Advanced Manufacturing of 2,6-Dichloro-4,8-Dipiperidinopyrimido[5,4-D]Pyrimidine for Global Pharmaceutical Supply Chains
Advanced Manufacturing of 2,6-Dichloro-4,8-Dipiperidinopyrimido[5,4-D]Pyrimidine for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously demands higher purity intermediates to ensure the safety and efficacy of final active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese patent CN108383844B, which details an innovative synthesis method for 2,6-dichloro-4,8-dipiperidinopyrimido[5,4-D]pyrimidine, a critical precursor for the coronary vasodilator Dipyridamole. Traditional manufacturing pathways have long struggled with impurity profiles that complicate downstream purification and limit yield. This new technical disclosure introduces a sophisticated strategy combining selective hydroxyl protection with microwave-assisted chlorination. By fundamentally altering the reaction sequence—starting from a tetrahydroxy precursor rather than a tetrachloro one—the process achieves exceptional selectivity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this methodology represents a paradigm shift towards greener, more efficient, and higher-yielding production capabilities that align with modern Good Manufacturing Practice (GMP) standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key dipyridamole intermediate relied on the direct nucleophilic substitution of 2,4,6,8-tetrachloropyrimido[5,4-D]pyrimidine with piperidine. While conceptually straightforward, this conventional route suffers from inherent chemical flaws that plague large-scale operations. The primary issue lies in the poor selectivity of piperidine towards the four chlorine atoms on the pyrimido-pyrimidine ring. Without precise control, piperidine indiscriminately attacks multiple positions, leading to a complex mixture of mono-, di-, tri-, and tetra-substituted byproducts. Separating the desired 4,8-disubstituted product from these structurally similar impurities is notoriously difficult and costly, often requiring extensive chromatography or multiple recrystallization steps that erode overall yield. Furthermore, the harsh conditions often required to drive these substitutions can degrade the sensitive heterocyclic core. As illustrated in the traditional pathway below, the lack of a directing group results in a chaotic reaction landscape that is ill-suited for the rigorous purity specifications demanded by modern regulatory bodies.
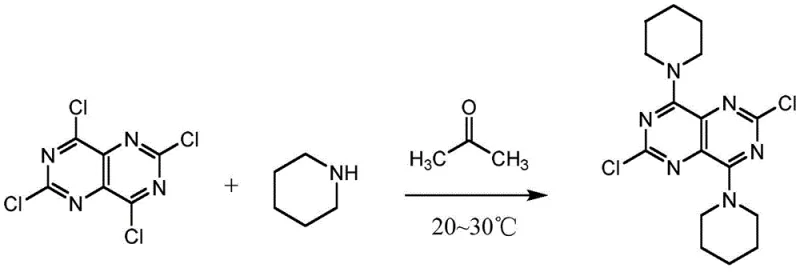
The Novel Approach
In stark contrast, the methodology disclosed in patent CN108383844B employs a "protection-first" strategy that elegantly solves the selectivity dilemma. Instead of starting with a fully chlorinated scaffold, the synthesis begins with 2,4,6,8-tetrahydroxypyrimido[5,4-d]pyrimidine. The process introduces 4-toluenesulfonyl chloride (TsCl) to selectively protect specific hydroxyl groups based on steric hindrance effects. This strategic masking ensures that subsequent nucleophilic attack by piperidine occurs exclusively at the unhindered 4 and 8 positions. Following this controlled substitution, the remaining protected groups are converted to chlorines using thionyl chloride under microwave irradiation. This inversion of the synthetic logic—from "chlorinate then substitute" to "protect, substitute, then chlorinate"—drastically simplifies the impurity profile. The result is a streamlined process that delivers the target molecule with yields exceeding 92% and purity greater than 99%, effectively eliminating the purification bottlenecks associated with legacy methods.
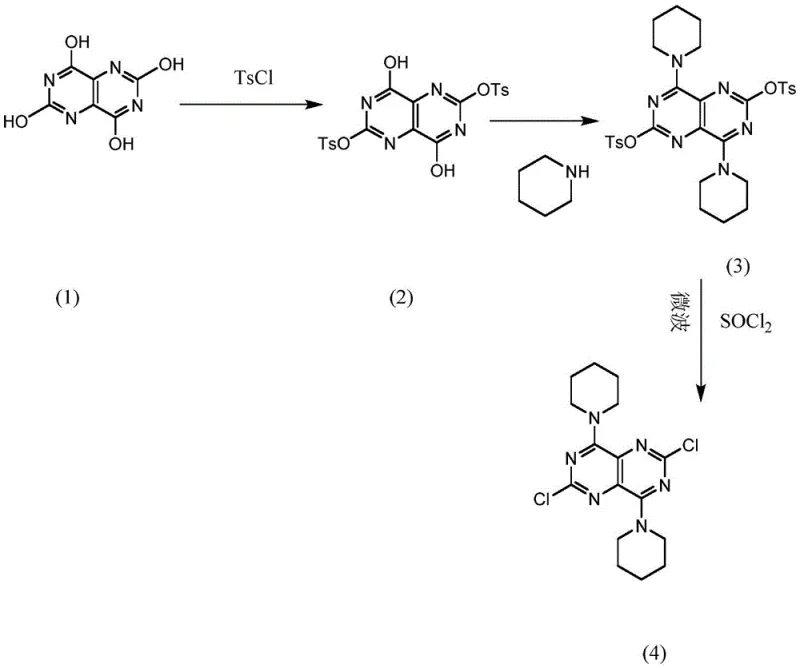
Mechanistic Insights into Selective Tosylation and Microwave Activation
The core innovation of this synthesis lies in the exploitation of steric hindrance during the tosylation step. When 2,4,6,8-tetrahydroxypyrimido[5,4-d]pyrimidine reacts with 4-toluenesulfonyl chloride, the bulky tosyl groups preferentially attach to the hydroxyls at the 2 and 6 positions due to the specific electronic and spatial environment of the fused ring system. This leaves the hydroxyl groups at the 4 and 8 positions free and accessible for nucleophilic attack. When piperidine is introduced, it acts as a nucleophile, displacing the unprotected hydroxyls (likely via an activated intermediate or direct substitution mechanism facilitated by the reaction conditions) to form the C-N bonds. This mechanistic precision is crucial; it prevents the formation of the unwanted 2,4,6,8-tetrapiperidino byproduct that plagues the direct chlorination route. The use of excess piperidine further drives this equilibrium to completion while simultaneously acting as an acid scavenger, neutralizing any acidic byproducts generated during the substitution, thus maintaining the integrity of the acid-sensitive heterocyclic core throughout the transformation.
Following the substitution, the conversion of the remaining tosyl-protected hydroxyls into chlorines is achieved through a rapid microwave-assisted reaction with thionyl chloride (SOCl2). Microwave chemistry offers distinct advantages over conventional conductive heating in this context. The dielectric heating mechanism allows for superheating of the polar reaction mixture, providing the activation energy required for the chlorination almost instantaneously. This reduces the reaction time from potentially several hours to merely 5-8 minutes at 400-500W power. Such rapid processing minimizes the thermal exposure of the product, thereby reducing thermal degradation and the formation of heat-induced impurities. The addition of triethylamine serves as a base to capture the HCl and SO2 gases evolved during the chlorination, driving the reaction forward and preventing acid-catalyzed decomposition of the newly formed piperidine linkages. This combination of selective protection and kinetic acceleration via microwaves creates a robust chemical environment conducive to high-purity manufacturing.
How to Synthesize 2,6-Dichloro-4,8-Dipiperidinopyrimido[5,4-D]Pyrimidine Efficiently
Implementing this advanced synthesis route requires careful attention to stoichiometry and thermal management, particularly during the microwave phase. The process is designed to be operationally simple, utilizing commercially available reagents such as ethanol, piperidine, and thionyl chloride. The initial protection and substitution steps are conducted under mild cooling (ice-water bath) to control exotherms and ensure regioselectivity. The subsequent microwave step requires specialized equipment capable of delivering consistent power output to ensure batch-to-batch reproducibility. For process chemists looking to transfer this technology from the lab to the pilot plant, the following standardized protocol outlines the critical operational parameters derived from the patent examples. These steps highlight the ease of workup, where the product precipitates upon quenching, simplifying isolation and reducing solvent consumption compared to extraction-heavy traditional methods.
- React 2,4,6,8-tetrahydroxypyrimido[5,4-d]pyrimidine with 4-toluenesulfonyl chloride in ethanol under ice-water bath conditions, followed by nucleophilic substitution with excess piperidine to form the protected intermediate.
- Subject the intermediate to chlorination using thionyl chloride and triethylamine in a microwave reactor at 400-500W for 5-8 minutes to replace hydroxyl/tosyl groups with chlorine.
- Quench the reaction mixture into cold water, filter the residue, wash with sodium chloride solution, and recrystallize from ethanol to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift away from complex purification sequences directly translates to reduced manufacturing costs and shorter cycle times. By eliminating the need for extensive chromatographic separation or multiple recrystallizations to remove isomeric byproducts, the overall processing time is significantly compressed. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations. Furthermore, the ability to recover and reuse excess piperidine distills out a major variable cost driver, enhancing the economic viability of the process. The reliance on standard, bulk-available chemicals like tosyl chloride and thionyl chloride ensures that raw material sourcing remains stable and不受 geopolitical supply shocks that might affect exotic catalysts. This stability is paramount for maintaining continuity in the supply of critical cardiovascular medication intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the downstream processing workflow. In traditional methods, a significant portion of the production budget is allocated to purification efforts aimed at removing closely related impurities. By engineering selectivity into the reaction itself through tosyl protection, the need for these expensive separation steps is virtually eliminated. Additionally, the dual role of piperidine as both reactant and solvent reduces the volume of auxiliary solvents required, lowering both procurement costs and waste disposal fees. The high yield (>92%) means less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS) and allowing for more competitive pricing structures in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of inputs. The process operates at relatively low temperatures (0-30°C) for the critical substitution steps, reducing the risk of thermal runaway incidents that can halt production lines. The microwave chlorination step is rapid, increasing the throughput capacity of existing reactor infrastructure without the need for massive capital expenditure on new vessels. Moreover, since the starting material (2,4,6,8-tetrahydroxypyrimido[5,4-d]pyrimidine) and reagents are commodity chemicals, the risk of supply disruption is minimized. This reliability ensures that downstream API manufacturers can maintain consistent production schedules for Dipyridamole, safeguarding patient access to essential heart medications.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns well with green chemistry principles. The reduction in solvent usage and the ability to recycle piperidine significantly lower the E-factor (mass of waste per mass of product). The microwave step, while requiring specific equipment, is highly amenable to continuous flow scale-up, a technology that is increasingly favored for handling hazardous reagents like thionyl chloride safely. The simplified workup, involving filtration and washing rather than complex extractions, reduces the generation of aqueous waste streams. These factors make the process easier to permit and operate within strict environmental regulations, facilitating smoother scale-up from pilot batches to multi-ton commercial production runs without encountering regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages outlined in the patent documentation and are intended to clarify the operational feasibility for potential partners. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement officers assessing long-term supply contracts. The answers reflect the consensus on how selective protection strategies and process intensification techniques contribute to modern pharmaceutical manufacturing excellence.
Q: How does this new synthesis method improve product purity compared to traditional routes?
A: The novel method utilizes a selective tosylation strategy that protects specific hydroxyl groups via steric hindrance. This forces the piperidine substitution to occur only at the desired 4,8-positions, significantly reducing the formation of multi-substituted byproducts common in direct chlorination routes, thereby achieving purity levels exceeding 99%.
Q: What are the scalability advantages of using microwave technology in this process?
A: Microwave irradiation provides rapid, uniform heating which drastically reduces reaction times from hours to minutes (5-8 min). This intensification allows for smaller reactor footprints and easier transition to continuous flow processing, enhancing throughput and energy efficiency for large-scale commercial production.
Q: Is the excess piperidine used in the reaction recoverable?
A: Yes, the process design accounts for sustainability and cost efficiency. The excessive piperidine serves dual roles as both a reactant and a solvent/base, and it can be distilled off under reduced pressure after the reaction for repeated reuse, minimizing raw material waste and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dichloro-4,8-Dipiperidinopyrimido[5,4-D]Pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a robust commercial process requires deep technical expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in patent CN108383844B are maintained at an industrial level. Our facilities are equipped with advanced microwave reactors and stringent purity specifications are enforced through our rigorous QC labs, which utilize HPLC and NMR to verify that every batch meets the >99% purity threshold required for cardiovascular API synthesis. We understand the critical nature of this intermediate in the global supply chain for Dipyridamole and are committed to delivering consistent quality.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize this synthesis route for your specific needs. Whether you require custom process development to further reduce costs or immediate supply of GMP-grade intermediates, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this microwave-assisted technology can enhance your supply chain efficiency and product quality.
