Industrial Scalability of 7-Methyl-3-Methylene-7-Octenal Acetal for Advanced Pheromone Manufacturing
The agricultural sector continuously demands more efficient and sustainable solutions for pest control, driving innovation in the synthesis of insect sex pheromones. Patent CN108164403B, filed in early 2022, introduces a groundbreaking methodology for the production of 7-methyl-3-methylene-7-octenal acetal compounds, which serve as pivotal intermediates for San Jose Scale (SJS) sex pheromones. This technology addresses critical bottlenecks in the existing supply chain by providing a stable, versatile precursor that can be converted into multiple active pheromone components through a unified synthetic pathway. By leveraging a robust coupling reaction between a nucleophilic 3-methyl-3-butenyl species and a functionalized acetal electrophile, the invention eliminates the reliance on hazardous organolithium reagents and complex photochemical isomerizations that have historically plagued this chemical space. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this patent represents a significant leap forward in process safety and economic viability.
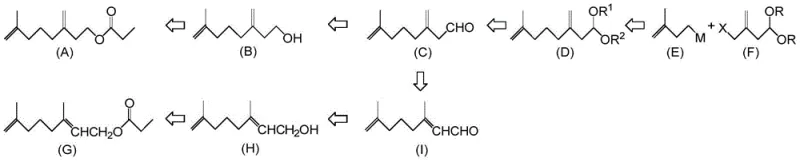
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of SJS sex pheromones such as 7-methyl-3-methylene-7-octenylpropionate has been fraught with significant technical and economic challenges that hinder large-scale adoption. Traditional routes, such as those described by Anderson et al. and Weiler et al., often depend heavily on stoichiometric amounts of expensive and pyrophoric reagents like n-butyllithium, methyllithium, and lithium aluminum hydride (LAH), which require stringent safety protocols and specialized equipment to handle safely. Furthermore, methods involving photochemical isomerization to establish the correct double bond geometry frequently result in complex mixtures of positional isomers that are exceptionally difficult to separate without resorting to preparative chromatography, a technique that is notoriously inefficient and costly at the metric-ton scale. These legacy processes also suffer from poor atom economy and generate substantial hazardous waste, creating a heavy burden on environmental compliance teams and inflating the overall cost of goods sold for the final pheromone product.
The Novel Approach
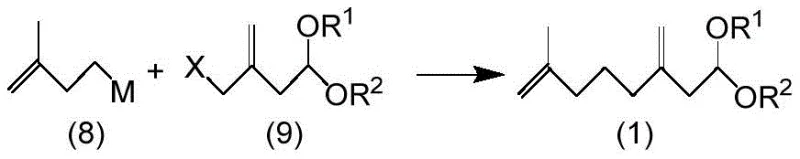
In stark contrast, the methodology disclosed in CN108164403B utilizes a convergent synthetic strategy centered around a stable 7-methyl-3-methylene-7-octenal acetal compound, designated as formula (1), which acts as a common hub for divergent synthesis. This novel approach enables the construction of the carbon skeleton via a copper-catalyzed coupling reaction between a Grignard reagent and an acetal bearing a leaving group, such as an acetoxy or halogen moiety, thereby avoiding the extreme conditions associated with organolithium chemistry. The resulting acetal intermediate possesses excellent storage stability, allowing manufacturers to produce and stockpile the precursor in large quantities without significant degradation, unlike the sensitive aldehydes or allyl chlorides used in prior art. From this single intermediate, manufacturers can selectively access different pheromone components through controlled hydrolysis and isomerization steps, offering unprecedented flexibility in production planning and inventory management for global supply chains.
Mechanistic Insights into Copper-Catalyzed Coupling and Isomerization
The core of this innovative process lies in the formation of the carbon-carbon bond through a transition metal-catalyzed coupling reaction, specifically utilizing a 3-methyl-3-butenyl nucleophile and an acetal electrophile. The reaction typically employs a Grignard reagent, such as 3-methyl-3-butenyl magnesium bromide, in the presence of a catalytic amount of a copper compound like copper(I) iodide and a phosphine ligand such as triethyl phosphite to enhance solubility and reactivity. This catalytic system facilitates an SN2 or SN2' substitution at the allylic position of the acetal substrate, effectively joining two five-carbon fragments to construct the ten-carbon backbone of the target molecule with high regioselectivity. The use of a catalytic amount of transition metal, rather than a stoichiometric quantity, drastically reduces the residual metal content in the final product, simplifying downstream purification and ensuring the high purity required for bioactive applications without the need for extensive chelating washes.
Following the coupling step, the process leverages the unique reactivity of the acetal protecting group to control the release of the reactive aldehyde functionality. Hydrolysis under mild acidic conditions converts the acetal into 7-methyl-3-methylene-7-octenal, which can then be subjected to base-catalyzed isomerization to shift the exocyclic double bond into a conjugated position, yielding 3,7-dimethyl-2,7-octadienal. This isomerization is thermodynamically driven and can be tuned by selecting specific bases, such as organic amines or alkoxides, to control the ratio of Z and E geometric isomers in the final mixture. The ability to manipulate the double bond position and geometry at this late stage allows for the precise tailoring of the pheromone blend to match the biological activity profiles required for effective pest control, while the subsequent reduction and esterification steps proceed with high chemoselectivity to preserve the sensitive olefinic bonds.
How to Synthesize 7-Methyl-3-Methylene-7-Octenal Efficiently
The synthesis of this high-value intermediate begins with the preparation of a stable acetal precursor, followed by a metal-catalyzed coupling reaction that joins two distinct carbon fragments under controlled thermal conditions. The process is designed to maximize yield while minimizing the formation of by-products through the careful selection of solvents like tetrahydrofuran and the optimization of reaction temperatures ranging from cryogenic conditions to reflux. Detailed standardized synthetic steps see the guide below, which outlines the specific molar ratios, addition rates, and workup procedures necessary to achieve pharmaceutical-grade purity suitable for agrochemical formulation.
- Couple 3-methyl-3-butenyl nucleophile with a stable acetal compound having a leaving group to form the 7-methyl-3-methylene-7-octenal acetal intermediate.
- Hydrolyze the acetal intermediate under acidic conditions to obtain 7-methyl-3-methylene-7-octenal, optionally isomerizing to 3,7-dimethyl-2,7-octadienal.
- Reduce the resulting aldehyde mixture to alcohols and subsequently esterify to yield the final propionate or other carboxylate pheromone components.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible improvements in cost structure and operational reliability, primarily driven by the elimination of hazardous reagents and the simplification of purification workflows. By replacing stoichiometric organometallic reagents with catalytic systems and utilizing distillation instead of chromatography for purification, the process significantly reduces the consumption of raw materials and solvents, leading to substantial cost savings in manufacturing operations. The enhanced stability of the acetal intermediate mitigates the risk of raw material spoilage during storage and transit, ensuring a consistent supply of quality inputs even in the face of logistical disruptions or extended lead times from upstream vendors. Furthermore, the scalability of the reaction conditions, which avoid extreme temperatures and pressures, facilitates a smoother technology transfer from pilot plant to commercial production, reducing the time and capital expenditure required to bring new pheromone products to market.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating the need for expensive and hazardous reagents such as n-butyllithium and lithium aluminum hydride, which require specialized handling and disposal protocols that drive up operational expenses. Additionally, the preference for distillation over chromatographic purification significantly lowers solvent usage and waste treatment costs, while the use of catalytic amounts of transition metals reduces the burden of heavy metal removal and recovery. These factors combine to create a leaner manufacturing process with a lower cost of goods sold, allowing for more competitive pricing in the global agrochemical market without compromising on product quality or purity specifications.
- Enhanced Supply Chain Reliability: The superior storage stability of the 3-acyloxymethyl-3-methylene-butenal acetal compound ensures that critical intermediates can be stockpiled safely for extended periods, decoupling production schedules from the immediate availability of fresh reagents and reducing the risk of production stoppages due to supply shortages. This stability also simplifies logistics and transportation requirements, as the materials do not require the same level of temperature control or hazard classification as pyrophoric organolithium reagents, thereby expanding the pool of qualified logistics providers and reducing shipping costs. Consequently, manufacturers can maintain higher service levels and shorter lead times for their customers, strengthening their position as a reliable agrochemical intermediate supplier in a volatile market.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for scale-up, utilizing common industrial solvents and reaction conditions that are easily replicated in large-scale reactors without the need for specialized photochemical equipment or cryogenic infrastructure. The reduction in hazardous waste generation, particularly the avoidance of stoichiometric metal salts and complex chromatographic fractions, aligns with increasingly stringent environmental regulations and corporate sustainability goals, minimizing the regulatory burden on manufacturing sites. This green chemistry approach not only future-proofs the production facility against evolving compliance standards but also enhances the brand reputation of the end-product among environmentally conscious consumers and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational advantages and technical feasibility for potential partners evaluating this technology for their own production lines or sourcing strategies.
Q: Why is the acetal intermediate preferred over direct aldehyde synthesis?
A: The acetal intermediate offers superior storage stability compared to the reactive aldehyde or unstable halides used in conventional methods, allowing for safer inventory management and flexible production scheduling.
Q: Can this process produce multiple pheromone components simultaneously?
A: Yes, the process allows for the controlled isomerization of the double bond, enabling the simultaneous production of 7-methyl-3-methylene-7-octenylpropionate and 3,7-dimethyl-2,7-octadienylpropionate mixtures from a common intermediate.
Q: What purification methods are suitable for industrial scale-up?
A: The patent explicitly favors distillation over chromatography for purification, which significantly reduces solvent waste and operational complexity, making the process highly viable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methyl-3-Methylene-7-Octenal Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108164403B for the efficient production of San Jose Scale sex pheromones and related agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 7-methyl-3-methylene-7-octenal derivatives meets the highest standards of quality and consistency required for effective pest control applications.
We invite you to collaborate with our technical procurement team to explore how this advanced chemistry can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements and quality needs. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, allowing us to tailor a solution that perfectly aligns with your strategic goals for cost reduction in pheromone manufacturing and supply chain resilience.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
