Scalable Preparation of Enantiopure Chiral Diene Ligands via Custom Stationary Phase Technology
The landscape of asymmetric synthesis is fundamentally dependent on the availability of high-purity chiral ligands, which serve as the cornerstone for inducing stereochemistry in complex pharmaceutical intermediates. Patent CN110283036B introduces a transformative approach to the preparation of enantiopure chiral diene ligands, addressing a critical bottleneck in the supply chain of advanced catalytic systems. Traditional methods often struggle with the economic and technical feasibility of obtaining single enantiomers at a scale relevant for industrial catalysis. This patent discloses a sophisticated column chromatography technique that utilizes a bespoke chiral stationary phase, specifically engineered to resolve racemic diene mixtures with exceptional efficiency. By leveraging a microcrystalline cellulose derivative modified with p-methoxybenzoyl chloride and immobilized on an aminopropyl silica gel support, the technology achieves enantiomeric excess (ee) values reaching 100% for specific isomers. This breakthrough not only enhances the purity profile required for sensitive downstream reactions but also establishes a robust framework for the commercial scale-up of complex chiral ligands, ensuring that process chemists have access to reliable materials without the prohibitive costs associated with conventional resolution techniques.
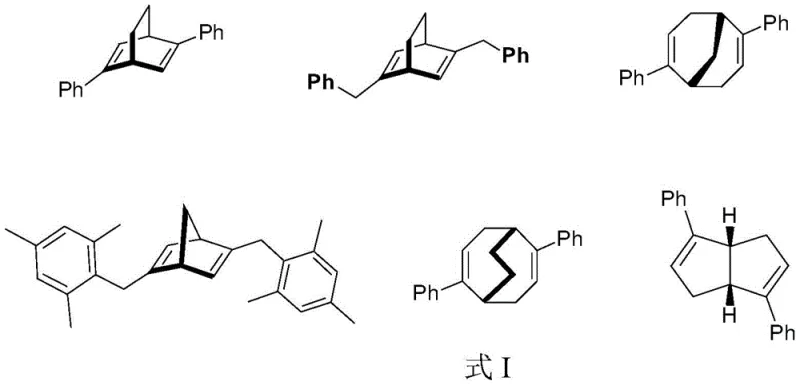
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of enantiopure chiral ligands has been plagued by significant inefficiencies inherent in classical resolution strategies. The most prevalent traditional method involves enzymatic resolution, which, while selective, suffers from a theoretical maximum yield of merely 50% for the desired enantiomer, effectively discarding half of the synthesized material and drastically inflating the cost of goods sold. Furthermore, enzymatic processes are often sensitive to reaction conditions, requiring precise control of pH, temperature, and solvent systems, which complicates process validation and scale-up. Alternative chemical resolution methods frequently rely on the formation of diastereomeric salts, a process that is labor-intensive, requires multiple recrystallization steps, and often fails to achieve the high optical purity demanded by modern regulatory standards for active pharmaceutical ingredients. Additionally, the reliance on commercial chiral HPLC columns for analytical or preparative separation presents a substantial financial barrier; these columns are exorbitantly priced, have limited loading capacities, and are prone to degradation under harsh preparative conditions, making them unsuitable for the cost reduction in pharmaceutical intermediate manufacturing that procurement teams desperately seek.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift by moving away from biological agents and towards a tailored physicochemical separation mechanism. The core innovation lies in the synthesis of a proprietary chiral filler, where microcrystalline cellulose is chemically modified to enhance its chiral recognition capabilities before being coated onto a robust silica support. This custom stationary phase creates a highly specific chiral environment within the chromatography column, allowing for the differential retention of enantiomers based on subtle stereoelectronic interactions. Unlike the rigid constraints of enzymatic pockets, this chromatographic system offers tunable selectivity through the adjustment of mobile phase composition, typically utilizing a binary mixture of n-hexane and isopropanol. The result is a process capable of resolving racemic diene ligands into distinct fractions with retention times that allow for clean collection, achieving ee values of 94.6% and 100% for the respective isomers. This approach bypasses the yield ceiling of kinetic resolutions and provides a scalable pathway for producing the high-purity chiral dienes depicted in the structural formulas, thereby securing a reliable chiral ligand supplier status for manufacturers adopting this technology.
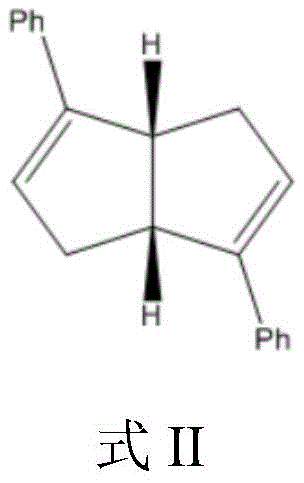
Mechanistic Insights into Cellulose-Based Chiral Recognition
The efficacy of this separation technology is rooted in the intricate molecular interactions between the analyte and the chiral stationary phase. The microcrystalline cellulose backbone provides a rigid, helical structure that acts as a chiral scaffold. When modified with aromatic groups such as p-methoxybenzoyl or p-toluoyl moieties, the polymer gains enhanced pi-pi stacking capabilities and dipole-dipole interaction sites. As the racemic chiral diene ligand passes through the column, the enantiomers transiently form diastereomeric complexes with the stationary phase. The spatial arrangement of the substituents on the cellulose derivative creates a 'chiral groove' that preferentially accommodates one enantiomer over the other through a combination of hydrogen bonding (facilitated by residual hydroxyl groups and the carbamate linkages on the silica) and steric hindrance. The aminopropyl silica gel support plays a critical role not merely as a carrier but as a functional participant; the amino groups improve the intermolecular forces between the cellulose derivative and the silica surface, ensuring a uniform and stable coating that prevents channeling and maintains column efficiency over repeated runs. This stability is crucial for maintaining consistent retention times and peak shapes, which are vital for the automated collection of high-purity fractions in a GMP environment.
Furthermore, the impurity control mechanism is inherently built into the chromatographic resolution process. In traditional synthesis, removing structurally similar impurities or regioisomers often requires extensive downstream purification. However, the high selectivity of the modified cellulose column ensures that not only are the enantiomers separated, but many achiral impurities are also eluted at different rates, effectively acting as a polishing step. The use of n-hexane and isopropanol as eluents provides a balance of polarity that optimizes the solubility of the diene ligands while maximizing the interaction with the chiral selector. The patent data indicates that the second eluting isomer can be obtained with an ee value of 100%, suggesting that the thermodynamic stability of the complex formed between this specific enantiomer and the stationary phase is sufficiently distinct to prevent co-elution. This level of precision eliminates the need for subsequent recrystallization steps, streamlining the workflow and reducing solvent consumption, which aligns with the principles of green chemistry and operational excellence.
How to Synthesize Enantiopure Chiral Diene Ligand Efficiently
The implementation of this resolution strategy requires a systematic approach to both the preparation of the stationary phase and the execution of the chromatographic run. The process begins with the rigorous modification of the cellulose polymer, followed by its immobilization on the silica support to create a durable column packing material. Once the column is packed and equilibrated, the racemic mixture is introduced, and the separation is driven by the optimized mobile phase gradient. This method transforms a challenging purification problem into a routine unit operation, enabling the production of kilogram quantities of enantiopure material. For detailed procedural specifics regarding reagent ratios, temperature controls, and packing densities, please refer to the standardized protocol below.
- Synthesize the chiral stationary phase by modifying microcrystalline cellulose with p-methoxybenzoyl chloride and coating it onto aminopropyl silica gel particles.
- Pack the modified silica gel into a medium-pressure glass column and equilibrate with n-hexane and isopropanol eluents.
- Load the racemic chiral diene ligand dissolved in n-hexane and perform gradient elution to collect enantiomers with up to 100% ee value.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented resolution method offers compelling economic and logistical benefits that directly impact the bottom line. By shifting from enzymatic or salt-based resolutions to a chromatographic process utilizing in-house or custom-prepared fillers, organizations can decouple their supply of critical chiral ligands from the volatility of the biocatalyst market. The ability to prepare the stationary phase from readily available raw materials like microcrystalline cellulose and silica gel significantly reduces the dependency on expensive commercial chiral columns, which are often subject to long lead times and price fluctuations. Moreover, the robustness of the aminopropyl silica support suggests a longer column lifetime compared to softer polymeric gels, allowing for hundreds of injection cycles without significant loss of resolution power. This durability translates directly into a lower cost per gram of purified product, facilitating cost reduction in pharmaceutical intermediate manufacturing without compromising on quality. The elimination of enzymatic steps also removes the regulatory burden associated with biological sourcing, simplifying the audit trail and accelerating the timeline for technology transfer to commercial manufacturing sites.
- Cost Reduction in Manufacturing: The economic model of this process is superior because it avoids the 50% yield loss inherent in kinetic enzymatic resolutions. By recovering both enantiomers (or at least the desired one in high yield from a racemate), the effective utilization of starting materials is doubled compared to destructive resolution methods. Additionally, the raw materials for the chiral filler—cellulose, silica, and simple acyl chlorides—are commodity chemicals with stable pricing, unlike specialized enzymes or chiral acids. The process operates at ambient temperatures and pressures, requiring no specialized high-energy equipment, which further drives down utility costs. The qualitative elimination of expensive metal catalysts or biocatalysts from the purification train means that the operational expenditure is primarily driven by solvent recovery, a well-understood and optimized cost center in any chemical plant.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and reproducibility of the column packing procedure. Since the chiral filler can be synthesized in-house or sourced from specialized material suppliers without intellectual property restrictions on the base materials, the risk of single-source supply disruption is minimized. The method uses standard normal-phase solvents (hexane/isopropanol) which are globally available and easily recycled, ensuring that production is never halted due to solvent shortages. The scalability of the medium-pressure column setup allows for a seamless transition from gram-scale R&D batches to multi-kilogram pilot runs, ensuring that the reducing lead time for high-purity chiral ligands is achievable as demand scales. This flexibility allows procurement teams to respond rapidly to fluctuating API demands without the long qualification timelines associated with new enzymatic vendors.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this chromatographic method is highly favorable. It avoids the generation of large volumes of aqueous waste streams typical of enzymatic hydrolysis or salt formation workups. The organic solvents used are amenable to efficient distillation and reuse, aligning with sustainability goals and reducing hazardous waste disposal costs. The solid waste generated (spent silica) is chemically inert and easier to handle than biological sludge. The process is inherently safer as it does not involve exothermic enzymatic reactions or the handling of sensitive biological cultures. This ease of waste management and the potential for solvent recycling make the process compliant with increasingly stringent environmental regulations, ensuring long-term operational continuity for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the experimental data and advantageous effects reported in the patent documentation, providing clarity on the practical application of the method for industrial partners seeking to optimize their chiral synthesis workflows.
Q: What is the primary advantage of this chromatographic method over enzymatic resolution?
A: Unlike enzymatic resolution which theoretically limits yield to 50% and requires expensive biocatalysts, this chromatographic method allows for the recovery of both enantiomers with high optical purity (up to 100% ee) and utilizes a reusable, chemically stable stationary phase.
Q: Can this method be scaled for industrial production of chiral ligands?
A: Yes, the patent specifically demonstrates the use of medium-pressure glass columns and custom-prepared fillers, which are significantly more cost-effective than commercial preparative HPLC columns, facilitating gram-to-kilogram scale production suitable for industrial catalysis applications.
Q: What specific chiral filler composition yields the best separation results?
A: The optimal performance is achieved using microcrystalline cellulose modified by p-methoxybenzoyl chloride (or p-toluoyl chloride derivatives) immobilized on aminopropyl-modified silica gel, which provides the necessary chiral environment for discriminating the diene enantiomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diene Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of asymmetric catalysis hinges on the consistent availability of ultra-high purity ligands. Our technical team has extensively analyzed the pathway described in CN110283036B and possesses the expertise to adapt this chromatographic resolution for large-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying chiral purity using state-of-the-art SFC and HPLC systems, guaranteeing that every batch meets stringent purity specifications required for GMP manufacturing. We understand the nuances of handling chiral dienes, which are often air-sensitive, and our infrastructure is designed to maintain product integrity throughout the packaging and shipping process.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential savings of switching to this chromatographic method versus your current resolution strategy. We encourage you to reach out for specific COA data from our pilot batches and comprehensive route feasibility assessments tailored to your target molecule. Let us help you secure a stable, cost-effective supply of enantiopure chiral dienes that will accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
