Advanced Manufacturing of High-Stability Tiopronin for Global Pharmaceutical Supply Chains
Introduction to Advanced Tiopronin Manufacturing Technology
The pharmaceutical industry continuously demands higher standards for active pharmaceutical ingredients (APIs), particularly for hepatoprotective agents where purity and stability are paramount for patient safety. Patent CN111217727A introduces a groundbreaking preparation method for Tiopronin, chemically known as N-(2-mercaptopropionyl)-glycine, which addresses critical deficiencies in existing manufacturing technologies. This novel synthetic route leverages a precise stoichiometric balance between glycine and 2-chloropropionyl chloride, coupled with a sophisticated sulfuration-reduction sequence, to achieve yields exceeding 75% and purity levels greater than 99.8%. Unlike traditional methods that often suffer from significant pollution and inconsistent product quality, this innovation ensures a robust impurity profile that remains stable even under rigorous environmental stress conditions. The structural integrity of the final molecule is preserved through meticulous control of reaction parameters, including pH regulation and temperature gradients during the crystallization phases. For global procurement teams, this represents a shift towards a more reliable Tiopronin supplier capable of delivering material that meets the stringent specifications required for modern therapeutic formulations.
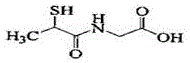
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tiopronin has been plagued by inherent inefficiencies that compromise both economic viability and product quality. Conventional routes often utilize harsh reaction conditions that lead to the formation of complex by-product mixtures, necessitating extensive and costly purification steps that erode overall yield. Furthermore, the resulting API frequently exhibits poor stability profiles, with related substances increasing dramatically upon exposure to standard storage conditions such as high temperature or humidity. This instability poses a severe risk to the supply chain, as degradation during transit or warehousing can render entire batches unsuitable for use, leading to significant financial losses and production delays. The reliance on less optimized catalytic systems in older patents often results in residual metal contaminants or unreacted starting materials that are difficult to remove to acceptable pharmacopeial limits. Consequently, manufacturers face a constant struggle to balance cost reduction in hepatoprotective API manufacturing with the uncompromising need for high-purity Tiopronin that ensures therapeutic efficacy.
The Novel Approach
The methodology disclosed in the patent fundamentally reengineers the synthesis pathway to overcome these historical bottlenecks through a refined two-step process. By strictly controlling the weight ratio of glycine to 2-chloropropionyl chloride and employing a specific extraction protocol with ethyl acetate, the first step efficiently generates the 2-chloropropionylglycine intermediate with minimal side reactions. The subsequent transformation involves a unique combination of sodium sulfide, sublimed sulfur, and zinc powder, which facilitates a clean conversion to the thiol functionality while suppressing the formation of disulfide dimers or other oxidative impurities. This approach not only enhances the chemical yield but also drastically improves the physical stability of the final crystal lattice. Comparative data indicates that while commercial samples may see impurity levels surge to over 10% under high-temperature stress, the product from this new method maintains impurity levels below 0.15%, demonstrating exceptional robustness. This technological leap ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed with confidence in the consistency and longevity of the final product.
Mechanistic Insights into Acylation and Reductive Thiolation
The core of this synthetic innovation lies in the precise mechanistic control exerted during the acylation and subsequent thiolation stages. In the initial step, the reaction is conducted under strongly alkaline conditions (pH 11-12) using sodium hydroxide, which serves to deprotonate the amino group of glycine, thereby enhancing its nucleophilicity for the attack on the acid chloride. This Schotten-Baumann type acylation is carefully managed with cooling to 5°C to prevent hydrolysis of the acid chloride, ensuring that the electrophile reacts exclusively with the amine rather than water. The use of ethyl acetate for extraction is critical, as it selectively partitions the organic intermediate away from inorganic salts, setting the stage for a high-purity downstream process. Following isolation, the intermediate undergoes a nucleophilic substitution where the chlorine atom is displaced by a sulfur species generated in situ. The generation of a sodium disulfide solution via the reaction of sodium sulfide and sublimed sulfur creates a potent nucleophile that effectively targets the alpha-carbon of the propionyl group. Finally, the addition of zinc powder acts as a reducing agent to cleave any formed disulfide bonds, ensuring the final product exists predominantly in the free thiol form, which is the biologically active configuration required for its chelating and detoxifying properties.
Impurity control is intrinsically built into this mechanism through the sequential addition of reagents and strict temperature controls. The slow dropwise addition of 2-chloropropionyl chloride prevents local exotherms that could trigger polymerization or degradation of the sensitive glycine backbone. Similarly, the post-reaction acidification with sulfuric acid is performed at low temperatures (5-15°C) to precipitate the product while keeping soluble impurities in the aqueous phase. The decolorization step using activated carbon further polishes the solution, removing trace organic colored bodies that often persist in crude thiol syntheses. By optimizing the vacuum drying parameters to between 45-55°C, the process avoids thermal degradation of the mercapto group, which is prone to oxidation at higher temperatures. This comprehensive attention to mechanistic detail results in a product with a remarkably clean impurity profile, satisfying the rigorous demands of regulatory bodies for high-purity Tiopronin used in treating viral and alcoholic hepatitis.
How to Synthesize Tiopronin Efficiently
The synthesis of Tiopronin via this patented route offers a streamlined pathway that balances operational simplicity with high-performance outcomes, making it an ideal candidate for technology transfer. The process begins with the preparation of the key intermediate, 2-chloropropionylglycine, which serves as the scaffold for the final molecule. Operators must adhere strictly to the specified weight ratios and pH controls to maximize the efficiency of the acylation step. Following the isolation of the intermediate, the focus shifts to the sulfuration reactor, where the generation of the active sulfur species requires careful thermal management to ensure complete dissolution of sublimed sulfur. The final reduction and crystallization steps are designed to maximize recovery while ensuring the crystal form possesses the desired stability characteristics. For a detailed breakdown of the specific operational parameters, reagent quantities, and safety protocols required to execute this synthesis, please refer to the standardized guide below.
- Prepare 2-chloropropionylglycine by reacting glycine with 2-chloropropionyl chloride in an aqueous sodium hydroxide solution, maintaining pH 11-12, followed by ethyl acetate extraction and crystallization.
- Generate a sodium disulfide solution by reacting sodium sulfide with sublimed sulfur in purified water at elevated temperatures.
- React the intermediate with the disulfide solution, followed by reduction with zinc powder and acidification to isolate high-purity Tiopronin crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced Tiopronin synthesis method offers substantial strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the drastic simplification of the purification train; because the reaction inherently produces fewer by-products, the need for extensive chromatographic purification or multiple recrystallizations is eliminated. This reduction in processing steps translates directly into lower utility consumption, reduced solvent waste, and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the exceptional stability of the final product mitigates the risk of inventory write-offs due to degradation, ensuring that stock held in warehouses retains its value over extended periods. This reliability allows for more flexible logistics planning and reduces the pressure on just-in-time delivery schedules, providing a buffer against global supply chain disruptions. Ultimately, this process enables a more resilient and cost-effective supply of critical hepatoprotective agents to the global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like zinc powder and sodium sulfide significantly lowers the raw material cost base. Additionally, the high yield (>75%) means that less starting material is required to produce the same amount of API, effectively spreading fixed costs over a larger output volume. The simplified workup procedure reduces labor hours and equipment occupancy time, further driving down the cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the synthesis route ensures consistent batch-to-batch quality, reducing the frequency of out-of-specification (OOS) results that can halt production lines. The use of widely available raw materials minimizes the risk of supply shortages for critical reagents, ensuring continuity of operations even during market fluctuations. Moreover, the high stability of the finished Tiopronin reduces the need for specialized cold-chain logistics, allowing for standard shipping methods that are faster and more economical.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor types and separation techniques that are easily replicated from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste and the efficient use of solvents facilitate easier compliance with increasingly stringent environmental regulations. This forward-looking design ensures that manufacturing facilities can operate sustainably while meeting the growing global demand for liver health therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Tiopronin synthesized via this novel method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the technology's value proposition. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing supplier capabilities. The answers highlight the specific advantages in terms of purity, stability, and operational efficiency that distinguish this method from legacy technologies.
Q: How does the new Tiopronin synthesis method improve stability compared to conventional processes?
A: The novel method utilizes a specific ratio of reactants and a controlled zinc powder reduction step that minimizes the formation of unstable impurities. Stress testing demonstrates that the resulting product maintains over 99% purity even under high-temperature conditions, whereas commercial alternatives show significant degradation.
Q: What are the critical quality parameters for Tiopronin in hepatoprotective applications?
A: Critical parameters include a purity level exceeding 99.8%, low related substance content (below 0.15% even after stress), and specific crystal form characteristics that ensure consistent dissolution and bioavailability in final drug formulations.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process relies on standard unit operations such as liquid-liquid extraction, vacuum concentration, and crystallization, using readily available raw materials like glycine and zinc powder, making it highly suitable for commercial scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tiopronin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of essential pharmaceutical ingredients like Tiopronin. Our team of expert chemists has extensively analyzed this patented route and possesses the technical capability to implement it seamlessly within our existing infrastructure. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, including the >99.8% benchmark set by this innovative technology. We are committed to being a partner that not only supplies materials but also adds value through technical expertise and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to bring high-quality, stable, and cost-effective Tiopronin to the patients who need it most, leveraging our shared commitment to excellence in pharmaceutical manufacturing.
