Advanced Enzymatic Synthesis of Deuterated Sulfamethazol-d4 for Commercial Scale-up and High-Purity Standards
The global demand for precise analytical standards in food safety monitoring has intensified, particularly concerning veterinary drug residues in animal-derived products. Patent CN110441447B introduces a groundbreaking synthetic methodology for deuterated labeled sulfamethazol-d4, addressing critical gaps in the availability of domestic isotopic standards. This technology leverages a sophisticated enzymatic deacetylation strategy to overcome the inherent instability of the isoxazole ring found in traditional acid-base hydrolysis methods. By utilizing stable isotope-labeled precursors, this process ensures high isotopic abundance and structural integrity, which are paramount for accurate LC-MS/MS quantification. As a reliable veterinary drug intermediate supplier, understanding this technological shift is essential for procurement teams aiming to secure high-purity deuterated standards that meet stringent national food safety regulations such as Ministry of Agriculture Announcement No. 1077-1-2008.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deacetylation of sulfonamide precursors relied heavily on harsh acidic or alkaline hydrolysis conditions, which posed significant challenges for molecules containing sensitive heterocyclic structures. In the specific case of sulfamethazol derivatives, the isoxazole ring is highly susceptible to nucleophilic attack under extreme pH conditions, leading to ring opening and irreversible degradation of the target molecule. This chemical instability results in substantially reduced yields and complicates the purification process, as numerous byproducts must be separated from the desired isotopic standard. Furthermore, the use of strong acids and bases necessitates specialized corrosion-resistant equipment and generates hazardous waste streams, increasing both the operational complexity and the environmental burden of the manufacturing process. For a procurement manager focused on cost reduction in pharmaceutical intermediate manufacturing, these inefficiencies translate directly into higher production costs and inconsistent supply quality.
The Novel Approach
The innovative route disclosed in the patent circumvents these chemical pitfalls by employing a protease-catalyzed hydrolysis mechanism under mild physiological conditions. This biocatalytic approach exhibits exceptional chemoselectivity, targeting only the amide bond of the acetyl group while leaving the fragile isoxazole ring completely intact. The reaction proceeds efficiently in a mixed solvent system of methanol and water at temperatures ranging from 25°C to 35°C, eliminating the need for energy-intensive heating or cooling cycles. This gentle processing environment not only preserves the structural fidelity of the deuterated label but also simplifies the workup procedure, allowing for straightforward isolation of the product through pH adjustment and filtration. By adopting this novel approach, manufacturers can achieve superior reproducibility and significantly enhanced product yields, establishing a robust foundation for the commercial scale-up of complex sulfonamides.
Mechanistic Insights into Protease-Catalyzed Deacetylation
The core mechanistic advantage of this synthesis lies in the specific interaction between the protease enzyme and the N-acetylsulfamethazol-d4 substrate. Proteases such as trypsin, chymotrypsin, and papain function by recognizing specific peptide-like bonds, and in this context, they effectively hydrolyze the acetamido group without affecting other functional groups on the benzene ring or the heterocycle. The reaction kinetics are optimized by maintaining a specific solute-to-water volume ratio of 4:1 in the solvent mixture, which balances enzyme activity with substrate solubility. This precise control over the reaction medium ensures that the enzymatic catalysis proceeds at an optimal rate, minimizing the reaction time required to reach completion compared to uncatalyzed thermal hydrolysis. For R&D directors evaluating process feasibility, this level of mechanistic control offers a predictable and scalable pathway that reduces the risk of batch-to-batch variability often associated with harsh chemical treatments.
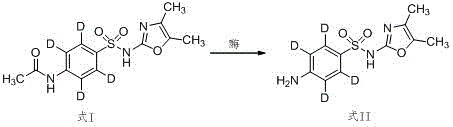
Impurity control is another critical aspect where this enzymatic mechanism excels, particularly regarding the preservation of isotopic purity. Traditional chemical hydrolysis often leads to hydrogen-deuterium exchange or structural scrambling due to the aggressive reaction conditions, which compromises the accuracy of the internal standard in mass spectrometry applications. In contrast, the mild enzymatic environment prevents such isotopic dilution, ensuring that the deuterium atoms remain securely positioned on the benzene ring throughout the synthesis. The subsequent workup involves simple acidification and neutralization steps that precipitate the product as a white solid, facilitating easy removal of the enzyme and solvent residues. This high level of purity is essential for meeting the rigorous quality specifications required for internal standard medicines used in regulatory testing laboratories.
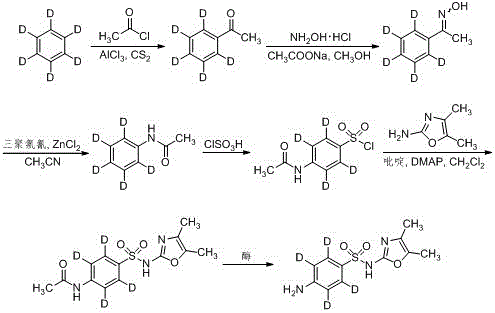
How to Synthesize Sulfamethazol-d4 Efficiently
The synthesis of this high-value isotopic standard follows a logical six-step sequence starting from readily available deuterated benzene, designed to maximize atom economy and operational safety. The initial steps involve classical organic transformations such as Friedel-Crafts acetylation and Beckmann rearrangement to construct the deuterated aniline core, which serves as the scaffold for the sulfonamide functionality. The final and most critical transformation is the enzymatic deacetylation, which unlocks the free amine necessary for the biological activity and detection characteristics of the molecule. Detailed standardized synthesis steps see the guide below, which outlines the specific reagent ratios, temperature controls, and workup procedures validated in the patent examples to ensure consistent high-yield production.
- Friedel-Crafts acetylation of benzene-d6 with acetyl chloride using aluminum trichloride to form acetophenone-d5.
- Oximation and Beckmann rearrangement to convert acetophenone-d5 into N-acetanilide-d5.
- Chlorosulfonylation followed by amidation and final enzymatic deacetylation using protease at 25-35°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this enzymatic synthesis route offers substantial commercial advantages by fundamentally altering the cost structure and risk profile of producing deuterated veterinary standards. The elimination of harsh chemical reagents reduces the dependency on specialized corrosion-resistant infrastructure, thereby lowering capital expenditure requirements for manufacturing facilities. Additionally, the mild reaction conditions decrease energy consumption and simplify waste treatment protocols, contributing to significant cost savings in utility and environmental compliance management. For supply chain heads concerned with continuity, the use of common industrial enzymes and solvents ensures that raw material availability remains stable, reducing the lead time for high-purity isotopic labels that are often subject to import restrictions. This process resilience translates into a more reliable supply chain capable of meeting the fluctuating demands of food safety monitoring programs without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The transition from aggressive acid-base hydrolysis to enzymatic catalysis removes the need for expensive corrosion-resistant reactors and extensive neutralization waste treatment systems. By operating at near-ambient temperatures, the process drastically reduces energy consumption associated with heating and cooling, leading to lower operational expenditures. Furthermore, the high selectivity of the enzyme minimizes the formation of byproducts, which reduces the complexity and cost of downstream purification processes. These cumulative efficiencies result in a more economically viable production model that allows for competitive pricing without sacrificing the high purity required for analytical standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available proteases and common organic solvents mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Unlike proprietary catalysts that may have single-source dependencies, the enzymes specified in this patent, such as trypsin and papain, are widely produced for various industries, ensuring a robust and diversified supply base. This accessibility allows manufacturers to maintain larger inventory buffers and respond more quickly to urgent procurement requests from regulatory bodies and testing laboratories. Consequently, partners can expect improved on-time delivery performance and greater flexibility in order volumes, strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: The aqueous-based nature of the enzymatic step aligns perfectly with green chemistry principles, significantly reducing the generation of hazardous organic waste streams. Scaling this process from laboratory to commercial production does not require exponential increases in safety measures, as the reaction does not involve exothermic runaways or toxic gas evolution. This inherent safety profile facilitates easier regulatory approval for new manufacturing sites and simplifies the permitting process for capacity expansion. Companies adopting this technology demonstrate a commitment to sustainable manufacturing practices, which is increasingly becoming a key criterion in vendor selection processes for multinational corporations and government agencies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterated sulfamethazol-d4, based on the specific innovations detailed in the patent literature. These insights are designed to clarify the operational benefits and quality assurances associated with this enzymatic synthesis route for stakeholders involved in procurement and quality control. Understanding these details helps in making informed decisions when selecting a partner for the supply of critical analytical reference materials.
Q: Why is enzymatic hydrolysis preferred over acid-base methods for Sulfamethazol-d4?
A: Traditional acid or base hydrolysis causes hydrolysis of the sensitive isoxazole ring, leading to low yields. Enzymatic hydrolysis offers specific selectivity for the amide bond, preserving the ring structure and ensuring high isotopic abundance.
Q: What are the optimal conditions for the protease catalysis step?
A: The process utilizes proteases such as trypsin or papain in a methanol-water solvent system (4:1 ratio) at mild temperatures between 25°C and 35°C, ensuring stability and reproducibility.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the route uses readily available raw materials like benzene-d6 and avoids extreme conditions. The mild enzymatic step simplifies downstream processing, making it highly suitable for commercial scale-up of complex veterinary intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfamethazol-d4 Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic technologies like the enzymatic deacetylation route described in CN110441447B to deliver superior isotopic standards to the global market. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards, guaranteeing the isotopic abundance and chemical integrity required for accurate LC-MS/MS analysis. We understand the critical nature of food safety testing and are committed to providing materials that empower your laboratory to achieve reliable and compliant results.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific analytical requirements. By requesting a Customized Cost-Saving Analysis, you can explore how our optimized enzymatic process translates into tangible value for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us partner with you to secure a stable supply of high-quality deuterated standards that enhance the accuracy and efficiency of your veterinary residue monitoring programs.
