Advanced Catalytic Synthesis of N-ethyl-3,4-(methylenedioxy)aniline for Commercial Scale-up
Advanced Catalytic Synthesis of N-ethyl-3,4-(methylenedioxy)aniline for Commercial Scale-up
The global demand for high-purity heterocyclic amines continues to surge, driven by their critical role as building blocks in the synthesis of complex therapeutic agents and agrochemical formulations. Among these, N-ethyl-3,4-(methylenedioxy)aniline (often abbreviated as NEBA) stands out as a pivotal intermediate for manufacturing drugs targeting gastrointestinal disorders and cardiovascular systems, as well as potent antibacterial quinazoline derivatives like oxolinic acid. A significant technological breakthrough in the production of this valuable compound is detailed in patent CN101875650A, which outlines a robust, industrially viable preparation process that fundamentally shifts away from hazardous halogenation chemistries. This report provides a deep technical analysis of this novel route, highlighting its mechanistic elegance and its profound implications for supply chain stability and cost efficiency in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-ethyl-3,4-(methylenedioxy)aniline has relied heavily on pathways originating from piperonyl cyclonene that involve electrophilic aromatic substitution with bromine followed by nucleophilic substitution. While conceptually straightforward, this traditional bromination-ammonolysis route presents severe engineering and safety challenges that hinder large-scale adoption. The ammonolysis step, in particular, necessitates extreme reaction conditions characterized by high temperatures and elevated pressures to force the displacement of the bromide leaving group with an amine source. These harsh parameters not only escalate energy consumption and equipment maintenance costs but also generate significant quantities of halogenated waste streams that require complex and expensive remediation. Furthermore, the post-reaction workup in these legacy processes is notoriously difficult, often resulting in poor product isolation and inconsistent quality profiles that fail to meet the stringent specifications required by modern pharmaceutical clients.
The Novel Approach
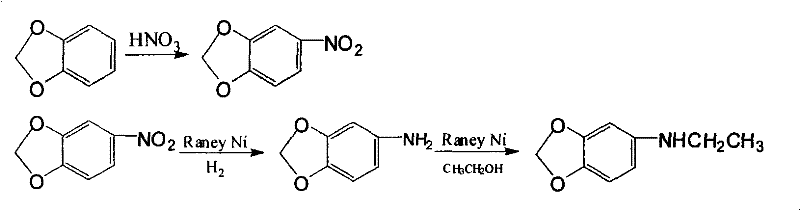
In stark contrast to the cumbersome halogenation strategies, the methodology disclosed in CN101875650A introduces a streamlined, two-stage sequence that leverages controlled nitration followed by a sophisticated one-pot reductive alkylation. This innovative approach begins with the nitration of piperonyl cyclonene using dilute nitric acid in the presence of a specialized dispersing agent, specifically alkyl naphthalene sulfonic acid. This modification is not merely a procedural tweak but a fundamental process intensification that controls the physical morphology of the intermediate, ensuring uniform particle size and minimizing the entrapment of corrosive acid residues. The subsequent transformation utilizes a dual-function catalytic system to simultaneously reduce the nitro group and alkylate the resulting amine using the solvent itself, thereby eliminating the need for separate alkylation reagents and drastically simplifying the operational workflow for manufacturers seeking a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into Dispersed Nitration and Reductive Alkylation
The core innovation of this synthesis lies in the precise control of the nitration step through the use of alkyl naphthalene sulfonic acid as a dispersing agent. In standard nitration reactions without such additives, the organic substrate and the aqueous acid phase often form heterogeneous mixtures with poor interfacial contact, leading to localized hot spots and over-nitration or oxidation byproducts. The inclusion of the dispersing agent, typically at a concentration of 1-2wt% relative to the reactant gross weight, creates a stable emulsion that significantly reduces the particle diameter of the forming nitro-intermediate. This micro-scale control ensures that the nitration proceeds uniformly at moderate temperatures of 55-70°C, preventing the formation of tars and reducing the amount of residual nitric acid trapped within the crystal lattice of the product. Such purity in the intermediate stage is critical because residual oxidants can poison the sensitive metal catalysts used in the downstream hydrogenation step, a common failure point in continuous manufacturing processes.
Following the isolation of the high-quality nitro-intermediate, the process transitions to a remarkable tandem catalytic transformation involving hydrogenation and N-alkylation. Using a composite catalyst system of Copper and Raney Nickel under a hydrogen pressure of 1.5-2.0 MPa, the nitro group is first reduced to the primary amine at approximately 70°C. However, rather than isolating this unstable amine, the reaction temperature is subsequently ramped to 140-180°C in the presence of absolute ethanol. Under these thermal conditions, the freshly generated amine undergoes a condensation reaction with the ethanol solvent, facilitated by the metallic catalyst surface, to yield the N-ethyl derivative. This mechanism effectively utilizes the solvent as a reagent, converting a potential waste stream into a value-added component of the final molecule and showcasing a high degree of atom economy that is highly desirable for green chemistry initiatives.
How to Synthesize N-ethyl-3,4-(methylenedioxy)aniline Efficiently
Implementing this synthesis route requires careful attention to the specific parameters outlined in the patent to ensure optimal yield and safety. The process is designed to be scalable, moving seamlessly from laboratory validation to multi-ton production with minimal re-engineering. Operators must strictly maintain the temperature gradients during the nitration phase to prevent runaway exotherms and ensure the dispersing agent is fully active before the addition of the nitrating agent. Following the filtration and washing of the nitro-intermediate, the hydrogenation step demands rigorous exclusion of oxygen and precise pressure control to maximize catalyst turnover. For a comprehensive understanding of the exact operational parameters, stoichiometry, and safety protocols required for execution, please refer to the standardized synthesis guide below.
- Perform nitration of piperonyl cyclonene using dilute nitric acid with an alkyl naphthalene sulfonic acid dispersing agent at 55-70°C.
- Hydrogenate the resulting nitro-intermediate using Cu/Raney Ni catalyst in absolute ethanol at 1.5-2.0 MPa pressure.
- Heat the reaction mixture to 140-180°C for 10 hours to effect N-ethylation using the ethanol solvent, then isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from bromination-based routes to this nitration-hydrogenation protocol represents a strategic opportunity to de-risk the supply of critical intermediates. The elimination of elemental bromine and the associated high-pressure ammonolysis reactors removes a significant bottleneck from the production schedule, allowing for faster batch turnover and more predictable delivery timelines. Moreover, the use of commodity chemicals like dilute nitric acid and ethanol, coupled with reusable heterogeneous catalysts, insulates the manufacturing cost structure from the volatility often seen in the market for specialized halogenating agents. This stability translates directly into more consistent pricing models for long-term contracts, providing a competitive edge in cost reduction in fine chemical manufacturing without compromising on the quality of the final API precursor.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of unit operations and the extension of catalyst life. By utilizing a dispersing agent to prevent catalyst poisoning, the frequency of catalyst replacement is significantly reduced, lowering the overall consumption of expensive metals like Raney Nickel. Additionally, the one-pot nature of the reduction and alkylation steps eliminates the need for intermediate isolation, drying, and re-dissolution, which substantially cuts down on solvent usage, energy consumption for heating and cooling, and labor hours. These cumulative efficiencies result in a leaner production cost base, enabling suppliers to offer more aggressive pricing while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory and logistical complexities associated with handling hazardous halogens and managing toxic waste. This new pathway relies on widely available raw materials that are not subject to the same stringent transport restrictions as bromine or other halogen sources. The robustness of the nitration step, with its high yield of over 97% for the intermediate, ensures a steady flow of material into the hydrogenation stage, minimizing the risk of production stoppages due to off-spec intermediates. This reliability is crucial for downstream pharmaceutical manufacturers who operate on Just-In-Time inventory models and cannot afford disruptions in their raw material feed.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a markedly cleaner profile. The absence of brominated byproducts simplifies wastewater treatment and reduces the burden on effluent processing facilities. The use of dilute nitric acid instead of concentrated fuming acids further enhances operational safety by reducing corrosion risks and the potential for severe chemical burns. As global regulations on industrial emissions tighten, adopting a process that inherently generates less hazardous waste positions the manufacturer as a preferred partner for multinational corporations committed to sustainability goals and strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of N-ethyl-3,4-(methylenedioxy)aniline using this advanced catalytic route. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines.
Q: What are the advantages of the nitration-dispersion method over traditional bromination routes?
A: The patented nitration method avoids the use of hazardous brominating agents and eliminates the need for high-temperature, high-pressure ammonolysis. By using a specific dispersing agent, the process controls particle size, reduces residual nitric acid, and prevents catalyst poisoning during the subsequent hydrogenation step, leading to easier industrial implementation.
Q: How is the N-ethyl group introduced in this synthesis pathway?
A: The N-ethyl group is introduced via a reductive alkylation mechanism. After the initial reduction of the nitro group to an amine using hydrogen and a Cu/Raney Ni catalyst, the reaction temperature is raised to 140-180°C. Under these conditions, the intermediate amine reacts with the absolute ethanol solvent to form the N-ethyl derivative in a single pot.
Q: What purity levels can be achieved with this catalytic process?
A: According to the patent data, the preparation method yields N-ethyl-3,4-(methylenedioxy)aniline with a purity reaching more than 92% and an overall yield in the range of 80-85%, demonstrating high efficiency suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-ethyl-3,4-(methylenedioxy)aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals depends on the availability of high-quality, consistently supplied intermediates. Our technical team has extensively analyzed the pathway described in CN101875650A and possesses the expertise to implement this efficient nitration and reductive alkylation strategy at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-ethyl-3,4-(methylenedioxy)aniline meets the exacting standards required for GMP-grade drug substance manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods. By leveraging our process engineering capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our advanced manufacturing solutions can support your next generation of therapeutic products.
