Optimizing Morabemide Production: A Three-Step High-Yield Synthetic Route for Industrial Scale-Up
Optimizing Morabemide Production: A Three-Step High-Yield Synthetic Route for Industrial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for monoamine oxidase inhibitors, a critical class of therapeutic agents. Patent CN109422675B introduces a transformative methodology for synthesizing Morabemide, addressing long-standing inefficiencies in prior art. This innovation shifts the paradigm from complex, hazardous multi-step sequences to a streamlined three-step protocol that prioritizes operational simplicity and high purity. By leveraging 5-chloro-2-picolinic acid as a stable starting material, the process circumvents the need for unstable halogenated amine intermediates. For R&D directors and procurement specialists, this represents a pivotal opportunity to enhance the reliability of their pharmaceutical intermediates supply chain while simultaneously driving down manufacturing overheads through improved atom economy and reduced waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Morabemide has been plagued by operational complexity and significant safety hazards associated with halogenated intermediates. The reference method, documented in application number 201710191955.3, necessitates a cumbersome four-step sequence that relies heavily on the preparation of 2-bromoethylamine hydrobromide. This specific intermediate requires the handling of 40% hydrobromic acid, a highly corrosive reagent that poses severe risks to reactor integrity and necessitates expensive corrosion-resistant equipment. Furthermore, the subsequent salification and elution steps required to manage hydrobromic acid residues add unnecessary time and solvent consumption to the workflow. The final condensation step in this legacy route, involving a substitution reaction with morpholine, suffers from inherently low kinetic efficiency, capping the yield at a suboptimal 46%. These factors collectively create a bottleneck for commercial scale-up of complex pharmaceutical intermediates, inflating both production costs and environmental waste profiles.
The Novel Approach
In stark contrast, the methodology disclosed in CN109422675B offers a refined, three-step trajectory that elegantly bypasses the pitfalls of brominated chemistry. By utilizing ethanolamine directly in an amidation reaction with 5-chloro-2-pyridinecarbonyl chloride, the process eliminates the need for corrosive hydrobromic acid entirely. This strategic shift not only simplifies the operational workflow but also drastically improves the safety profile of the manufacturing environment. The final transformation involves a direct condensation with morpholine under dehydrating conditions, which proves kinetically superior to the substitution mechanism of the prior art, boosting the final step yield to 56%. This reduction in step count from four to three, coupled with the use of benign, commercially abundant reagents, establishes a new benchmark for cost reduction in pharmaceutical intermediates manufacturing. The streamlined nature of this route ensures faster batch turnover and reduced solvent inventory requirements.
Mechanistic Insights into Amidation and Acid-Catalyzed Condensation
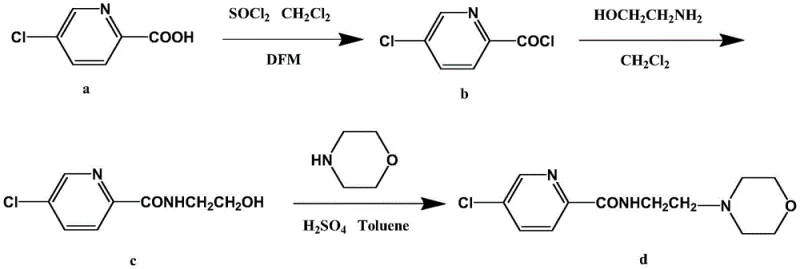
The core of this synthetic breakthrough lies in the precise control of nucleophilic acyl substitution and subsequent dehydration kinetics. The initial activation of 5-chloro-2-picolinic acid using thionyl chloride, catalyzed by trace amounts of N,N-Dimethylformamide (DMF), generates a highly reactive acid chloride species. This intermediate is then immediately subjected to nucleophilic attack by ethanolamine. The reaction is meticulously controlled at 40-50°C to balance reaction rate against potential side reactions, ensuring the formation of 5-chloro-N-(2-hydroxyethyl)-2-picolinamide with exceptional purity (99.3%). The presence of the hydroxyl group in this intermediate is crucial, as it serves as the leaving group precursor in the final cyclization step. Unlike the bromine leaving group in the old method, the hydroxyl group requires acid catalysis for effective displacement, which is achieved in the final stage using concentrated sulfuric acid in refluxing toluene.
The final condensation step is a masterpiece of thermodynamic control, utilizing toluene as both a solvent and an azeotropic agent to remove water continuously. The addition of concentrated sulfuric acid protonates the hydroxyl group of the amide intermediate, converting it into a superior leaving group (water). This facilitates the nucleophilic attack by the secondary amine nitrogen of morpholine. The reflux conditions (120-125°C) provide the necessary thermal energy to overcome the activation barrier for this dehydration, driving the equilibrium towards the product side. This mechanism avoids the formation of toxic alkyl halides and ensures that the only byproduct is water, which is easily separated. Such mechanistic elegance translates directly to high-purity pharmaceutical intermediates that require minimal downstream purification, thereby preserving yield and reducing processing time.
How to Synthesize Morabemide Efficiently
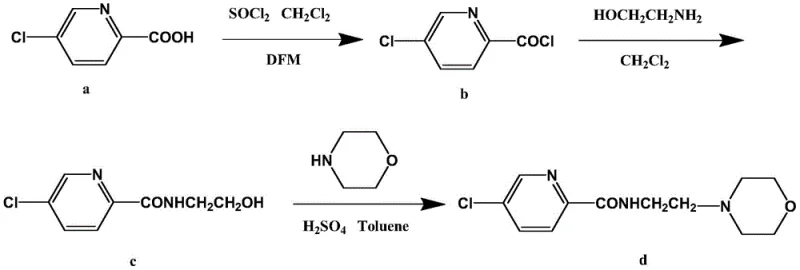
Implementing this synthesis requires strict adherence to the optimized stoichiometric ratios and temperature profiles defined in the patent to maximize the 56% yield in the final step. The process begins with the activation of the carboxylic acid, followed by a controlled amidation, and concludes with an acid-catalyzed cyclization. Each stage is designed to minimize impurity carryover, ensuring that the final crystalline product meets rigorous quality standards without extensive chromatographic purification. The detailed standardized operating procedures below outline the specific molar ratios, such as the 1:2 ratio of acid to thionyl chloride, which are critical for reproducibility. For process chemists looking to adopt this technology, understanding these nuances is key to reducing lead time for high-purity pharmaceutical intermediates.
- Convert 5-chloro-2-picolinic acid to 5-chloro-2-pyridinecarbonyl chloride using thionyl chloride and DMF catalyst in dichloromethane.
- React the acid chloride intermediate with ethanolamine at 40-50°C to form 5-chloro-N-(2-hydroxyethyl)-2-picolinamide.
- Perform acid-catalyzed condensation with morpholine in refluxing toluene to cyclize and dehydrate, yielding pure Morabemide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of corrosive hydrobromic acid from the supply chain removes a significant logistical burden, as sourcing and storing hazardous acids require specialized infrastructure and regulatory compliance measures. By switching to ethanolamine and morpholine, which are bulk commodity chemicals with stable global supply networks, manufacturers can insulate themselves from volatile raw material markets. Furthermore, the reduction in reaction steps from four to three inherently shortens the production cycle time, allowing for increased throughput within existing facility footprints. This operational efficiency translates directly into a more resilient supply chain capable of meeting fluctuating market demands for monoamine oxidase inhibitors without the risk of bottlenecks associated with complex multi-step syntheses.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the reaction sequence and the optimization of reagent usage. By reducing the molar equivalent of thionyl chloride from 4 parts in the prior art to just 2 parts, and morpholine from 3 parts to 2 parts, the direct material costs are significantly lowered. Additionally, the avoidance of expensive corrosion-resistant reactors and the elimination of neutralization steps for hydrobromic acid waste result in substantial capital expenditure savings. The higher overall yield means less starting material is wasted per kilogram of final product, further enhancing the cost-efficiency profile. These cumulative factors create a compelling business case for transitioning to this greener, leaner manufacturing protocol.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is often compromised by the dependency on niche or hazardous intermediates. This new method relies exclusively on 5-chloro-2-picolinic acid, ethanolamine, and morpholine, all of which are widely available from multiple global suppliers. This diversification of the raw material base mitigates the risk of supply disruptions caused by single-source dependencies or regulatory shutdowns of hazardous chemical plants. Moreover, the simplified workflow reduces the probability of batch failures due to operational complexity, ensuring a consistent and predictable output of Morabemide. This stability is crucial for maintaining uninterrupted production schedules for downstream API formulations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns perfectly with modern green chemistry principles. The absence of halogenated organic byproducts simplifies waste treatment protocols, reducing the load on effluent treatment plants and lowering disposal costs. The use of toluene, a common industrial solvent, allows for efficient recovery and recycling via distillation, minimizing solvent waste. The process is inherently scalable, as the exothermic nature of the acyl chlorination and amidation steps can be easily managed in large-scale reactors with standard cooling systems. This ease of scale-up ensures that production can be ramped from pilot batches to multi-ton commercial quantities without requiring fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the comparative data and experimental results presented in patent CN109422675B. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific advantages in yield, safety, and operational simplicity that distinguish this method from legacy technologies.
Q: How does the new synthesis method improve yield compared to conventional routes?
A: The novel three-step route achieves a final step yield of 56%, significantly outperforming the 46% yield of the previous four-step method which relied on corrosive hydrobromic acid intermediates.
Q: What are the primary safety advantages of this manufacturing process?
A: By eliminating the use of 40% hydrobromic acid and 2-bromoethylamine hydrobromide, the process removes highly corrosive hazards, reducing equipment maintenance costs and improving operator safety.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method utilizes readily available raw materials like ethanolamine and morpholine, operates under standard reflux conditions, and simplifies purification, making it highly scalable for industrial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Morabemide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the route disclosed in CN109422675B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific thermal and safety requirements of acyl chlorination and acid-catalyzed condensations. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of Morabemide we produce meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative synthesis route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this method can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of Morabemide for your next generation of therapeutic developments.
