Advanced Synthesis of Long-Chain Alkanoic Acid Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of Long-Chain Alkanoic Acid Derivatives for Scalable Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient and cost-effective routes to complex molecules. A recent technological breakthrough, documented in patent CN112939925A, introduces a novel preparation method for long-chain alkanoic acid derivatives that addresses critical bottlenecks in the production of high-value medical intermediates. This innovation is particularly relevant for the synthesis of glucagon-like peptide-1 (GLP-1) analogs, such as Semaglutide, where the demand for precise long-chain alkyl diacid mono-tert-butyl esters is surging. By utilizing long-chain hydroxy alkanoic acid derivatives as raw materials and employing a sodium borohydride reduction strategy, this method offers a pathway that is not only chemically robust but also economically superior to traditional approaches. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is key to securing a reliable supply chain for next-generation diabetes treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of long-chain alkyl diacid mono-tert-butyl esters has been plagued by significant economic and technical hurdles. The conventional routes often rely on expensive starting materials that drive up the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient (API). Furthermore, the synthetic difficulty associated with constructing these long-chain architectures frequently results in lower yields and complex purification requirements. In the context of manufacturing intermediates for blockbuster drugs like Semaglutide, these inefficiencies translate into supply chain vulnerabilities and inflated pricing structures. The reliance on harsh reaction conditions or scarce reagents in older methodologies further complicates the scale-up process, making it challenging for manufacturers to meet the rigorous demands of global pharmaceutical markets without compromising on quality or delivery timelines.
The Novel Approach
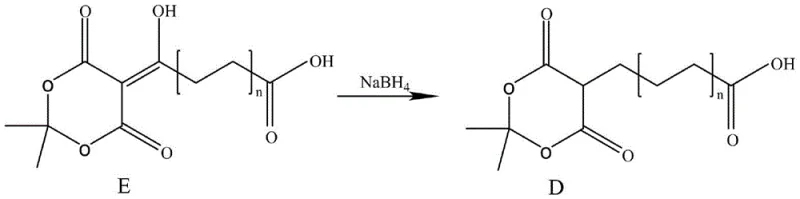
In stark contrast, the methodology outlined in patent CN112939925A presents a streamlined solution that leverages readily available long-chain hydroxy alkanoic acid derivatives. This new route bypasses the need for prohibitively expensive precursors by establishing a direct transformation via reduction. The core of this innovation lies in the conversion of a hydroxy alkanoic acid derivative (Structure E) into the target long-chain alkanoic acid derivative (Structure D) using sodium borohydride. This shift represents a fundamental optimization of the synthetic tree, reducing the number of steps and the complexity of the operation. By simplifying the molecular construction, this approach not only lowers the barrier to entry for production but also enhances the overall robustness of the manufacturing process, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Sodium Borohydride-Mediated Reduction
The chemical elegance of this process is rooted in the selective reduction capabilities of sodium borohydride when modulated by acetic acid. In the transformation of compound E to compound D, the reaction targets specific unsaturated functionalities within the dioxanone ring system while preserving the integrity of the long alkyl chain and the terminal carboxylic acid group. The mechanism likely involves the in situ generation of a more reactive borane species or a coordinated hydride transfer that facilitates the saturation of the exocyclic double bond. This chemoselectivity is paramount for R&D teams focused on impurity control, as it minimizes the formation of over-reduced byproducts or side reactions that could compromise the purity profile of the intermediate. The use of dichloromethane as a solvent at controlled low temperatures (-5°C) further ensures that the reaction kinetics are managed precisely, preventing thermal runaway and maintaining high stereochemical fidelity where applicable.
Furthermore, the preceding condensation step, which generates the precursor E from long-chain alkyl diacids and isopropylidene malonate, utilizes a classic carbodiimide-mediated coupling strategy. Employing N,N'-diisopropylcarbodiimide (DIC) alongside 4-dimethylaminopyridine (DMAP) activates the carboxylic acid for nucleophilic attack, forming the crucial carbon-carbon bond that extends the chain. This step is critical because it sets the stage for the subsequent reduction. The efficiency of this condensation directly impacts the overall yield of the final derivative. By optimizing the molar ratios of the coupling agents and the catalyst, the process ensures high conversion rates, thereby reducing the burden on downstream purification. This dual-stage mechanism—condensation followed by selective reduction—provides a comprehensive framework for synthesizing complex long-chain architectures with high precision and reliability.
How to Synthesize Long-Chain Alkanoic Acid Derivatives Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high purity. The process begins with the careful preparation of the hydroxy alkanoic acid intermediate, followed by the critical reduction step that defines the final product structure. Operators must maintain an inert atmosphere, typically using nitrogen protection, to prevent moisture interference which could deactivate the reagents. The temperature control during the addition of sodium borohydride is particularly vital; cooling the reaction mixture to approximately -5°C before reagent addition mitigates exothermic risks and controls the reaction rate. Following the reaction, a standardized workup procedure involving extraction with saturated sodium bicarbonate and washing with brine ensures the removal of acidic byproducts and inorganic salts. For a detailed breakdown of the specific operational parameters and stoichiometry required for successful execution, please refer to the standardized guide below.
- Condense long-chain alkyl diacid with isopropylidene malonate using DIC and DMAP catalysts in dichloromethane to form the hydroxy alkanoic acid derivative intermediate.
- Reduce the intermediate using sodium borohydride and acetic acid at low temperatures (-5°C) to obtain the final long-chain alkanoic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the substantial cost savings achieved through the utilization of more accessible raw materials. By shifting away from expensive, specialized precursors to common long-chain diacids and malonate derivatives, the input cost profile of the intermediate is significantly improved. This reduction in raw material expense translates directly into a more competitive pricing structure for the final API, allowing pharmaceutical companies to better manage their production budgets. Additionally, the simplified operational workflow reduces the labor and utility costs associated with complex multi-step syntheses, further enhancing the economic viability of the project.
- Cost Reduction in Manufacturing: The elimination of costly starting materials and the use of standard reagents like sodium borohydride and DIC drastically lower the bill of materials. This approach avoids the need for exotic catalysts or high-pressure equipment, which often require significant capital expenditure and maintenance. Consequently, the overall manufacturing overhead is reduced, allowing for better margin management in the production of high-volume pharmaceutical intermediates. The efficiency of the reaction also implies less waste generation, which can lower disposal costs and environmental compliance fees.
- Enhanced Supply Chain Reliability: Sourcing long-chain alkyl diacids and isopropylidene malonate is generally more straightforward than procuring specialized mono-tert-butyl esters, which may have limited suppliers. This diversification of the supply base reduces the risk of shortages and price volatility. Furthermore, the robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent output quality. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions, operating near ambient or slightly sub-zero temperatures, make this process highly scalable from pilot plant to commercial tonnage without requiring extensive re-engineering. The use of dichloromethane, while requiring proper handling, is a well-understood solvent in the industry with established recovery protocols. The simplicity of the workup, involving standard aqueous washes and crystallization or pulping steps, facilitates easy scale-up. Moreover, the high atom economy of the condensation and reduction steps aligns with green chemistry principles, minimizing the environmental footprint and simplifying regulatory approval processes for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what stakeholders can expect during technology transfer. Understanding these details is essential for risk assessment and strategic planning when integrating this new route into existing production portfolios. The answers reflect the practical realities of scaling this chemistry and the specific advantages it holds over legacy methods.
Q: Why is this new synthetic route significant for GLP-1 analog production?
A: Traditional methods for synthesizing octadecyl diacid mono-tert-butyl ester, a key intermediate for Semaglutide, suffer from high raw material costs and significant synthetic difficulty. This patent introduces a route using more accessible long-chain hydroxy alkanoic acid derivatives, drastically simplifying the process.
Q: What are the specific reaction conditions for the reduction step?
A: The reduction is performed under mild conditions, typically cooling the reaction mixture to -5°C in dichloromethane, adding acetic acid, and then introducing sodium borohydride. The reaction proceeds for 1-2 hours, ensuring high safety and operational simplicity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method creates a route suitable for industrial production. The use of common reagents like sodium borohydride and mild temperatures avoids the need for extreme pressure or specialized equipment, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Long-Chain Alkanoic Acid Derivative Supplier
As the global demand for GLP-1 analogs continues to skyrocket, the need for a dependable source of high-quality intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN112939925A to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of long-chain alkanoic acid derivative meets the exacting standards required for pharmaceutical synthesis. Our commitment to technical excellence ensures that you receive a product that facilitates smooth downstream processing and final API approval.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of modern pharmaceutical manufacturing with a partner dedicated to innovation, reliability, and mutual success.
