Optimized Synthesis of 2-Bromo-5-Fluoroaniline for High-Volume Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands high-purity intermediates to support the manufacturing of complex active pharmaceutical ingredients (APIs), particularly for breakthrough therapies like Cystic Fibrosis treatments. Patent CN112110824B introduces a robust and highly efficient method for preparing 2-bromo-5-fluoroaniline, a critical building block in the synthesis of Tezacaftor. This novel synthetic route addresses long-standing challenges in regioselectivity and yield that have plagued traditional manufacturing processes. By employing a strategic acetyl protection group, the method ensures precise positioning of substituents on the aromatic ring, thereby minimizing the formation of difficult-to-remove isomers. The technical breakthrough lies not only in the chemical elegance but also in its practical applicability for industrial scale-up, offering a reliable pathway for producing high-purity pharmaceutical intermediates. As global demand for CFTR modulators rises, securing a stable supply chain for such key precursors becomes paramount for drug developers and generic manufacturers alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated anilines has relied on direct nitration or reduction of nitro-halobenzenes, approaches that often suffer from significant drawbacks in terms of selectivity and safety. Conventional routes, such as the direct reduction of 2-bromo-5-fluoronitrobenzene using iron, frequently encounter issues with incomplete conversion and the generation of hazardous waste streams. Furthermore, direct nitration of fluoroaniline without protection often leads to a mixture of ortho and para isomers, necessitating costly and yield-reducing purification steps like column chromatography or repeated recrystallization. These inefficiencies result in elevated production costs and extended lead times, which are detrimental to the fast-paced environment of pharmaceutical development. Additionally, older methods disclosed in prior art often utilize harsh conditions that compromise the integrity of the halogen substituents, leading to debromination side reactions that further degrade the overall process efficiency and product quality.
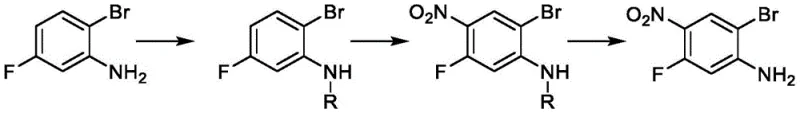
The Novel Approach
In stark contrast, the methodology disclosed in CN112110824B utilizes a sophisticated four-step sequence that prioritizes atomic economy and operational simplicity. The innovation begins with the acetylation of 4-fluoroaniline, which serves a dual purpose: protecting the sensitive amine group from oxidation and acting as a directing group for subsequent electrophilic substitution. This strategic modification allows for highly regioselective nitration, ensuring that the nitro group is installed exclusively at the desired position relative to the fluorine atom. The subsequent conversion of the acetamido group to a bromine atom via a diazonium intermediate demonstrates exceptional control over the reaction parameters, avoiding the harsh conditions typical of direct halogenation. This novel approach not only streamlines the workflow but also significantly enhances the purity profile of the final product, making it an ideal candidate for GMP-compliant manufacturing environments where consistency is key.
Mechanistic Insights into Acetyl-Directed Nitration and Sandmeyer Bromination
The core of this synthetic success lies in the precise manipulation of electronic effects on the benzene ring during the nitration phase. When 4-fluoroacetanilide is subjected to a mixture of concentrated sulfuric acid and fuming nitric acid, the acetamido group exerts a strong ortho/para directing influence. However, due to the steric hindrance and the electronic withdrawal of the fluorine atom at the para position, the electrophilic attack by the nitronium ion is funneled specifically to the ortho position relative to the acetamido group. This electronic steering is critical for avoiding the formation of the 3-nitro isomer, which would be a persistent impurity in unprotected systems. The reaction temperature is meticulously controlled between 0 to 5°C to suppress poly-nitration and hydrolysis of the acetyl group, ensuring that the intermediate 2-nitro-4-fluoroacetanilide is formed with exceptional fidelity. Such mechanistic control is the foundation of the high yields reported in the patent examples, often exceeding 95% for this specific transformation step.
Following nitration, the transformation of the acetamido group into a bromine atom represents a classic yet optimized Sandmeyer-type reaction sequence. The process involves the hydrolysis of the acetamido group to regenerate the free amine, followed immediately by diazotization using sodium nitrite under acidic conditions. The resulting diazonium salt is highly reactive and is subsequently treated with cuprous bromide and hydrobromic acid to effect the replacement of the diazo group with a bromine atom. This step is particularly sensitive to temperature and pH, with the patent specifying a range of 50 to 55°C to facilitate the radical mechanism required for bromine insertion while minimizing the risk of phenol formation via hydrolysis. The use of cuprous bromide as a catalyst ensures a smooth transition, and the subsequent reduction of the nitro group using iron powder in an ethanol-acetic acid mixture completes the synthesis. This reduction step is chosen for its cost-effectiveness and ease of workup, avoiding the need for expensive hydrogenation catalysts while maintaining high selectivity for the amino group.
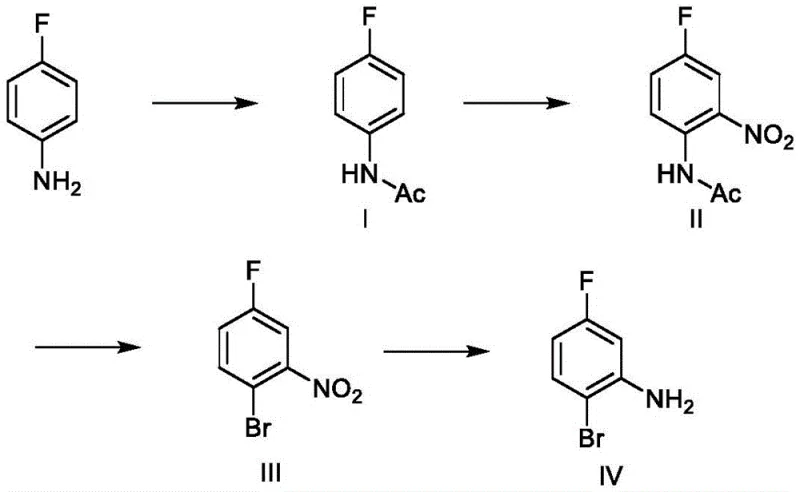
How to Synthesize 2-Bromo-5-Fluoroaniline Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to replicate the high yields observed in the patent data. The process begins with the acylation of 4-fluoroaniline in a solvent like toluene or methanol, where acetic anhydride is added dropwise to maintain a temperature below 25°C, preventing exothermic runaway. Following isolation of the acetanilide intermediate, the nitration is performed in dichloromethane with strict temperature control to ensure regioselectivity. The subsequent bromination step involves generating the diazonium salt in situ and reacting it with a copper-bromide complex, a procedure that demands precise pH adjustment to stabilize the diazonium species before substitution. Finally, the nitro group is reduced using iron powder in a mixture of ethanol and acetic acid at elevated temperatures, followed by a basic workup to liberate the free amine. For detailed standard operating procedures and specific safety guidelines regarding the handling of diazonium salts and strong acids, please refer to the technical documentation provided below.
- Acetylation of 4-fluoroaniline using acetic anhydride to form 4-fluoroacetanilide.
- Nitration of 4-fluoroacetanilide with fuming nitric acid and sulfuric acid to yield 2-nitro-4-fluoroacetanilide.
- Diazotization and bromination using sodium nitrite and cuprous bromide to generate 2-bromo-5-fluoronitrobenzene.
- Reduction of the nitro group using iron powder in acidic ethanol to obtain the final 2-bromo-5-fluoroaniline.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing commodity chemicals that are readily available in the global market, thereby reducing supply chain vulnerability. The reliance on 4-fluoroaniline as a starting material is strategic, as it is a mass-produced chemical with a stable price point, unlike specialized nitro-halobenzenes which can suffer from supply fluctuations. The elimination of expensive noble metal catalysts, such as palladium or platinum, which are often required in cross-coupling alternatives, translates directly into significant cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the high yield at each step minimizes the consumption of raw materials per kilogram of final product, enhancing the overall material efficiency of the process. This efficiency is crucial for procurement managers looking to optimize cost structures without compromising on the quality specifications required for downstream API synthesis.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the use of inexpensive reagents like iron powder for reduction and acetic anhydride for protection, avoiding the need for high-pressure hydrogenation equipment or precious metal catalysts. By maximizing the yield at every stage, the amount of waste generated per unit of product is drastically simplified, leading to lower disposal costs and reduced raw material consumption. The operational simplicity also means lower energy requirements, as most reactions proceed at moderate temperatures without the need for cryogenic cooling or extreme heating, further contributing to substantial cost savings in utility expenses.
- Enhanced Supply Chain Reliability: The use of robust, well-understood chemical transformations ensures that the manufacturing process is less prone to unexpected failures or batch rejections, guaranteeing a consistent supply of high-purity 2-bromo-5-fluoroaniline. Since the raw materials are common industrial chemicals, the risk of supply disruption due to raw material scarcity is significantly mitigated, providing a stable foundation for long-term production planning. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream manufacturers to maintain their own production schedules without interruption due to precursor shortages.
- Scalability and Environmental Compliance: The synthetic route is designed with commercial scale-up of complex pharmaceutical intermediates in mind, utilizing standard reactor types and workup procedures that can be easily transferred from pilot plant to multi-ton production. The avoidance of heavy metal catalysts simplifies the purification process and reduces the burden on wastewater treatment systems, aligning with increasingly stringent environmental regulations. The solid waste generated, primarily iron oxides from the reduction step, is easier to handle and dispose of compared to the complex organic waste streams associated with palladium-catalyzed reactions, ensuring a more sustainable and compliant manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-bromo-5-fluoroaniline based on the patented technology. These insights are derived from the specific reaction conditions and beneficial effects described in the patent documentation, aimed at clarifying the feasibility and advantages of this method for potential partners. Understanding these details is crucial for R&D and procurement teams evaluating this route for their supply chain integration.
Q: What are the key advantages of the acetyl protection strategy in this synthesis?
A: The acetyl protection strategy prevents over-oxidation of the amine group during nitration and directs the nitro group to the desired ortho position, significantly improving regioselectivity and overall yield compared to direct nitration methods.
Q: How does this method address impurity control in large-scale production?
A: By utilizing a stepwise approach with intermediate isolation and specific reagents like cuprous bromide for the Sandmeyer reaction, the process minimizes side reactions such as debromination or poly-nitration, ensuring high HPLC purity suitable for API synthesis.
Q: Is this synthetic route scalable for commercial manufacturing?
A: Yes, the route uses readily available raw materials like 4-fluoroaniline and standard reagents (acetic anhydride, iron powder), and operates at moderate temperatures, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-5-Fluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to implement the optimized synthetic route described in CN112110824B allows us to offer a competitive advantage in terms of both quality and consistency, making us a trusted partner for your complex chemical needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our advanced process technologies. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project goals. By partnering with us, you gain access to a reliable source of high-purity intermediates that can accelerate your development timelines and enhance your market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
