Advanced Nickel-Catalyzed Asymmetric Synthesis of Chiral Quaternary Carbon Alpha-Amino Acid Esters for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral quaternary carbon centers, particularly within alpha-amino acid scaffolds, due to their critical role in the rigidity and metabolic stability of non-natural polypeptides and bioactive compounds. A groundbreaking development in this domain is detailed in patent CN114516814A, which discloses a highly efficient catalytic asymmetric preparation method for chiral quaternary carbon alpha-amino acid ester compounds. This innovation leverages a chiral Nickel (Ni) catalyst to facilitate a direct asymmetric propargyl substitution reaction between propargyl alcohol ester compounds and N-acyl amino acid ester compounds. Unlike traditional approaches that often suffer from narrow substrate scope or complex catalyst preparation, this method offers a versatile platform capable of accommodating both terminal and internal alkyne substrates while utilizing commercially available chiral ligands. For R&D directors and process chemists, this represents a significant leap forward in accessing diverse libraries of chiral building blocks with exceptional stereocontrol.
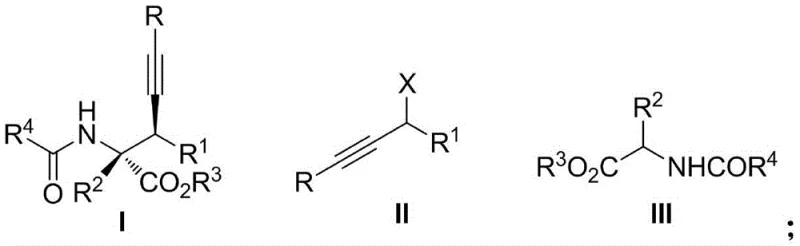
The strategic value of this technology lies in its ability to streamline the synthesis of complex amino acid derivatives, which are pivotal intermediates in the development of next-generation therapeutics. By employing a metal complex generated in situ from a Ni metal precursor and an axial chiral diphosphine ligand, the reaction proceeds under mild conditions, typically at 0 °C in solvents such as dichloromethane. This operational simplicity contrasts sharply with more demanding catalytic systems, offering a practical route for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio. The broad applicability of the reaction, covering various substituted phenyl, naphthyl, and heterocyclic groups, ensures that medicinal chemists can rapidly iterate on lead compounds without being constrained by synthetic limitations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the asymmetric synthesis of chiral quaternary carbon alpha-amino acids has often relied on copper-catalyzed decarboxylation reactions involving azlactone-based propargyl enol carbonates. While effective in specific contexts, these conventional methods present substantial drawbacks that hinder their widespread adoption in industrial settings. Firstly, the requisite propargyl alcohol enol carbonate derived from azlactone must be prepared in advance through a multi-step synthesis, which is not only complex but also results in low overall yields, thereby inflating the cost of goods. Secondly, the chiral tridentate ligands employed in these copper-catalyzed systems are typically not commercial products; they require lengthy synthetic sequences, difficult purification steps, and incur high costs, making them impractical for large-scale manufacturing. Furthermore, these legacy methods exhibit a restricted substrate scope, generally limited to terminal alkyne substrates, failing to accommodate internal alkyne variants which are often necessary for specific biological activities.
The Novel Approach
The novel approach described in the patent data overcomes these historical bottlenecks by introducing a direct asymmetric propargyl substitution catalyzed by a chiral Nickel system. This methodology eliminates the need for pre-functionalized azlactone derivatives, allowing for the direct coupling of readily available propargyl alcohol esters and N-acyl amino acid esters. A key advantage is the utilization of commercially available axial chiral diphosphine ligands, such as the L1, L2, and L3 series, which drastically simplifies the supply chain and reduces raw material costs. The reaction demonstrates remarkable versatility, successfully processing both terminal and internal alkyne substrates with high efficiency. Moreover, the operational simplicity—conducting the reaction at 0 °C with standard bases like cesium carbonate—makes this process highly attractive for cost reduction in API manufacturing, as it minimizes energy consumption and equipment requirements compared to cryogenic or high-pressure alternatives.
Mechanistic Insights into Ni-Catalyzed Asymmetric Propargyl Substitution
The core of this technological advancement is the chiral Ni catalyst, formed in situ from a Ni metal precursor and an axially chiral diphosphine ligand. The preferred Ni precursors include Ni(COD)2, Ni(acac)2, or Ni(OTf)2, with Ni(COD)2 often yielding superior catalytic activity and enantioselectivity. The ligand architecture plays a pivotal role in inducing chirality; specifically, ligands featuring a binaphthyl or similar backbone with diphenylphosphino groups (as seen in structures L1, L2, and L3) create a rigid chiral environment around the nickel center. This steric bulk effectively differentiates the enantiotopic faces of the reacting species during the formation of the pi-allyl nickel intermediate, ensuring high stereochemical fidelity. The interaction between the soft nickel center and the alkyne moiety facilitates the activation of the propargyl ester, enabling the nucleophilic attack by the amino acid ester enolate with precise spatial control.
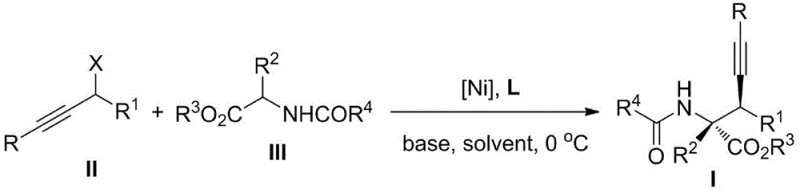
Impurity control is inherently managed through the high selectivity of the catalyst system. The use of specific ligands like L3-1, where the methylenedioxy bridges lock the conformation, has been shown to provide the best catalytic activity and stereoselectivity, often resulting in products with >99% ee. This high level of stereocontrol minimizes the formation of diastereomers and racemic byproducts, simplifying downstream purification processes. The reaction mechanism likely involves an oxidative addition of the propargyl ester to the Ni(0) species to form a cationic pi-allyl Ni(II) complex, followed by nucleophilic attack from the deprotonated N-acyl amino acid ester. The choice of base, such as Cs2CO3 or K2CO3, is critical for generating the nucleophile without promoting side reactions like elimination or hydrolysis, thereby maintaining a clean reaction profile suitable for high-purity pharmaceutical intermediate production.
How to Synthesize Chiral Quaternary Carbon Alpha-Amino Acid Esters Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the ease of handling air-sensitive catalysts under inert atmosphere. The process begins with the generation of the active catalyst species, followed by the controlled addition of substrates at low temperature to maximize selectivity.
- Prepare the chiral Ni catalyst by stirring a Ni metal precursor (e.g., Ni(COD)2) and an axial chiral diphosphine ligand (e.g., L3-1) in anhydrous dichloromethane under nitrogen protection.
- Dissolve the propargyl alcohol ester compound and N-acyl amino acid ester compound in anhydrous dichloromethane and cool the solution to 0 °C.
- Add the prepared Ni catalyst solution and a base additive (e.g., Cs2CO3) to the reaction mixture, stir at 0 °C for at least 12 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Ni-catalyzed methodology offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from custom-synthesized, expensive ligands to commercially available off-the-shelf chiral diphosphines significantly de-risks the supply chain, ensuring consistent availability of critical reagents. Additionally, the elimination of the multi-step precursor synthesis required in older copper-catalyzed methods drastically reduces the number of unit operations, leading to substantial cost savings in terms of labor, solvent usage, and waste disposal. The ability to use simple, inexpensive nickel salts further enhances the cost-effectiveness of the process compared to precious metal alternatives.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives down manufacturing costs through several mechanisms. By utilizing commercially available ligands and nickel precursors, the raw material expenditure is significantly lower than processes requiring bespoke chiral catalysts. The high yields and excellent stereoselectivity observed across a wide range of substrates mean that less starting material is wasted, and the need for costly chiral separation techniques (like preparative HPLC) is minimized or eliminated. Furthermore, the mild reaction conditions (0 °C) reduce energy consumption associated with heating or deep cooling, contributing to a leaner and more economical production process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely accessible starting materials. Propargyl alcohol esters and N-acyl amino acid esters are commodity chemicals or easily synthesized from common feedstocks, reducing dependency on single-source suppliers for exotic intermediates. The robustness of the reaction, which tolerates various functional groups and substrate types (including internal alkynes), allows for flexibility in sourcing; if one specific substrate variant is unavailable, alternative analogs can often be substituted without re-optimizing the entire catalytic system. This flexibility ensures continuous production schedules and reduces the risk of delays caused by raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by its operational simplicity and safety profile. The reaction does not require extreme pressures or temperatures, making it compatible with standard stainless steel reactors found in most multipurpose chemical plants. From an environmental perspective, the high atom economy of the direct substitution reaction generates less waste compared to stepwise approaches. The use of standard organic solvents like dichloromethane or toluene allows for established recovery and recycling protocols, aligning with modern green chemistry initiatives and regulatory requirements for waste minimization in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed asymmetric synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and practical application.
Q: What are the advantages of this Ni-catalyzed method over previous Cu-catalyzed routes?
A: This novel Ni-catalyzed method eliminates the need for complex pre-synthesis of azlactone-based propargyl enol carbonates, utilizes commercially available chiral ligands instead of difficult-to-prepare tridentate ligands, and significantly broadens the substrate scope to include both terminal and internal alkyne substrates.
Q: Which ligand provides the best stereoselectivity for this transformation?
A: Among the tested axially chiral diphosphine ligands (L1, L2, L3), the ligand L3-1 (where n=1 and Ar=Ph) demonstrates superior catalytic activity and enantioselectivity, consistently delivering products with excellent ee values.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly suitable for scale-up due to its use of readily available commercial reagents, mild reaction conditions (0 °C), simple operational procedures, and high yields, which collectively reduce production complexity and cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quaternary Carbon Alpha-Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Ni-catalyzed asymmetric preparation method for advancing drug discovery and development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm enantiomeric excess, guaranteeing that every batch of high-purity chiral amino acid derivatives meets the exacting standards of the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your supply chain and accelerate your project timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced catalytic capabilities can deliver superior value and reliability for your critical API intermediates.
