Scalable Manufacturing of Tebipenem Intermediate V via Optimized Four-Step Synthesis
The pharmaceutical industry's relentless pursuit of effective oral antibiotics has placed significant focus on carbapenems, specifically Tebipenem Pivoxil (VI), a next-generation oral carbapenem prodrug. The commercial viability of such advanced therapeutics hinges entirely on the robustness and cost-efficiency of their key intermediates. Patent CN102250080A discloses a breakthrough preparation method for 1-(4,5-dihydro-2-thiazolinyl)-3-mercaptoazetidine hydrochloride (V), a critical building block in the Tebipenem synthesis pathway. This patent addresses long-standing manufacturing bottlenecks by replacing laborious purification techniques with streamlined crystallization processes. As illustrated in the structural relationship below, Intermediate V serves as the foundational scaffold upon which the complex beta-lactam architecture of Tebipenem Pivoxil is constructed.
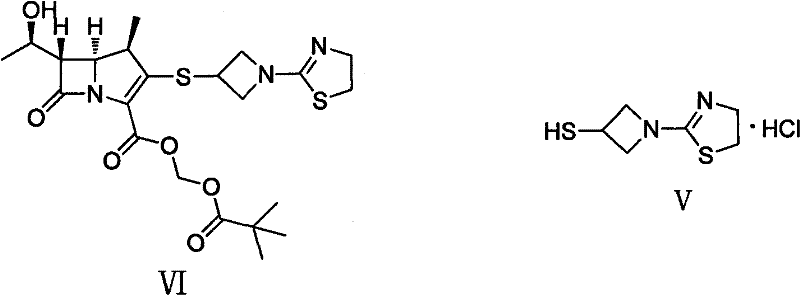
For R&D Directors and Process Chemists, the significance of this patent lies in its departure from traditional, low-yield methodologies. Historically, the synthesis of this mercaptoazetidine derivative has been plagued by excessive step counts and reliance on silica gel chromatography, a technique notoriously difficult to scale beyond the laboratory bench. The disclosed method utilizes 2-methylthio-2-thiazoline as a cheap, accessible starting material, executing a logical four-step sequence that maximizes atom economy. By rigorously optimizing reaction conditions—such as the specific use of potassium carbonate in alcoholic solvents for the initial condensation—the inventors have achieved a process that not only improves overall yield but also drastically reduces the generation of hazardous waste, aligning perfectly with modern green chemistry mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Intermediate V was characterized by prohibitive complexity and cost. For instance, the method described in Japanese Patent JP8053453 involves a three-step reaction where every single intermediate requires purification via silica gel chromatography. In an industrial context, this is a logistical nightmare; chromatography consumes vast quantities of organic solvents, requires specialized equipment with large footprints, and inevitably leads to significant product loss on the column. Similarly, routes developed by Hayashi et al. utilizing allylamine involve five steps with distillation or chromatography, while Isoda's eight-step route from benzylamine suffers from severe reaction conditions and low cumulative yields. These legacy methods create a fragile supply chain where minor deviations in purification can halt production, rendering them unsuitable for the reliable, high-volume manufacturing required for global antibiotic distribution.
The Novel Approach
The novel approach detailed in CN102250080A fundamentally re-engineers the synthetic pathway to prioritize scalability. By selecting 2-methylthio-2-thiazoline as the nucleophile source, the process bypasses the need for complex amine protections found in older routes. The innovation is most evident in the purification strategy: instead of chromatography, the intermediates are purified via recrystallization or, in some cases, telescoped directly into the next reaction vessel without isolation. This shift from batch-wise chromatographic separation to continuous or semi-continuous crystallization-based processing dramatically reduces the operational expenditure (OPEX). Furthermore, the reaction conditions are mild and utilize common industrial solvents like ethanol, dichloromethane, and DMF, ensuring that the process can be transferred to standard stainless steel reactors without requiring exotic metallurgy or extreme pressure conditions.
Mechanistic Insights into the Four-Step Substitution Cascade
The core of this synthesis is a carefully orchestrated sequence of nucleophilic substitutions and functional group transformations. The first step involves the nucleophilic attack of 3-hydroxyazetidine on the electrophilic carbon of the thiazoline ring, facilitated by a mild base like potassium carbonate. This step is critical as it establishes the azetidine-thiazoline linkage; the choice of base and temperature (80-100°C) ensures complete conversion while minimizing ring-opening side reactions. Following this, the hydroxyl group is activated via mesylation using methanesulfonyl chloride (MsCl) and triethylamine. This transformation converts a poor leaving group (-OH) into an excellent leaving group (-OMs), setting the stage for the subsequent sulfur introduction. The precision here is vital, as residual acidity from the HCl byproduct must be neutralized to prevent degradation of the acid-sensitive azetidine ring.
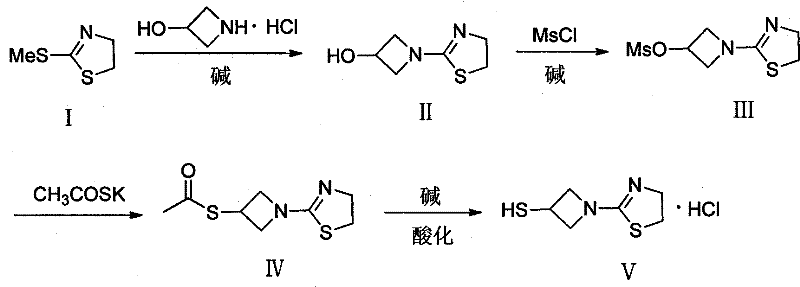
The third and fourth steps demonstrate elegant control over sulfur chemistry. The mesylate intermediate undergoes nucleophilic displacement with potassium thioacetate. Using the thioacetate anion rather than a free thiol prevents the formation of disulfide impurities, a common plague in thiol chemistry. Finally, the thioester is hydrolyzed under basic conditions (KOH/MeOH) and immediately acidified with dilute hydrochloric acid. This one-pot hydrolysis-acidification sequence ensures the thiol is generated in situ and trapped immediately as the stable hydrochloride salt (V). From an impurity control perspective, this route is superior because each step produces distinct, separable byproducts (e.g., potassium mesylate, potassium acetate) that are easily removed during the aqueous workup or crystallization phases, resulting in a final product with a clean impurity profile suitable for GMP manufacturing.
How to Synthesize 1-(4,5-dihydro-2-thiazolinyl)-3-mercaptoazetidine hydrochloride Efficiently
Executing this synthesis at a commercial scale requires strict adherence to the optimized parameters defined in the patent to ensure safety and yield. The process begins with the condensation reaction in an alcoholic solvent, followed by a controlled mesylation at low temperatures to manage exotherms. The subsequent thioacetate substitution requires polar aprotic solvents like DMF to solubilize the salts, and the final hydrolysis must be monitored to prevent over-reaction.
- Condense 2-methylthio-2-thiazoline with 3-hydroxyazetidine hydrochloride using potassium carbonate in alcohol at 80-100°C to form the hydroxy-intermediate.
- React the hydroxy-intermediate with methanesulfonyl chloride (MsCl) and triethylamine in dichloromethane at 0-10°C to generate the mesylate leaving group.
- Displace the mesylate with potassium thioacetate in DMF at 80-100°C to install the protected thiol group as a thioester.
- Hydrolyze the thioester using potassium hydroxide in methanol followed by acidification with dilute hydrochloric acid to isolate the final mercaptoazetidine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology represents a significant opportunity for cost reduction and risk mitigation. The primary driver of value is the elimination of silica gel chromatography. In traditional fine chemical manufacturing, chromatography is a major cost center due to the high price of silica, the volume of solvents required for elution, and the energy costs associated with solvent recovery. By replacing this with crystallization, the variable cost per kilogram of the intermediate drops precipitously. Additionally, the use of 2-methylthio-2-thiazoline as a starting material leverages a commodity chemical supply chain, insulating the manufacturer from the volatility often seen with specialized, custom-synthesized starting materials.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived from both material and processing efficiencies. By avoiding chromatography, the facility saves on consumables (silica) and reduces solvent purchase and disposal costs. The high atom economy of the substitution reactions means less raw material is wasted as byproducts. Furthermore, the ability to telescope intermediates (using the crude product of one step directly in the next) reduces the number of isolation cycles, saving labor hours and reducing the time the product spends in the reactor, thereby increasing asset turnover rates.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the chemistry. The reagents used—potassium carbonate, triethylamine, methanesulfonyl chloride, and potassium thioacetate—are all bulk commodities available from multiple global suppliers. This diversifies the supply base and reduces the risk of single-source bottlenecks. Moreover, the process tolerances described in the patent suggest a forgiving reaction profile, meaning that minor fluctuations in raw material quality or reaction temperature are less likely to result in batch failures, ensuring consistent on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory pressure regarding pharmaceutical waste intensifies, this process offers a clear compliance advantage. The reduction in solvent usage directly correlates to a lower carbon footprint and reduced hazardous waste generation. The "three wastes" (waste water, waste gas, waste residue) are minimized because the byproducts are largely inorganic salts that are easier to treat than complex organic mixtures from column chromatography. This environmental efficiency simplifies the permitting process for new production lines and aligns with the ESG (Environmental, Social, and Governance) goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for technology transfer teams evaluating the feasibility of adopting this method for commercial production.
Q: Why is this synthesis route superior to prior art methods like JP8053453?
A: Unlike prior art which requires silica gel chromatography purification after every step, this patented method utilizes crystallization or direct telescoping of intermediates. This eliminates the massive solvent consumption and product loss associated with column chromatography, making it viable for multi-kilogram production.
Q: What are the critical quality control points for Intermediate V?
A: Critical control points include the purity of the mesylate intermediate (III) before substitution to prevent carryover of unreacted alcohol, and strict temperature control during the final acidification step to ensure the correct hydrochloride salt formation without degrading the sensitive beta-lactam precursor structure.
Q: Is the starting material 2-methylthio-2-thiazoline commercially available?
A: Yes, 2-methylthio-2-thiazoline is an inexpensive and readily available commodity chemical. Its use as the starting point significantly lowers the raw material cost basis compared to routes starting from complex amines or requiring allylamine derivatization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(4,5-dihydro-2-thiazolinyl)-3-mercaptoazetidine hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antibiotics like Tebipenem depends on a secure and high-quality supply of critical intermediates. Our technical team has extensively analyzed the route disclosed in CN102250080A and possesses the expertise to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Intermediate V meets the exacting standards required for beta-lactam antibiotic synthesis.
We invite you to collaborate with us to optimize your supply chain for this vital component. By leveraging our process engineering capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
