Advanced Synthesis of Prasugrel Intermediates: Scalable Routes for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for antithrombotic agents continues to evolve, driven by the need for safer and more efficient synthetic routes for key active ingredients. Patent CN101402556A introduces a groundbreaking methodology for the preparation of 1-cyclopropyl-2-(2-fluorophenyl)-2-hydroxyethanone, a critical intermediate in the synthesis of Prasugrel. This novel compound addresses significant bottlenecks found in legacy manufacturing processes, offering a pathway that aligns with modern green chemistry principles while maintaining rigorous purity standards required by global regulatory bodies. For R&D Directors and Supply Chain Heads, understanding the technical nuances of this patent is essential for optimizing production pipelines and securing a competitive edge in the cardiovascular drug market. The innovation lies not just in the molecule itself, but in the strategic re-engineering of the synthetic sequence to eliminate hazardous reagents and improve overall process mass intensity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates for Prasugrel has relied heavily on halogenation strategies that pose severe environmental and operational challenges. The conventional route, as disclosed in prior art such as EP542411, necessitates the use of liquid bromine and carbon tetrachloride to generate the alpha-bromo ketone precursor. These reagents are notoriously toxic, presenting significant health risks to laboratory personnel and requiring extensive safety infrastructure to manage exposure. Furthermore, the reaction generates hydrogen bromide as a strongly acidic byproduct, which causes severe corrosion to standard stainless steel reactor equipment, leading to increased maintenance costs and potential downtime. Perhaps most critically from a commercial perspective, the condensation step in this traditional pathway suffers from a remarkably low yield of approximately 30%, rendering the overall process economically inefficient and unsuitable for large-scale industrial production where material throughput is paramount.
The Novel Approach
In stark contrast to the hazardous legacy methods, the technology described in CN101402556A pivots towards a safer and more atom-economical strategy centered on enol ester chemistry. This novel approach bypasses the need for elemental bromine entirely, instead utilizing isopropenyl acetate to form a stable enol ester intermediate through an acid-catalyzed reaction. This shift eliminates the generation of corrosive hydrogen bromide and removes the requirement for carcinogenic carbon tetrachloride solvents. The subsequent oxidation step is carefully controlled to convert the enol ester into the target hydroxy ketone with high fidelity. By replacing dangerous halogenation with a mild oxidation protocol, the new method drastically simplifies the waste treatment process and reduces the burden on environmental compliance teams. The operational simplicity, combined with significantly improved yields, makes this route a superior candidate for reliable pharmaceutical intermediates supplier networks aiming to stabilize the global supply of antithrombotic medications.
Mechanistic Insights into Acid-Catalyzed Enolization and Oxidation
The core of this technological advancement rests on a sophisticated two-step mechanistic sequence that ensures high conversion rates while minimizing side reactions. The first stage involves the reaction of cyclopropyl-2-fluorobenzyl ketone with isopropenyl acetate in the presence of a catalyst. Suitable catalysts include organic acids like methanesulfonic acid or p-toluenesulfonic acid, as well as inorganic acids, operating effectively within a temperature range of 25-120°C. This step facilitates the formation of the enol ester (Compound 3) with exceptional efficiency, often achieving yields as high as 95.0% as demonstrated in the patent examples. The mechanism relies on the acid catalyst activating the carbonyl group, promoting nucleophilic attack by the enol ether, and driving the equilibrium forward by the continuous removal of acetone byproduct. This high-yielding transformation is crucial for establishing a robust foundation for the subsequent oxidation step, ensuring that the bulk of the starting material is successfully converted into the reactive intermediate without significant loss.
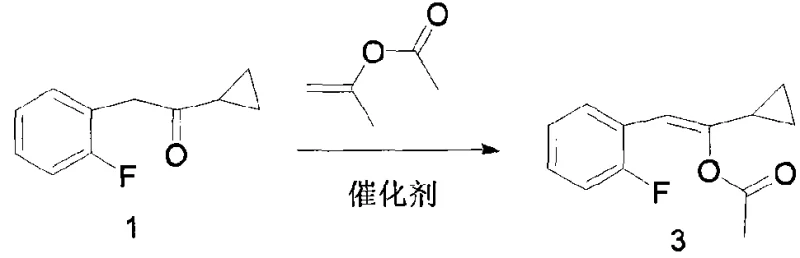
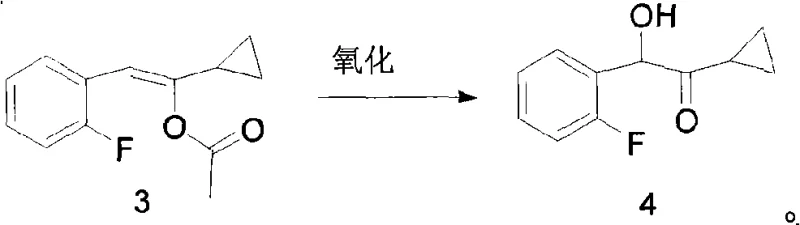
Following the formation of the enol ester, the process moves to the critical oxidation phase where the carbon-carbon double bond is cleaved to install the hydroxyl functionality. This transformation can be achieved using a variety of oxidants, including m-chloroperoxybenzoic acid, hydrogen peroxide, or peracetic acid. To further enhance reaction kinetics and selectivity, the patent describes the optional use of metal catalysts, specifically complexes formed by Mn or Fe ions with ligands such as bipyridine or phenanthroline. These metal-ligand complexes facilitate the oxygen transfer process at mild temperatures, typically between -40-60°C, with a preference for -30-0°C to control exotherms and prevent over-oxidation. The mechanistic precision here allows for the production of 1-cyclopropyl-2-(2-fluorophenyl)-2-hydroxyethanone with high purity, as the mild conditions minimize the formation of degradation byproducts that are common in harsher oxidative environments. This level of control is vital for meeting the stringent impurity profiles demanded by R&D Directors for downstream API synthesis.
How to Synthesize 1-cyclopropyl-2-(2-fluorophenyl)-2-hydroxyethanone Efficiently
Implementing this synthesis route in a commercial setting requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the precise mixing of the ketone starting material and isopropenyl acetate under reflux conditions with a catalytic amount of p-toluenesulfonic acid, ensuring continuous distillation of acetone to drive the reaction to completion. Once the enol ester is isolated, it is dissolved in a solvent such as dichloromethane or acetonitrile and cooled to sub-zero temperatures before the controlled addition of the oxidant. Whether using stoichiometric peracids or catalytic metal-peroxide systems, maintaining the specified temperature range is critical to maximizing yield and minimizing safety risks associated with exothermic oxidation. The detailed standardized synthesis steps see the guide below.
- React cyclopropyl-2-fluorobenzyl ketone with isopropenyl acetate using an acid catalyst like p-toluenesulfonic acid at reflux to form the enol ester intermediate.
- Oxidize the resulting enol ester using an oxidant such as m-chloroperoxybenzoic acid or hydrogen peroxide, optionally with a metal catalyst, at controlled low temperatures.
- Isolate the final 1-cyclopropyl-2-(2-fluorophenyl)-2-hydroxyethanone product through extraction and drying, achieving high yields suitable for industrial scaling.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the transition to this novel synthetic route offers profound strategic benefits that extend beyond simple chemical efficiency. The elimination of highly regulated and hazardous materials like liquid bromine and carbon tetrachloride simplifies the procurement process, as these substances often require special handling permits and incur high disposal costs. By switching to low-toxic, cheap, and easily obtainable reagents, manufacturers can significantly reduce the total cost of ownership associated with raw material acquisition and waste management. This shift not only lowers direct material costs but also mitigates the risk of supply chain disruptions caused by regulatory crackdowns on hazardous chemical transport. Furthermore, the simplified operational procedure reduces the need for specialized corrosion-resistant equipment, allowing for production in standard glass-lined or stainless steel reactors, which enhances asset utilization and flexibility across multi-purpose manufacturing facilities.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and hazardous halogenation reagents, which directly lowers the variable cost per kilogram of the intermediate. By avoiding the use of liquid bromine, facilities eliminate the need for costly scrubbing systems required to neutralize hydrogen bromide off-gas, resulting in substantial capital expenditure savings. Additionally, the high yield of the enolization step ensures that raw material consumption is minimized, reducing the overall material intensity of the process. The ability to use common oxidants like hydrogen peroxide further drives down reagent costs compared to specialized brominating agents. These factors combine to create a manufacturing profile that is significantly more cost-competitive, allowing for better margin protection in the face of fluctuating raw material markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the reliance on commodity chemicals that are widely available from multiple global vendors. Unlike specialized brominating agents which may have limited suppliers and long lead times, reagents like isopropenyl acetate and acetic acid are produced at massive scales for various industries, ensuring consistent availability. The robustness of the reaction conditions also means that production is less susceptible to minor variations in raw material quality, reducing the rate of batch failures and ensuring a steady flow of product. This reliability is crucial for maintaining the continuity of API production schedules, preventing costly delays in the formulation and packaging of the final antithrombotic medication. Reducing lead time for high-purity pharmaceutical intermediates becomes achievable when the synthesis is not bottlenecked by hazardous material handling protocols.
- Scalability and Environmental Compliance: From an environmental perspective, this route generates significantly less three-waste pollution, aligning with increasingly strict global environmental regulations. The absence of halogenated solvents and corrosive byproducts simplifies wastewater treatment and reduces the volume of hazardous solid waste requiring incineration. This environmental friendliness facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates, as regulatory approval processes are often faster for greener technologies. The simplicity of the operation also means that scaling from pilot plant to full commercial production involves fewer engineering challenges, allowing for rapid capacity expansion to meet market demand. This scalability ensures that the supply chain can adapt quickly to surges in demand for Prasugrel without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patent technology. These answers are derived directly from the experimental data and beneficial effects described in CN101402556A, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines. The focus is on practical implementation, safety improvements, and the tangible benefits observed in the patent examples.
Q: How does this new synthesis method improve upon conventional bromination routes?
A: The conventional method utilizes toxic liquid bromine and carbon tetrachloride, generating corrosive hydrogen bromide byproducts and suffering from low condensation yields around 30%. The new patent method replaces these hazardous reagents with low-toxic isopropenyl acetate and mild oxidants, significantly improving operational safety and overall yield.
Q: What catalysts are recommended for the oxidation step in this process?
A: The patent specifies that while oxidants like m-chloroperoxybenzoic acid can work alone, the addition of metal complexes formed by Mn or Fe ions with ligands such as bipy or phen can drastically accelerate reaction speed and improve efficiency under mild temperature conditions.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial production. It utilizes cheap and easily obtainable raw materials, generates less three-waste pollution, and involves simple operations with high yields, making it highly viable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-cyclopropyl-2-(2-fluorophenyl)-2-hydroxyethanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of life-saving cardiovascular medications. Our CDMO expertise allows us to translate complex patent methodologies like CN101402556A into reliable commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1-cyclopropyl-2-(2-fluorophenyl)-2-hydroxyethanone meets the highest quality standards required for API synthesis. We are committed to being a partner that not only supplies chemicals but also optimizes the underlying technology for maximum efficiency.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this greener, more efficient method. Contact us today to discuss how we can support your production goals with high-quality intermediates and expert technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
