Advanced Synthesis of Linezolid Degradation Impurities for Global Quality Control Standards
The pharmaceutical industry faces relentless pressure to ensure the highest standards of safety and efficacy for critical antibiotics like Linezolid, particularly in injectable formulations where stability is paramount. Patent CN111925343A introduces a groundbreaking synthesis method for a specific Linezolid degradation impurity, designated as Formula (II), which serves as an essential reference standard for quality control protocols. This innovation addresses a significant gap in the market where effective synthetic routes for this specific degradation product were previously scarce or inefficient. By leveraging a sophisticated dual-catalyst system involving alkali metal alkoxides and solid base catalyst resins, the patented process achieves exceptional purity levels exceeding 98% and yields greater than 92%. For R&D directors and quality assurance teams, this development represents a pivotal advancement in the ability to monitor and control the degradation profile of Linezolid injections, ensuring compliance with stringent global regulatory requirements such as those mandated by the FDA and EMA.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific degradation impurities for oxazolidinone antibiotics has been fraught with challenges related to low selectivity and cumbersome purification processes. Conventional hydrolysis methods often rely on harsh conditions or单一 catalysts that fail to distinguish between the desired degradation pathway and competing side reactions, leading to complex mixtures of byproducts. These impurities not only reduce the overall yield of the target compound but also necessitate extensive downstream processing, such as multi-step column chromatography, to achieve the purity required for analytical reference standards. Furthermore, traditional solvents may not provide the optimal environment for the specific hydrolytic cleavage required to generate Formula (II) from Defluorinated Linezolid, resulting in incomplete conversions and significant material loss. For procurement managers, these inefficiencies translate into higher costs of goods sold and extended lead times due to the complexity of isolating the target molecule from a dirty reaction matrix.
The Novel Approach
The methodology disclosed in CN111925343A revolutionizes this landscape by introducing a highly selective hydrolysis protocol that utilizes a unique combination of solvents and catalysts. By employing a solvent system composed of esters with fewer than six carbon atoms, such as ethyl acetate or isopropyl acetate, mixed with water in a precise volume ratio, the reaction environment is optimized for both solubility and reactivity. The true breakthrough lies in the dual-catalyst strategy, which pairs a homogeneous alkali metal alkoxide with a heterogeneous solid base catalyst resin, specifically A-8XMP. This synergistic effect dramatically improves the conversion rate of Defluorinated Linezolid while suppressing the formation of unwanted byproducts. As illustrated in the reaction scheme below, this approach allows for the direct transformation of the starting material into the target degradation impurity with remarkable efficiency.
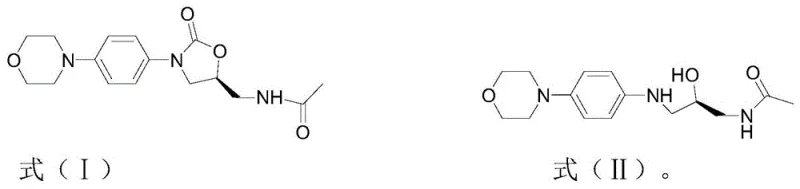
Consequently, the need for labor-intensive purification steps is virtually eliminated, as the crude product obtained after simple workup already possesses high purity. This streamlined workflow not only accelerates the production timeline but also significantly reduces the consumption of silica gel and organic solvents typically associated with chromatographic separation, offering a greener and more economically viable alternative for industrial-scale manufacturing.
Mechanistic Insights into Dual-Catalyst Hydrolysis
The mechanistic elegance of this synthesis lies in the cooperative interaction between the homogeneous and heterogeneous catalytic species. The alkali metal alkoxide, such as sodium methoxide or sodium tert-butoxide, acts as a strong base that initiates the nucleophilic attack on the oxazolidinone ring of the Defluorinated Linezolid substrate. This initial activation is crucial for overcoming the energy barrier associated with the ring-opening hydrolysis. However, relying solely on a strong homogeneous base can lead to over-reaction or non-selective degradation of other sensitive functional groups within the molecule. This is where the solid base catalyst resin A-8XMP plays a pivotal role by providing a controlled basic environment on its surface. The porous structure of the resin likely facilitates the adsorption of reactants in a specific orientation that favors the formation of the desired hydrolysis product, Formula (II), while sterically hindering pathways that lead to impurities.
Furthermore, the choice of the ester-water solvent system is not merely for dissolution but actively participates in stabilizing the transition states of the reaction. The ester component ensures that the organic substrate remains in solution, while the water acts as the nucleophile for the hydrolysis step. The specific volume ratio of 2:1 to 3:1 creates a micro-emulsion or biphasic interface that enhances mass transfer between the solid catalyst, the liquid reagents, and the substrate. This optimized interfacial contact ensures that the reaction proceeds rapidly at reflux temperatures, typically completing within 2.5 to 3.5 hours. From an impurity control perspective, this mechanism effectively minimizes the generation of regio-isomers or over-hydrolyzed species, which are common pitfalls in less optimized protocols. The result is a clean reaction profile that simplifies the isolation process to a straightforward extraction and washing sequence, yielding a product that meets the rigorous specifications required for pharmaceutical reference materials.
How to Synthesize Linezolid Degradation Impurity Efficiently
The implementation of this synthesis route is designed for operational simplicity and robustness, making it highly suitable for technology transfer from laboratory to pilot plant scales. The process begins with the preparation of the reaction vessel, where the specific ester solvent and water are combined to create the reaction medium. Defluorinated Linezolid is then introduced, followed by the sequential addition of the alkali metal alkoxide and the solid base catalyst resin. The mixture is heated to reflux, maintaining a temperature that ensures vigorous mixing and optimal reaction kinetics without degrading the thermal sensitivity of the intermediates. Monitoring via TLC indicates the disappearance of the starting material, signaling the completion of the transformation. Following the reaction, the workup procedure is notably mild, involving the addition of dichloromethane for phase separation and subsequent washing with dilute hydrochloric acid to neutralize residual bases.
- Prepare the reaction mixture by dissolving Defluorinated Linezolid in a solvent system comprising an ester (e.g., ethyl acetate) and water in a specific volume ratio.
- Introduce the dual catalyst system consisting of an alkali metal alkoxide and a solid base catalyst resin (A-8XMP) to the mixture under stirring.
- Heat the reaction to reflux for approximately 2.5 to 3.5 hours, followed by a simplified workup involving dichloromethane extraction and acid washing to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers substantial strategic advantages that extend beyond mere technical feasibility. The elimination of complex purification steps, specifically the removal of column chromatography, represents a significant reduction in operational expenditures. Chromatography is notoriously resource-intensive, requiring large quantities of high-grade silica gel and vast volumes of organic solvents, both of which contribute heavily to waste disposal costs and raw material expenses. By achieving high purity directly from the reaction workup, manufacturers can drastically lower their variable costs per kilogram of product. Additionally, the use of commodity chemicals such as ethyl acetate, sodium methoxide, and commercially available catalyst resins ensures a stable and resilient supply chain. These materials are produced globally in massive quantities, mitigating the risk of shortages that often plague specialty reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the simplification of the downstream processing train. Traditional methods for synthesizing complex pharmaceutical impurities often require multiple recrystallizations or preparative HPLC separations to reach reference-grade purity, which are time-consuming and expensive. In contrast, this novel route achieves purity levels exceeding 98% through a simple extractive workup. This reduction in unit operations translates directly into lower labor costs, reduced energy consumption for solvent recovery, and minimized equipment occupancy time. Furthermore, the high yield reported in the patent examples implies that less starting material is wasted, maximizing the return on investment for the raw Defluorinated Linezolid. The avoidance of transition metals or exotic catalysts also removes the need for costly metal scavenging steps, further streamlining the cost structure.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for the production of quality control standards, as any disruption can halt the release of finished drug batches. This synthesis method relies on a robust set of reagents that are widely available from multiple global suppliers, reducing dependency on single-source vendors. The solid base catalyst resin, being a heterogeneous catalyst, can potentially be recovered and reused, adding another layer of supply security and cost efficiency. Moreover, the reaction conditions are relatively mild, utilizing standard glass-lined or stainless steel reactors capable of handling reflux temperatures, which means the process can be executed in most existing multipurpose chemical facilities without requiring capital-intensive retrofitting. This flexibility allows for rapid scaling to meet fluctuating demand for Linezolid impurity standards.
- Scalability and Environmental Compliance: As regulatory bodies increasingly emphasize green chemistry principles, the environmental footprint of synthesis routes becomes a key selection criterion. This process scores highly on sustainability metrics due to its high atom economy and the use of less hazardous solvents compared to traditional chlorinated or aromatic solvents often found in older literature. The significant reduction in solvent usage, driven by the absence of chromatography, lowers the volume of hazardous waste generated per batch. This not only reduces disposal fees but also simplifies the permitting process for manufacturing sites. The scalability is further enhanced by the fact that the reaction kinetics are well-defined and reproducible, minimizing the risk of batch failures during scale-up. This reliability ensures a consistent supply of high-quality impurity standards, supporting the long-term commercial viability of Linezolid formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this Linezolid degradation impurity. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their quality control workflows.
Q: Why is the synthesis of Linezolid degradation impurities critical for pharmaceutical manufacturers?
A: Linezolid injections are prone to generating degradation impurities during sterilization and storage. Having a high-purity reference standard for these impurities is mandatory for regulatory compliance and ensuring patient safety.
Q: How does the dual catalyst system improve the synthesis compared to traditional methods?
A: The combination of alkali metal alkoxide and solid base resin A-8XMP significantly enhances reaction selectivity, minimizing side reactions and eliminating the need for complex column chromatography purification.
Q: What are the scalability benefits of this specific hydrolysis process?
A: The process utilizes readily available solvents like ethyl acetate and robust catalysts, allowing for straightforward scale-up from laboratory to commercial production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linezolid Degradation Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity reference standards play in the lifecycle management of essential antibiotics like Linezolid. Our team of expert chemists has thoroughly analyzed the synthetic route described in CN111925343A and is fully prepared to execute this protocol with precision and scalability. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply of Linezolid degradation impurities remains uninterrupted regardless of market demand fluctuations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of material we deliver meets the exacting standards required for regulatory submissions and stability testing.
We invite pharmaceutical manufacturers and quality control laboratories to collaborate with us to optimize their supply chains for Linezolid impurities. By leveraging our technical expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your quality control strategies are supported by the most reliable and efficient synthesis technologies available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
