Advanced Chiral Resolution Technology for High-Purity Rociverine Intermediates and Commercial Scalability
The pharmaceutical industry continuously seeks robust pathways for the synthesis of chiral intermediates, particularly for antispasmodic agents like Rociverine. Patent CN108069846B introduces a groundbreaking resolution method for cis-1-hydroxy-[1,1'-bis(cyclohexyl)]-2-carboxylic acid, a critical precursor in the manufacturing of single-isomer Rociverine. This technology addresses the longstanding challenges associated with stereochemical control by employing S-(-)-α-phenethylamine or R-(+)-α-phenethylamine as resolving agents. By shifting away from catalytic hydrogenation towards diastereomeric salt formation, this process offers a safer, more environmentally benign route that aligns with modern green chemistry principles. The ability to access both (1S,2S) and (1R,2R) enantiomers with high fidelity provides pharmaceutical manufacturers with the flexibility needed for rigorous quality control and regulatory compliance. This technical advancement represents a significant leap forward in the production of high-purity pharmaceutical intermediates, ensuring that supply chains can meet the stringent purity specifications required for global drug registration.
![Chemical structures of cis-1-hydroxy-[1,1'-bis(cyclohexyl)]-2-carboxylic acid racemate and its enantiomers](/insights/img/rociverine-intermediate-chiral-resolution-supplier-20260315172859-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
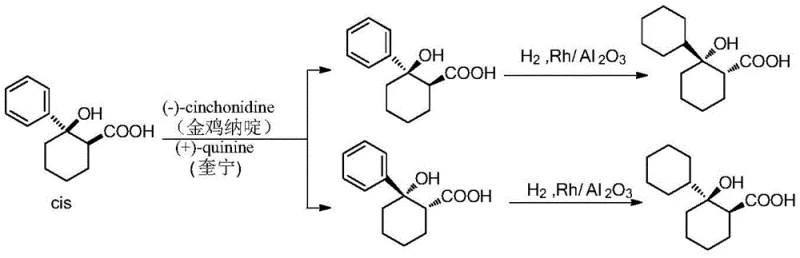
Historically, the synthesis of key Rociverine intermediates has relied on routes that are both economically and operationally burdensome. Prior art, such as the method described by Pini D et al., necessitates the high-pressure hydrogenation of benzene rings to achieve the necessary cyclohexyl structures. This step inherently requires the use of precious metal catalysts, specifically rhodium on alumina (Rh/Al2O3), which introduces significant cost volatility and supply chain risks due to the scarcity of rhodium. Furthermore, high-pressure hydrogenation operations demand specialized reactor equipment capable of withstanding extreme conditions, thereby increasing capital expenditure and operational safety hazards. The use of expensive resolving agents like cinchonidine and quinine in these traditional pathways further exacerbates the cost structure, with yields often remaining suboptimal. These factors collectively create a bottleneck for manufacturers aiming to scale production efficiently while maintaining competitive pricing structures in the global pharmaceutical market.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN108069846B circumvents these heavy industrial constraints by utilizing a direct resolution strategy on the pre-formed cyclohexyl scaffold. By employing S-(-)-α-phenethylamine or R-(+)-α-phenethylamine, the process eliminates the need for high-pressure hydrogenation steps entirely. These resolving agents are not only significantly more cost-effective, reported to be approximately one-tenth the price of traditional alkaloids like quinine, but they are also readily available in bulk quantities. The reaction proceeds under mild conditions, typically involving simple salt formation in alcohol solvents at temperatures ranging from 10°C to 60°C. This shift from catalytic reduction to crystallization-based separation drastically simplifies the process flow, reducing the number of unit operations and minimizing the generation of hazardous waste. The result is a streamlined manufacturing protocol that enhances overall process safety and economic viability without compromising the stereochemical integrity of the final product.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technological breakthrough lies in the precise formation and separation of diastereomeric salts. When the racemic cis-1-hydroxy-[1,1'-bis(cyclohexyl)]-2-carboxylic acid interacts with a chiral amine such as S-(-)-α-phenethylamine, it forms two distinct diastereomeric salts with different physical properties. The (1S,2S)-acid forms a salt with the S-amine that exhibits significantly lower solubility in specific alcoholic solvents compared to the (1R,2R)-acid salt. This differential solubility is the driving force behind the separation, allowing the desired isomer to crystallize out of the solution while the unwanted isomer remains in the mother liquor. The process is highly sensitive to solvent choice, with absolute ethanol and isopropanol proving particularly effective in modulating solubility profiles. By carefully controlling the cooling rate and stirring time, manufacturers can optimize the crystal lattice formation to exclude impurities, thereby achieving high optical purity. This mechanism ensures that the final free acid, obtained after acidification, meets the rigorous enantiomeric excess requirements necessary for downstream API synthesis.
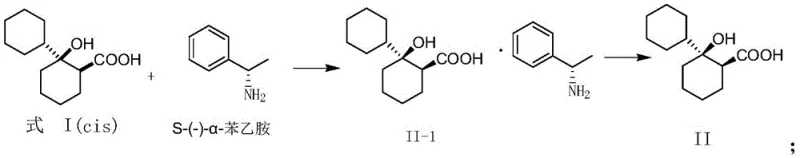
Furthermore, the purification mechanism is reinforced through recrystallization steps that further enhance optical purity. The patent details the use of ketone solvents like acetone or nitrile solvents like acetonitrile for recrystallization, which selectively dissolve minor impurities while retaining the target diastereomeric salt. The molar ratio of the resolving agent to the racemic acid is critically controlled, typically between 1:0.8 and 1:1.1, to maximize yield without sacrificing purity. This precise stoichiometric control prevents the co-crystallization of the undesired enantiomer, a common pitfall in less optimized resolution processes. The final liberation of the chiral acid is achieved through treatment with dilute hydrochloric acid, which protonates the carboxylate and releases the free acid into the organic phase. This multi-stage purification strategy ensures that the final product possesses the stringent quality attributes required for pharmaceutical applications, effectively mitigating the risk of chiral contamination in the final drug product.
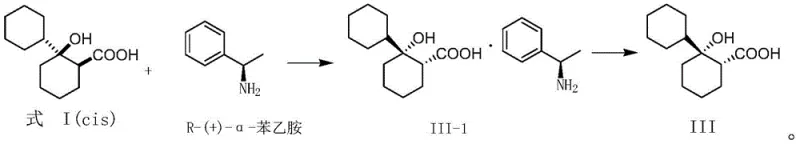
How to Synthesize cis-1-hydroxy-[1,1'-bis(cyclohexyl)]-2-carboxylic Acid Efficiently
Implementing this resolution technology requires strict adherence to the optimized parameters regarding solvent volume, temperature, and reaction time to ensure consistent batch-to-batch quality. The process begins with the dissolution of the racemic starting material in a defined volume of anhydrous alcohol, followed by the controlled addition of the chiral amine resolving agent. Maintaining the reaction temperature within the 30°C to 45°C range during the salt formation phase is crucial for initiating proper nucleation. Subsequent cooling to ice-bath temperatures facilitates the complete precipitation of the diastereomeric salt, which is then isolated via filtration. For detailed operational parameters, including specific solvent-to-mass ratios and recrystallization protocols, please refer to the standardized synthesis guide below which outlines the exact procedural steps for industrial replication.
- Dissolve the racemic cis-1-hydroxy-[1,1'-bis(cyclohexyl)]-2-carboxylic acid in a C1-C4 alkyl alcohol solvent such as absolute ethanol and heat to 45°C.
- Add the chiral resolving agent, either S-(-)-α-phenethylamine or R-(+)-α-phenethylamine, in a molar ratio of 1: 0.8 to 1:1.1 while maintaining temperature.
- Cool the reaction mixture slowly to induce crystallization of the diastereomeric salt, filter the solid, and treat with dilute hydrochloric acid to liberate the pure enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented resolution method offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of precious metal catalysts removes a significant variable cost component, shielding production budgets from the volatility of the rhodium market. Additionally, the use of commodity chemicals like alpha-phenethylamine and ethanol ensures that raw material sourcing is stable and geographically diverse, reducing the risk of supply disruptions. The simplified process flow, which avoids high-pressure equipment, lowers the barrier to entry for contract manufacturing organizations, thereby increasing the pool of potential suppliers and fostering competitive pricing. These factors combine to create a more robust and cost-efficient supply chain for this critical pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The replacement of expensive alkaloid resolving agents with alpha-phenethylamine derivatives results in a drastic reduction in raw material costs. By avoiding the need for high-pressure hydrogenation reactors and precious metal catalysts, capital expenditure and maintenance costs are significantly lowered. The simplified workup procedures also reduce labor and utility consumption, contributing to overall manufacturing efficiency and allowing for more competitive pricing strategies in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade solvents and reagents ensures that production is not bottlenecked by specialized chemical shortages. The ambient pressure conditions reduce the logistical complexities associated with transporting and storing hazardous high-pressure gases. This operational simplicity enhances the reliability of supply, ensuring that downstream API manufacturers can maintain consistent production schedules without the fear of unexpected delays caused by equipment failures or raw material scarcity.
- Scalability and Environmental Compliance: The process is inherently scalable, transitioning smoothly from laboratory benchtop to multi-ton commercial production without the need for complex engineering redesigns. The absence of heavy metal waste streams simplifies environmental compliance and waste treatment protocols, aligning with increasingly stringent global environmental regulations. This green chemistry approach not only reduces the environmental footprint but also minimizes the regulatory burden associated with hazardous waste disposal, facilitating faster approval times for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: How does this resolution method improve upon traditional hydrogenation routes?
A: Unlike prior art methods requiring high-pressure hydrogenation of benzene rings and expensive rhodium catalysts, this patent utilizes ambient pressure salt formation with low-cost phenethylamine derivatives, significantly reducing equipment hazards and raw material expenses.
Q: What is the optical purity achievable with this resolution technique?
A: Through optimized recrystallization steps using solvents like isopropanol or acetone, the process yields diastereomeric salts with high optical rotation values, such as -9.99° to -10.00° for the S-enantiomer salt, ensuring high stereochemical integrity for downstream API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids complex high-pressure reactors and uses commercially available solvents like ethanol and reagents like alpha-phenethylamine, making it highly scalable from kilogram to multi-ton production levels with simplified waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rociverine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of effective antispasmodic therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative resolution method described in patent CN108069846B can be seamlessly integrated into your supply chain. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles. Our facility is equipped to handle the specific solvent and crystallization requirements of this process, guaranteeing consistent quality and reliable delivery for your pharmaceutical projects.
We invite you to collaborate with us to optimize your sourcing strategy for this key intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this technology can reduce your overall cost of goods. Please contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and economic efficiency of your drug manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
