Advanced BTC-Mediated Vilsmeier Cyclization for High-Purity 3-Formyl Chromone Derivatives Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with environmental compliance, a challenge effectively addressed by the methodology disclosed in patent CN100436441C. This intellectual property introduces a refined protocol for the preparation of 3-formyl chromone derivatives, which serve as critical scaffolds in the development of antibiotics, antivirals, and cardiovascular agents. Unlike conventional approaches that rely heavily on corrosive and polluting phosphorus reagents, this innovation utilizes bis(trichloromethyl) carbonate (BTC) in conjunction with N,N-dimethylformamide (DMF) to generate the active Vilsmeier reagent in situ. The strategic substitution of traditional reagents not only mitigates the severe environmental burden associated with phosphorus waste but also stabilizes the reaction profile, allowing for the efficient synthesis of complex heterocyclic structures under remarkably mild thermal conditions.
Furthermore, the versatility of this synthetic route is evidenced by its ability to accommodate a wide array of substituents on the starting o-hydroxyacetophenone backbone without significant loss in yield or purity. The process operates within a controlled temperature window, initially maintaining cryogenic conditions to manage exotherms during reagent mixing, followed by a gentle warming phase to drive the cyclization to completion. This level of control is paramount for industrial applications where reproducibility and safety are non-negotiable. By leveraging this technology, manufacturers can access high-value chromone intermediates with reduced downstream purification burdens, positioning this patent as a cornerstone for modernizing the supply chain of bioactive heterocycles.
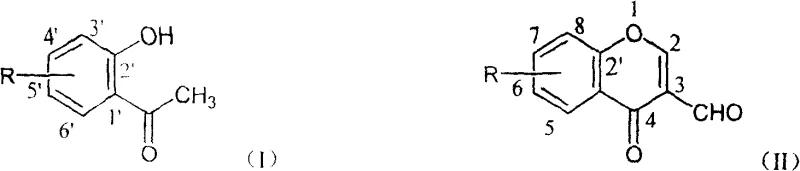
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-formyl chromone derivatives has been predominantly achieved through the classical Vilsmeier-Haack reaction utilizing phosphorus oxychloride (POCl3) and DMF. While chemically effective, this legacy methodology presents substantial operational and environmental liabilities that hinder its sustainability in modern green chemistry frameworks. The primary drawback lies in the generation of significant quantities of phosphorus-containing wastewater, which requires complex and costly treatment processes to meet increasingly stringent global environmental regulations regarding eutrophication and aquatic toxicity. Additionally, the use of POCl3 poses severe handling risks due to its corrosive nature and tendency to release hazardous hydrogen chloride gas upon exposure to moisture, necessitating specialized corrosion-resistant equipment and rigorous safety protocols that inflate capital expenditure.
Beyond the environmental and safety concerns, the conventional POCl3-mediated route suffers from inconsistent performance when applied to substrates with varying electronic properties. Literature and practical experience indicate that yields fluctuate dramatically depending on the nature of the substituents on the aromatic ring; specifically, substrates bearing strong electron-donating groups or strong electron-withdrawing groups often result in markedly lower conversion rates and difficult-to-remove impurities. This lack of universality forces process chemists to develop customized, substrate-specific optimization campaigns for each new derivative, leading to prolonged development timelines and inefficient resource allocation. Furthermore, the harsh acidic conditions often promote side reactions such as polymerization or decomposition of sensitive functional groups, complicating the isolation of the target chromone and reducing the overall atom economy of the process.
The Novel Approach
In stark contrast to the limitations of phosphorus-based reagents, the novel approach detailed in the patent data employs bis(trichloromethyl) carbonate (BTC), commonly known as triphosgene, as a safe and solid alternative for generating the chlorinating species required for the Vilsmeier transformation. This method fundamentally alters the reaction landscape by eliminating phosphorus from the waste stream entirely, thereby transforming a hazardous waste management problem into a manageable organic solvent recovery operation. The reaction proceeds in C1-C4 halogenated hydrocarbon solvents, such as 1,2-dichloroethane or dichloromethane, which allows for precise temperature control and homogeneous mixing of the solid BTC precursor with liquid DMF. The resulting Vilsmeier reagent is generated smoothly at low temperatures, minimizing the risk of runaway exotherms that are characteristic of liquid POCl3 additions.
The operational simplicity of this new route is further enhanced by its broad substrate tolerance, effectively solving the yield variability issues plaguing earlier methods. Whether the starting o-hydroxyacetophenone derivative contains electron-rich methoxy groups or electron-deficient nitro and halogen substituents, the BTC-mediated system consistently delivers high yields, often exceeding 80% and reaching up to 93% in optimized embodiments. This robustness implies that a single, standardized operating procedure can be applied to a diverse library of chromone precursors, significantly accelerating the scale-up process for new drug candidates. Moreover, the mild reaction temperatures, ranging from 0°C during addition to a maximum of 30°C during the cyclization phase, preserve the integrity of sensitive functional groups, ensuring that the final product possesses the high purity required for pharmaceutical applications without the need for extensive chromatographic purification.
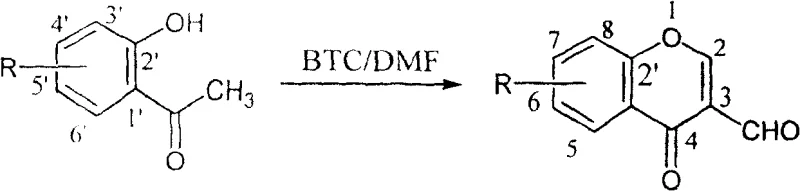
Mechanistic Insights into BTC-Mediated Vilsmeier Cyclization
The mechanistic pathway of this transformation begins with the nucleophilic attack of the DMF oxygen on the carbonyl carbon of the bis(trichloromethyl) carbonate, leading to the formation of an unstable intermediate that rapidly decomposes to release phosgene equivalents in situ. These reactive species immediately interact with a second equivalent of DMF to generate the active chloroiminium salt, which serves as the potent electrophile in the subsequent Vilsmeier-Haack reaction. This in-situ generation is crucial as it avoids the handling of free phosgene gas while maintaining a steady, controlled concentration of the active reagent throughout the reaction vessel. The electrophilic chloroiminium ion then attacks the electron-rich enol tautomer of the o-hydroxyacetophenone derivative at the alpha-carbon position, forming a C-C bond and establishing the precursor for the heterocyclic ring closure.
Following the initial formylation, the intramolecular cyclization occurs through the nucleophilic attack of the phenolic hydroxyl group onto the activated carbonyl carbon, facilitated by the elimination of dimethylamine and hydrochloric acid. The mild thermal conditions of 25-30°C provide sufficient activation energy for this cyclization step without promoting the degradation of the newly formed chromone ring or the aldehyde functionality. From an impurity control perspective, the use of BTC minimizes the formation of phosphorylated byproducts that are notoriously difficult to separate from the target molecule in traditional methods. The clean reaction profile ensures that the crude product primarily consists of the desired 3-formyl chromone, unreacted starting material, and soluble amine salts, all of which can be effectively removed through simple aqueous workup and recrystallization, thereby guaranteeing a superior impurity profile for downstream pharmaceutical synthesis.
How to Synthesize 3-Formyl Chromone Efficiently
The execution of this synthesis requires careful attention to stoichiometry and thermal management to maximize the efficiency of the Vilsmeier reagent generation and subsequent cyclization. The process is designed to be scalable, utilizing readily available industrial solvents and reagents that do not require specialized storage infrastructure beyond standard safety protocols for halogenated compounds. Operators must ensure that the addition rates of the BTC solution and the substrate are controlled to maintain the reaction temperature within the specified 0-5°C range during the initial phase, preventing localized hot spots that could lead to reagent decomposition. Detailed standard operating procedures regarding the specific molar ratios, solvent volumes, and workup techniques are essential for reproducing the high yields reported in the patent embodiments.
- Prepare the Vilsmeier reagent in situ by reacting bis(trichloromethyl) carbonate (BTC) with N,N-dimethylformamide (DMF) in a C1-C4 halogenated hydrocarbon solvent at 0-5°C.
- Add the solution of the o-hydroxyacetophenone derivative dropwise to the reaction mixture while maintaining the temperature at 0-5°C, and stir for 1-4 hours.
- Warm the reaction mixture to 25-30°C and maintain stirring for 12-16 hours to complete the cyclization, followed by hydrolysis with ice water and standard organic extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this BTC-based synthesis route offers compelling economic and logistical advantages that extend far beyond the laboratory bench. The most significant value driver is the drastic reduction in environmental compliance costs associated with waste disposal. By completely eliminating phosphorus from the process effluent, manufacturers avoid the hefty fees and complex permitting processes required for treating phosphorus-contaminated wastewater, which represents a substantial hidden cost in traditional chromone production. Additionally, the reduction in DMF consumption, as highlighted in the patent data, lowers the raw material spend and reduces the energy load on solvent recovery distillation columns, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The transition to a phosphorus-free process inherently lowers the total cost of ownership by removing the need for expensive corrosion-resistant reactors often mandated for handling hot POCl3. The solid nature of BTC allows for safer and cheaper transportation and storage compared to liquid acid chlorides, reducing logistics overheads. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall mass balance and ensuring that a higher percentage of input costs are converted into saleable high-purity product. The simplified workup procedure, which relies on standard extraction and crystallization rather than complex chromatography, further drives down labor and consumable costs per kilogram of output.
- Enhanced Supply Chain Reliability: Utilizing BTC as a phosgene equivalent enhances supply chain resilience because it is a stable solid that is less susceptible to degradation during long-term storage compared to liquid reagents. This stability ensures that raw material quality remains consistent over time, reducing the risk of batch failures due to reagent hydrolysis or contamination. The broad substrate scope of the method means that a single supply chain configuration can support the production of multiple chromone derivatives, allowing for greater flexibility in responding to market demand fluctuations without the need for retooling or sourcing niche reagents for specific analogues. This standardization simplifies inventory management and reduces the risk of stockouts for critical intermediates.
- Scalability and Environmental Compliance: The mild exothermic profile of the BTC-DMF reaction makes it exceptionally well-suited for large-scale commercial production, as heat removal can be managed with standard jacketed reactors without the need for extreme cooling capacities. This thermal safety margin facilitates a smoother technology transfer from pilot plant to multi-ton manufacturing suites. From a regulatory standpoint, the "cleaner" nature of the process aligns perfectly with the increasing pressure from global regulatory bodies and end-users for greener manufacturing practices. Companies adopting this technology can leverage their reduced environmental footprint as a competitive differentiator when bidding for contracts with sustainability-focused multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of how this technology resolves historical bottlenecks in chromone synthesis. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the BTC method improve upon traditional POCl3 Vilsmeier reactions?
A: The BTC method eliminates the use of phosphorus oxychloride (POCl3), thereby removing significant phosphorus pollution sources and simplifying wastewater treatment protocols while maintaining high yields across diverse substrates.
Q: What is the substrate scope for this 3-formyl chromone synthesis?
A: The method demonstrates excellent tolerance for various substituents on the aromatic ring, including electron-donating groups like methoxy and hydroxyl, as well as electron-withdrawing groups like nitro and halogens, ensuring consistent yields where traditional methods often fail.
Q: What are the optimal molar ratios for the reagents in this process?
A: The preferred molar ratio of o-hydroxyacetophenone derivative to bis(trichloromethyl) carbonate to DMF is approximately 1:2~2.3:8~12, which balances reagent cost with reaction efficiency and minimizes excess DMF usage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Formyl Chromone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, environmentally sustainable synthetic routes is a strategic imperative for the modern pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in patent literature are faithfully translated into robust industrial reality. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-formyl chromone derivative meets the exacting standards required for API synthesis. Our commitment to process excellence means we can deliver these complex intermediates with the consistency and reliability necessary to support your clinical and commercial programs.
We invite you to engage with our technical procurement team to discuss how this BTC-mediated technology can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering high-quality fine chemical intermediates.
