Advanced Synthesis of 3-(Halogenated Phenoxy) Benzenesulfonyl Chloride Derivatives for Pharmaceutical Applications
The pharmaceutical industry constantly seeks robust synthetic pathways for complex intermediates that balance efficiency with environmental stewardship. Patent CN113563235B introduces a significant advancement in the preparation of 3-(halogenated phenoxy) benzenesulfonyl chloride derivatives, which serve as critical building blocks for matrix metalloproteinase (MMP) inhibitors used in treating tissue ulcers and aortic aneurysms. This technology addresses long-standing challenges in traditional synthesis by replacing hazardous reagents with milder, more selective alternatives. The core innovation lies in a three-step sequence that begins with a copper-catalyzed Ullmann coupling, proceeds through a unique diazotization-sulfenylation transformation, and concludes with a controlled oxidative chlorination. For R&D teams and procurement specialists alike, this methodology represents a shift towards safer, more sustainable pharmaceutical intermediate manufacturing without compromising on yield or purity.
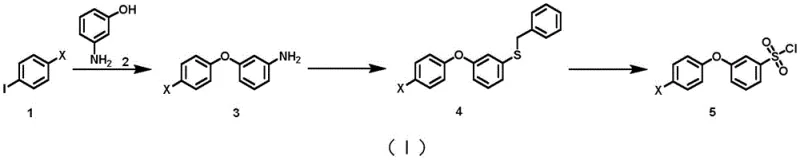
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonyl chloride derivatives has relied heavily on aggressive sulfonation and chlorination protocols involving chlorosulfonic acid and thionyl chloride. These reagents are not only highly corrosive to reactor vessels, necessitating expensive Hastelloy or glass-lined equipment, but they also generate substantial volumes of acidic wastewater that require complex neutralization and treatment processes. Furthermore, prior art strategies, such as those disclosed in European patent EP0457140A2, often utilize benzyl mercaptan for introducing the sulfur moiety. Benzyl mercaptan is notorious for its extremely unpleasant odor and high toxicity, posing severe occupational health risks and requiring specialized containment systems to prevent atmospheric contamination. The reliance on chlorine gas for oxidative chlorination in these older routes further exacerbates safety concerns due to the difficulty in storing and handling this high-toxicity gas on an industrial scale.
The Novel Approach
The methodology outlined in CN113563235B fundamentally reengineers the synthetic logic to bypass these hazards. Instead of direct sulfonation, the route constructs the carbon-sulfur bond via a diazonium intermediate reacting with sodium thiosulfate and benzyl chloride. This clever substitution eliminates the need for foul-smelling thiols entirely, replacing them with stable, odorless solids and liquids that are easier to handle and transport. The final oxidative chlorination step utilizes reagents like N-chlorosuccinimide (NCS) or dichlorohydantoin rather than elemental chlorine. This shift allows the reaction to proceed at mild temperatures, typically between 0°C and 10°C, drastically reducing energy consumption and the risk of thermal runaway. By optimizing the design of the synthetic route, the invention achieves cost reduction in pharmaceutical intermediate manufacturing through simplified post-treatment procedures and reduced waste disposal costs, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Ullmann Coupling and Diazotization
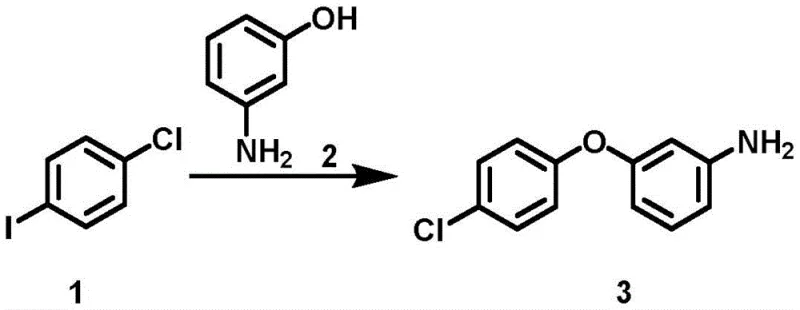
The first critical transformation in this sequence is the formation of the aryl ether bond via Ullmann coupling. In this step, a p-haloiodobenzene derivative reacts with m-hydroxyaniline in the presence of a copper catalyst and a specialized ligand, such as 2,9-diphenyl-1,10-phenanthroline. The mechanistic advantage here is twofold: the ligand stabilizes the active copper species, facilitating the oxidative addition of the aryl iodide, while simultaneously suppressing the homocoupling of the iodobenzene, a common side reaction that plagues uncatalyzed or poorly ligated systems. This high selectivity ensures that the crude product contains fewer impurities, simplifying downstream purification. The use of bases like tripotassium phosphate further drives the equilibrium forward by deprotonating the phenol, generating a more nucleophilic phenoxide species that efficiently attacks the copper-aryl complex.
Following the ether formation, the synthesis employs a sophisticated one-pot conversion of the aniline group to a benzyl sulfide. This involves the in situ generation of a diazonium salt using a nitrite ester, which then reacts with an active sulfur species derived from sodium thiosulfate and benzyl chloride. This mechanism effectively bypasses the isolation of unstable diazonium salts and avoids the use of volatile thiols. The subsequent oxidative chlorination converts the sulfide directly to the sulfonyl chloride. Unlike traditional methods that might over-oxidize or degrade sensitive functional groups, the use of N-chlorosuccinimide provides a controlled source of electrophilic chlorine. This precision is vital for maintaining the integrity of the halogenated phenoxy moiety, ensuring that the final high-purity pharmaceutical intermediate meets the stringent specifications required for API synthesis.
How to Synthesize 3-(Halogenated Phenoxy) Benzenesulfonyl Chloride Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety in a pilot or production plant setting. The process begins with the careful control of temperature and atmosphere during the coupling phase to maximize yield, followed by a sequential addition strategy in the sulfenylation step to manage exotherms. The final oxidation is conducted under ice-cooled conditions to prevent decomposition of the sulfonyl chloride product. Detailed standard operating procedures regarding solvent ratios, stirring speeds, and quenching methods are essential for consistent results. For a comprehensive breakdown of the specific molar ratios, reaction times, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Perform Ullmann coupling between p-haloiodobenzene and m-hydroxyaniline using a copper catalyst and ligand to form the ether linkage.
- Convert the resulting aniline intermediate to a benzyl sulfide via diazotization followed by reaction with sodium thiosulfate and benzyl chloride.
- Execute oxidative chlorination of the benzyl sulfide using N-chlorosuccinimide or dichlorohydantoin to yield the final sulfonyl chloride derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The substitution of hazardous raw materials with commodity chemicals significantly de-risks the supply chain. Sourcing benzyl mercaptan or chlorine gas often involves complex logistics and regulatory hurdles, whereas sodium thiosulfate, benzyl chloride, and N-chlorosuccinimide are widely available bulk chemicals with stable pricing and reliable delivery schedules. This shift enhances supply continuity and reduces the administrative burden associated with handling restricted substances.
- Cost Reduction in Manufacturing: The elimination of corrosive reagents like chlorosulfonic acid translates directly into lower capital expenditure and maintenance costs. Reactors do not require exotic alloys to withstand extreme acidity, and the lifespan of equipment is extended. Furthermore, the simplified waste stream, devoid of heavy acidic loads and toxic sulfide odors, reduces the cost of effluent treatment. By shortening the synthetic steps and avoiding expensive purification cycles needed to remove mercaptan residues, the overall cost of goods sold is substantially optimized.
- Enhanced Supply Chain Reliability: The reliance on cheap and easily obtained starting materials, such as p-haloiodobenzene and m-hydroxyaniline, ensures that production is not bottlenecked by scarce reagents. The robustness of the Ullmann coupling and the mild conditions of the final oxidation step mean that the process is less susceptible to batch failures caused by minor fluctuations in temperature or reagent quality. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify, this green chemistry approach positions the product favorably for long-term production. The absence of high-toxicity gases and the reduction in wastewater volume align with modern environmental, social, and governance (ESG) goals. The process is inherently safer to scale from kilogram to tonne quantities because the exothermic profiles are manageable, and the reagents do not pose immediate catastrophic risks in the event of a leak, facilitating smoother regulatory approvals for commercial plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield optimization and safety protocols.
Q: What are the primary safety advantages of this new synthesis route compared to conventional methods?
A: The novel route eliminates the use of highly corrosive chlorosulfonic acid and toxic chlorine gas. Furthermore, it replaces foul-smelling and hazardous benzyl mercaptan with safer sodium thiosulfate and benzyl chloride, significantly improving workplace safety and environmental compliance.
Q: How does the Ullmann coupling step ensure high selectivity?
A: By utilizing specific ligands such as 2,9-diphenyl-1,10-phenanthroline alongside a copper catalyst, the reaction promotes the cross-coupling between the aryl iodide and phenol while minimizing the self-coupling of the haloiodobenzene, leading to higher purity of the ether intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It uses cheap and readily available raw materials like p-haloiodobenzene and operates under mild conditions (0-10°C for the final step), reducing energy consumption and equipment corrosion risks associated with strong acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Halogenated Phenoxy) Benzenesulfonyl Chloride Supplier
The synthetic pathway detailed in CN113563235B exemplifies the kind of innovative process chemistry that drives modern API manufacturing. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such laboratory-scale breakthroughs into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to multi-tonne manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for downstream drug synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify how our manufacturing prowess can support your project timelines and quality objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
