Advanced Rhodium-Catalyzed Synthesis of Chiral Aminocyclopropane Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks robust methodologies for constructing conformationally constrained amino acid scaffolds, which are pivotal for enhancing the metabolic stability and bioactivity of peptide-based therapeutics. Patent CN113896662A introduces a groundbreaking preparation method for aminocyclopropane carboxylic acid compounds, specifically targeting chiral aryl-beta-aminocyclopropane structures that have historically been challenging to synthesize with high stereocontrol. This innovation leverages a sophisticated chiral rhodium catalytic system to facilitate the cyclopropanation of enamine compounds with diazo precursors, delivering products with exceptional enantiomeric excess and yield. For R&D directors and procurement specialists, this technology represents a significant leap forward in accessing high-purity pharmaceutical intermediates that were previously limited by costly or inefficient synthetic routes. The ability to produce these complex scaffolds reliably opens new avenues for drug discovery programs focused on peptidomimetics and protease inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating beta-aminocyclopropane carboxylic acids often suffer from severe limitations regarding stereochemical control and operational complexity. Conventional methods frequently rely on multi-step sequences involving hazardous reagents or harsh reaction conditions that compromise the integrity of sensitive functional groups present in the molecule. Furthermore, achieving high enantioselectivity in the formation of the cyclopropane ring has historically required expensive chiral auxiliaries or resolution processes that drastically reduce overall atom economy and increase waste generation. The lack of efficient catalytic systems capable of distinguishing between enantiotopic faces of the alkene substrate has resulted in racemic mixtures that require tedious separation, thereby inflating production costs and extending lead times for critical drug development projects. These inefficiencies create substantial bottlenecks in the supply chain for advanced peptide therapeutics.
The Novel Approach
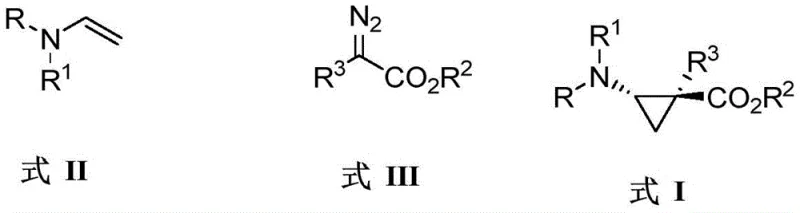
The novel approach detailed in the patent overcomes these historical barriers by employing a direct, catalytic asymmetric cyclopropanation strategy using transition metal rhodium complexes. By reacting sulfonyl enamine compounds with diazo compounds in the presence of specialized chiral rhodium catalysts, the process achieves direct construction of the cyclopropane ring with precise stereochemical definition. This method eliminates the need for stoichiometric chiral reagents and reduces the number of synthetic steps, thereby streamlining the manufacturing process significantly. The reaction conditions are remarkably mild, operating effectively at temperatures ranging from -78°C to 60°C, which allows for the preservation of sensitive ester and amine functionalities. This technological advancement provides a concise and efficient preparation method that transforms the economic and technical feasibility of producing chiral aryl-beta-aminocyclopropane carboxylic acid compounds for commercial applications.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Cyclopropanation
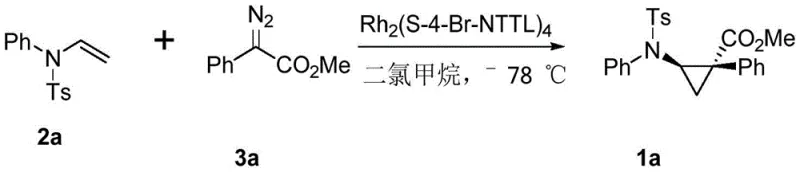
The core of this technological breakthrough lies in the unique ability of chiral dirhodium tetracarboxylate catalysts to generate metal-carbene intermediates that react with high fidelity. When the diazo compound decomposes in the presence of the rhodium catalyst, it forms a reactive rhodium-carbene species that is sterically encumbered by the chiral ligands surrounding the metal center. This chiral environment dictates the trajectory of the subsequent attack on the enamine double bond, ensuring that the cyclopropane ring forms with a specific spatial orientation. The use of catalysts such as tetrakis[N-4-bromo-naphthalic anhydride-(S)-tert-leucine]dirhodium creates a highly defined pocket that favors the formation of one enantiomer over the other, leading to the observed high enantioselectivity values up to 99%. Understanding this mechanistic nuance is crucial for R&D teams aiming to optimize reaction parameters for specific substrate classes.
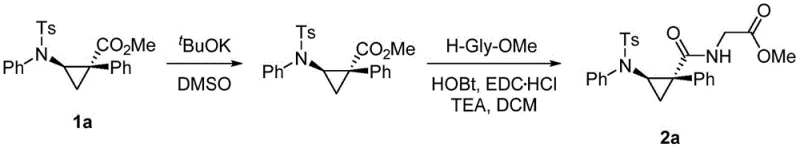
Beyond the primary cyclization event, the mechanism also inherently supports impurity control through the specificity of the catalyst-substrate interaction. The steric bulk of the catalyst ligands prevents non-productive side reactions such as dimerization of the diazo compound or insertion into non-target C-H bonds, which are common pitfalls in carbene chemistry. This selectivity ensures that the crude reaction mixture contains a high proportion of the desired product, simplifying downstream purification and reducing solvent consumption during chromatography. Furthermore, the resulting aminocyclopropane scaffolds serve as versatile building blocks for further derivatization, as demonstrated by their successful conversion into polypeptide structures. The ability to couple these rigid amino acid analogs with standard amino acids like glycine or phenylalanine highlights their utility in constructing proteolytically stable peptide therapeutics, adding significant value to the synthetic route.
How to Synthesize Chiral Aryl-Beta-Aminocyclopropane Carboxylic Acid Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the addition rate of the diazo component and the maintenance of an inert atmosphere to prevent catalyst deactivation. The process typically involves dissolving the chiral rhodium catalyst and the sulfonylenamine substrate in a dry chlorinated solvent such as dichloromethane, followed by the slow, controlled addition of the diazo compound solution via a syringe pump. This slow addition is critical to managing the concentration of the reactive carbene intermediate, minimizing side reactions and maximizing the yield of the desired cyclopropane product. Following the reaction period, which can range from 2 to 24 hours depending on the specific substrate reactivity, the mixture is worked up by solvent removal and purified using standard silica gel chromatography techniques to isolate the high-purity target compound.
- Dissolve the chiral rhodium catalyst and sulfonylenamine compound in dichloromethane under nitrogen protection to form the initial reaction mixture.
- Slowly inject the diazo compound solution into the reaction mixture using a syringe pump while maintaining strict temperature control between -78°C and room temperature.
- Stir the reaction for 2 to 24 hours, then isolate the crude product via silica gel column chromatography after solvent removal to obtain the pure target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages by addressing key pain points associated with the sourcing of complex chiral intermediates. The reliance on cheap and easily available raw materials, such as substituted enamines and diazo esters, significantly reduces the raw material cost base compared to routes requiring exotic starting materials. Additionally, the high yields reported in the patent examples, often exceeding 90%, mean that less raw material is required to produce a given amount of final product, directly contributing to substantial cost savings in manufacturing. The operational convenience of the method, which does not require extreme pressures or temperatures beyond standard cryogenic cooling, allows for implementation in existing multipurpose reactors without the need for specialized capital investment, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric chiral auxiliaries and the reduction in synthetic steps lead to a drastic simplification of the production workflow. By utilizing a catalytic amount of rhodium complex rather than equivalent amounts of chiral reagents, the process minimizes the cost of goods sold associated with chirality induction. Furthermore, the high atom economy of the cyclopropanation reaction ensures that the majority of the mass of the starting materials ends up in the final product, reducing waste disposal costs and improving overall process efficiency. This streamlined approach allows for a more competitive pricing structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The use of robust and commercially available catalysts alongside stable enamine and diazo precursors ensures a reliable supply chain that is less susceptible to disruptions caused by the scarcity of niche reagents. The versatility of the reaction conditions, which tolerate a wide range of substituents on the aromatic rings, means that a single platform technology can be used to produce a diverse library of analogues, reducing the need for multiple distinct supply chains for different drug candidates. This flexibility allows manufacturers to respond quickly to changing demand patterns in the pharmaceutical sector without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The reaction employs common organic solvents like dichloromethane and ethyl acetate, which are well-understood in terms of handling and recycling on an industrial scale, facilitating easy scale-up from gram to ton quantities. The high selectivity of the reaction minimizes the formation of by-products, thereby reducing the environmental burden associated with waste treatment and solvent recovery. This alignment with green chemistry principles not only lowers operational costs related to waste management but also ensures compliance with increasingly stringent environmental regulations, making the process sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and utility of this chiral rhodium-catalyzed synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this methodology into existing drug development pipelines or manufacturing operations.
Q: What level of enantioselectivity can be achieved with this rhodium catalytic system?
A: According to patent CN113896662A, the use of specific chiral rhodium catalysts, such as Rh2(S-4-Br-NTTL)4, can achieve enantioselectivity values as high as 99% ee, ensuring high optical purity for pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production of peptide building blocks?
A: Yes, the method utilizes cheap and easily available raw materials like sulfonyl enamines and diazo compounds, operates under mild conditions, and provides high yields, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What are the primary downstream applications for these aminocyclopropane compounds?
A: These conformationally constrained beta-aminocyclopropane carboxylic acid derivatives serve as critical building blocks for the synthesis of polypeptides, specifically designed to improve proteolytic stability in drug design.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Aryl-Beta-Aminocyclopropane Carboxylic Acid Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this innovative patent technology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and enantioselectivity demonstrated in the laboratory are maintained at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to verify the identity and quality of every batch, providing our clients with the confidence needed to advance their clinical programs. Our commitment to excellence ensures that we can deliver reliable pharmaceutical intermediates that meet the exacting standards of the global market.
We invite prospective partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this catalytic method for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your drug development efforts.
