Advanced Manufacturing of Miriplatin Hydrate: A Scalable, Chloroform-Free Route for Global Oncology Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical oncology agents. Patent CN102329338A introduces a groundbreaking preparation method for Miriplatin Hydrate, a lipophilic platinum-based antitumor drug primarily used for the treatment of liver cancer. This innovation addresses significant historical bottlenecks in platinum complex synthesis by replacing hazardous halogenated solvents with safer aqueous and alcohol-based systems. The core breakthrough lies in a novel hydrolysis strategy using silver sulfate and barium hydroxide, which ensures exceptional control over ionic impurities while maintaining high stereochemical integrity. By shifting away from traditional chloroform-dependent protocols, this technology offers a cleaner, more efficient route that aligns perfectly with modern Green Chemistry principles and stringent regulatory requirements for API manufacturing. 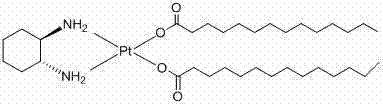
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lipophilic platinum complexes like Miriplatin has been plagued by significant technical and safety challenges. Early methodologies, such as those reported in foreign patent JP 1987000096, relied on the generation of sodium tetradecanoate, which frequently became entrapped within the crystal lattice of the final product, creating persistent impurity profiles that were extremely difficult to purge. Furthermore, subsequent approaches detailed in patents like WO94/14470 and JP11-315088 necessitated the use of chloroform as a primary reaction medium. 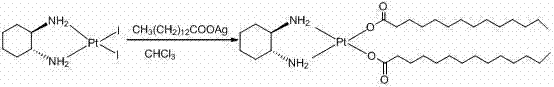
The reliance on chloroform presents severe occupational health risks and environmental liabilities, requiring complex solvent recovery systems and strict emission controls that drive up operational expenditures. Additionally, these legacy processes often suffered from prolonged reaction cycles and inconsistent yields due to the difficulty in managing the solubility of long-chain fatty acid salts in halogenated solvents. The combination of toxic solvent exposure, difficult purification workflows, and suboptimal yield consistency rendered these conventional methods increasingly obsolete for modern, high-volume commercial manufacturing.
The Novel Approach
The methodology disclosed in CN102329338A represents a paradigm shift by utilizing a water-based hydrolysis system followed by ligand exchange in n-butanol. Instead of struggling with insoluble sodium salts or toxic chloroform suspensions, this process converts the diiodo-platinum precursor into a soluble hydroxy-platinum species using silver sulfate. This transformation allows for the precise removal of iodide ions as insoluble silver iodide, which is mechanically separated via filtration. The subsequent addition of barium hydroxide effectively scavenges residual sulfate ions, generating a ultra-pure hydroxy-platinum intermediate in situ. This aqueous-compatible intermediate then reacts smoothly with tetradecanoic acid dissolved in n-butanol, precipitating the final Miriplatin Hydrate product with exceptional purity and crystallinity.
Mechanistic Insights into Silver Sulfate-Mediated Hydrolysis
The heart of this synthetic innovation is the controlled ligand exchange mechanism facilitated by silver cations. The process begins with the high-purity intermediate cis-diiodo-((1R,2R)-1,2-cyclohexanediamine) platinum (II), often referred to as cycloplatin. 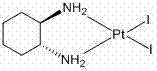
When treated with silver sulfate in an aqueous environment at moderate temperatures (50-60°C), the soft silver ions exhibit a high affinity for the iodide ligands coordinated to the platinum center. This drives the displacement of iodide to form insoluble silver iodide precipitate, leaving behind a coordinatively unsaturated platinum species that is immediately stabilized by water molecules to form the aquo-complex. The addition of barium hydroxide serves a dual purpose: it adjusts the pH to facilitate the deprotonation of the incoming fatty acid and simultaneously precipitates any remaining sulfate ions as barium sulfate. This sequential precipitation strategy ensures that the final reaction mixture contains minimal inorganic contaminants, allowing the tetradecanoic acid to coordinate cleanly to the platinum center without competing side reactions or salt entrapment.
How to Synthesize Miriplatin Hydrate Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for laboratory and pilot-scale production. The process initiates with the preparation of the cycloplatin precursor from potassium tetrachloroplatinate, followed by the critical silver-mediated hydrolysis and final fatty acid coupling. Detailed operational parameters regarding stoichiometry, temperature control, and washing procedures are essential to achieving the reported high yields and purity specifications. For process engineers and R&D teams looking to implement this technology, the following standardized workflow encapsulates the critical unit operations required for successful execution.
- Synthesize the intermediate cis-diiodo-((1R,2R)-1,2-cyclohexanediamine) platinum (II) by reacting K2PtCl4 with KI and (1R,2R)-1,2-diaminocyclohexane in water.
- Hydrolyze the intermediate using silver sulfate in purified water at 50-60°C to replace iodide ligands, followed by filtration to remove silver iodide precipitate.
- Treat the filtrate with barium hydroxide octahydrate to remove sulfate ions, then react the resulting hydroxy-platinum solution with tetradecanoic acid in n-butanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of chloroform removes a major regulatory hurdle and significantly reduces the costs associated with hazardous waste disposal and solvent recovery infrastructure. By utilizing water and n-butanol, the process leverages commodity solvents that are readily available globally, mitigating supply chain volatility and reducing raw material procurement risks. Furthermore, the simplified purification workflow, which relies on straightforward filtration and washing rather than complex chromatography or recrystallization from hazardous mixtures, drastically shortens the overall production cycle time.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive solvent recovery steps and the reduction of batch failure rates associated with difficult-to-remove impurities. By avoiding the formation of entrapped sodium salts, the need for repetitive recrystallization is minimized, leading to substantial savings in energy consumption and labor hours. Additionally, the use of non-halogenated solvents lowers the insurance and compliance costs typically associated with handling Class 1 or Class 2 carcinogenic solvents, resulting in a leaner cost structure for the final API.
- Enhanced Supply Chain Reliability: Reliance on chloroform often subjects manufacturers to strict quota limitations and fluctuating market prices due to its status as a controlled substance. In contrast, the reagents used in this novel method, such as silver sulfate, barium hydroxide, and n-butanol, are bulk industrial chemicals with stable supply chains. This shift ensures consistent production scheduling and protects against interruptions caused by regulatory changes in solvent availability, thereby guaranteeing a more reliable supply of high-purity Miriplatin Hydrate to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, operating under normal atmospheric pressure and moderate temperatures that do not require specialized high-pressure reactors or cryogenic cooling systems. The aqueous nature of the hydrolysis step facilitates excellent heat transfer and mixing in large vessels, ensuring uniform reaction kinetics even at metric-ton scales. Moreover, the reduction in toxic solvent usage aligns with increasingly stringent global environmental regulations, positioning manufacturers as responsible corporate citizens and simplifying the environmental impact assessment process for new facility approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and operational safety. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the new synthesis method for Miriplatin Hydrate superior to conventional chloroform-based routes?
A: The novel method described in patent CN102329338A eliminates the use of chloroform, a toxic solvent harmful to human health and the environment. Furthermore, it avoids the formation of sodium tetradecanoate impurities which are difficult to remove in older methods, ensuring higher product purity and easier downstream processing.
Q: How does the silver sulfate hydrolysis step improve impurity control?
A: By utilizing silver sulfate for hydrolysis, the process effectively precipitates iodide ions as silver iodide, which is easily filtered out. Subsequent treatment with barium hydroxide removes residual sulfate ions as barium sulfate, resulting in a highly pure trans-hydroxy-platinum intermediate ready for final ligand exchange.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for scalability. It operates under normal temperature and pressure conditions, uses water and n-butanol instead of hazardous halogenated solvents, and achieves high yields with simplified purification steps, making it ideal for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Miriplatin Hydrate Supplier
The technological advancements detailed in patent CN102329338A underscore the immense potential for optimizing the supply chain of critical oncology intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards for pharmaceutical intermediates.
We invite global partners to collaborate with us on optimizing their Miriplatin Hydrate supply chain through this advanced, eco-friendly methodology. By leveraging our technical expertise, you can achieve significant operational efficiencies and secure a stable source of high-quality material. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your volume requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
