity OLED materials. By combining a rigid carbazole derivative with an electron-deficient triazine core, the material achieves a stable structure that is easy to store and process, making it an ideal candidate for the commercial scale-up of complex optoelectronic materials. This novel chemistry not only boosts performance metrics like EQE to over 34% in specific configurations but also lowers the turn-on voltage, contributing to lower power consumption in end-user applications.
Mechanistic Insights into Cu-Catalyzed N-Arylation and TADF Mechanism
The core of the synthetic success lies in the robust copper-catalyzed N-arylation reaction that links the triazine intermediate with the carbazole derivative, a process that requires precise control over reaction parameters to ensure high purity. The mechanism involves the activation of the aryl halide bond by the copper catalyst in the presence of 1,10-phenanthroline as a ligand, facilitating the nucleophilic attack by the nitrogen atom of the carbazole moiety. This catalytic cycle is highly efficient, as evidenced by the reported yields of approximately 85.8% in the final coupling step, indicating a well-optimized reaction pathway that minimizes side products and impurities. For technical teams, understanding this mechanism is vital because it highlights the importance of ligand selection and temperature control (130°C in DMF) in achieving the desired molecular geometry that supports TADF properties. The resulting molecular structure exhibits a small energy gap between the singlet and triplet states (ΔEst), which is the fundamental requirement for enabling thermal up-conversion of triplet excitons to the singlet state.
Impurity control is another critical aspect of this synthesis, as trace metal residues or unreacted starting materials can severely degrade OLED device performance and lifespan. The patent outlines a rigorous purification protocol involving silica gel column chromatography with specific eluent ratios, such as dichloromethane and petroleum ether, followed by recrystallization to ensure stringent purity specifications. This multi-step purification process effectively removes catalyst residues like copper iodide and unreacted intermediates, which is essential for meeting the rigorous QC labs standards required by top-tier display manufacturers. The stability of the final Br-DMIC-TRZ material is further enhanced by the steric hindrance provided by the dimethylindeno group, preventing molecular aggregation that could lead to quenching effects. Such attention to chemical detail ensures that the high-purity OLED material delivered to production lines maintains consistent batch-to-batch performance, a key requirement for maintaining yield in mass production environments.
How to Synthesize Br-DMIC-TRZ Efficiently
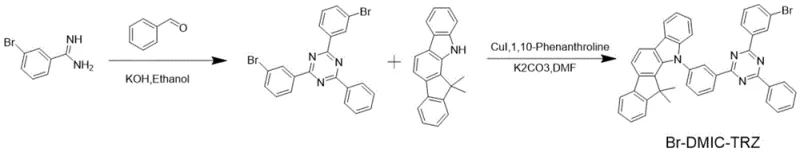
The synthesis of this high-efficiency TADF host material follows a logical two-step sequence that begins with the formation of a triazine intermediate followed by a copper-catalyzed coupling reaction. The process is designed to be operationally simple, utilizing standard laboratory equipment such as round-bottom flasks and oil baths, which translates well to larger reactor scales. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during handling of reagents like potassium hydroxide and dimethylformamide.
- Condense 3-bromobenzamidine hydrochloride with benzaldehyde in ethanol using potassium hydroxide at 70°C to form the triazine intermediate.
- Perform copper-catalyzed N-arylation between the triazine intermediate and 5,7-dihydro-7,7-dimethylindeno[2,1-b]carbazole in DMF at 130°C.
- Purify the final crude product via silica gel column chromatography and recrystallization to achieve high-purity Br-DMIC-TRZ.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of the Br-DMIC-TRZ synthesis route offers substantial cost savings by eliminating the need for precious metal catalysts often required in palladium-coupled alternatives. The use of copper iodide as a catalyst significantly reduces raw material costs, and the high yields reported in the patent minimize waste generation, contributing to a more sustainable and economically viable manufacturing process. For supply chain heads, the availability of starting materials like benzaldehyde and bromobenzamidine hydrochloride ensures that production is not bottlenecked by scarce reagents, thereby enhancing supply chain reliability and reducing the risk of delays. The simplified purification process also means shorter processing times per batch, allowing for faster turnover and more responsive fulfillment of customer orders in the fast-paced electronics market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of high-yield reaction steps directly translate to lower cost of goods sold (COGS) for the final host material. By optimizing the molar ratios of reagents and utilizing efficient purification methods, manufacturers can achieve significant cost savings without compromising on the quality or performance of the electronic chemical. This economic efficiency is crucial for maintaining competitive pricing in the global OLED material market while preserving healthy profit margins for all stakeholders involved in the value chain.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the synthesis of Br-DMIC-TRZ ensures a stable supply base that is less susceptible to geopolitical disruptions or market volatility. This stability allows procurement managers to negotiate long-term contracts with greater confidence, knowing that the raw material pipeline is secure and robust. Furthermore, the straightforward nature of the synthesis reduces the dependency on specialized contract manufacturing organizations, giving companies more flexibility in managing their production capacity and inventory levels.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory experiments to kilogram and ton-scale commercial production with minimal process re-engineering. The use of standard solvents and established purification techniques simplifies waste management and ensures compliance with increasingly strict environmental regulations regarding chemical discharge. This scalability ensures that the material can meet the growing demand for high-resolution displays and lighting solutions without encountering technical barriers during technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this TADF host material in industrial settings. These answers are derived directly from the patent data to provide accurate and actionable insights for decision-makers evaluating this technology for their product roadmaps.
Q: What is the primary advantage of Br-DMIC-TRZ over traditional CBP host materials?
A: Br-DMIC-TRZ utilizes a bromine-induced heavy atom effect to enhance reverse intersystem crossing (RISC), achieving significantly higher external quantum efficiency (EQE) compared to CBP.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the patent describes a robust two-step process using readily available raw materials and standard purification techniques like column chromatography, facilitating commercial scale-up.
Q: What are the key performance metrics for devices using this host?
A: Devices utilizing Br-DMIC-TRZ have demonstrated maximum EQE values up to 34.1% with lower turn-on voltages, indicating superior energy transfer and device stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Br-DMIC-TRZ Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is fully equipped to handle the specific synthesis requirements of complex optoelectronic materials like Br-DMIC-TRZ, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand the critical nature of supply continuity in the electronics sector and have established robust logistics networks to deliver high-purity OLED materials on time, every time. Our commitment to quality and reliability makes us the ideal partner for companies looking to integrate advanced TADF technologies into their next-generation display products.
We invite you to engage with our technical procurement team to discuss how we can support your specific material needs through a Customized Cost-Saving Analysis. By collaborating with us, you can access specific COA data and route feasibility assessments that will help you optimize your supply chain and reduce overall manufacturing costs. Let us help you bridge the gap between patent innovation and commercial reality, ensuring your products lead the market in performance and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
