Advanced Synthesis of Benzisoselenazolone Amino Sugar Derivatives for Oncology Applications
Advanced Synthesis of Benzisoselenazolone Amino Sugar Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that can offer improved therapeutic indices, particularly in the challenging field of oncology. Patent CN101016319A introduces a significant advancement in this domain by disclosing a series of benzisoselenazolone amino sugar derivatives. These compounds represent a strategic fusion of organoselenium chemistry with carbohydrate biology, aiming to leverage the known antioxidant and anti-tumor properties of selenium while utilizing the targeting potential of amino sugars. As a leading manufacturer in the fine chemical sector, we recognize the immense potential of this chemical class to serve as critical intermediates for next-generation anti-cancer therapeutics. The structural innovation lies in the modification of the benzisoselenazolone core, traditionally associated with drugs like Ebselen, by attaching hydroxyl-protected amino sugar units. This modification is not merely a structural exercise but a calculated move to address the historical limitations of organoselenium drugs, specifically regarding toxicity and bioavailability. By integrating these sugar moieties, the resulting derivatives exhibit remarkable anti-tumor efficacy in vitro, suggesting a promising pathway for developing safer and more effective cancer treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organoselenium drugs has been hindered by a narrow therapeutic window. While compounds like Ebselen (2-phenyl-1,2-benzisoselenazol-3(2H)-one) have demonstrated potent anti-inflammatory and antioxidant activities, their clinical progression has often been stalled by concerns over toxicity at higher doses. Conventional synthesis methods for simple benzisoselenazolones often yield products that lack specific targeting mechanisms, leading to non-specific distribution in the body and potential adverse effects on healthy tissues. Furthermore, the chemical stability of some early-generation selenium compounds can be problematic, requiring complex formulation strategies to maintain efficacy during storage and administration. The reliance on simple aromatic substituents limits the chemical diversity available for structure-activity relationship (SAR) studies, making it difficult to optimize the pharmacokinetic profile. Consequently, there has been a pressing need in the R&D community for a modular synthetic approach that allows for the systematic variation of substituents to enhance potency while mitigating toxicological risks.
The Novel Approach
The methodology outlined in the patent data presents a transformative solution by introducing amino sugar derivatives as key structural components. This novel approach utilizes the inherent biological recognition of sugars by cell surface receptors to potentially improve the cellular uptake of the selenium payload specifically in tumor environments. The synthesis strategy is elegantly designed to be flexible, allowing for the incorporation of various protecting groups such as acetyl, benzoyl, or benzyl groups on the sugar moiety. This flexibility enables chemists to fine-tune the lipophilicity and solubility of the final molecule, which are critical parameters for drug absorption and distribution. Moreover, the synthetic route avoids the use of exotic or prohibitively expensive catalysts, relying instead on robust organic transformations like amide coupling and nucleophilic substitution. This practical aspect of the chemistry ensures that the transition from laboratory discovery to commercial-scale manufacturing is seamless, addressing a major pain point for procurement teams who often struggle with the scalability of complex medicinal chemistry routes.
Mechanistic Insights into Benzisoselenazolone Ring Formation
The core of this technology lies in the efficient construction of the benzisoselenazolone ring system onto the amino sugar scaffold. The mechanism typically proceeds through the reaction of a hydroxyl-protected 2-amino sugar with 2-chloroselenobenzoyl chloride. This acyl chloride acts as a potent electrophile, reacting with the nucleophilic amine group on the sugar to form an amide intermediate. In the presence of a base such as triethylamine, an intramolecular cyclization occurs where the selenium atom attacks the carbonyl carbon, closing the ring to form the stable benzisoselenazolone structure. This cyclization is highly favorable due to the formation of the five-membered heterocyclic ring containing the selenium-nitrogen bond. The patent describes two distinct pathways: one involving a direct reaction and another involving an intermediate amino acid coupling. The latter pathway allows for the introduction of a spacer arm between the sugar and the selenium core, providing additional conformational freedom that may enhance binding affinity to biological targets. Understanding this mechanistic nuance is vital for R&D directors aiming to replicate or modify the synthesis for specific analogues.
Control of impurities is paramount in the synthesis of such complex heterocycles, particularly given the sensitivity of selenium chemistry. The process described utilizes mild reaction conditions, typically maintaining temperatures between 0°C and room temperature during the critical coupling steps to prevent degradation of the sensitive sugar protecting groups. The use of palladium on carbon (Pd/C) for hydrogenation in the alternative pathway effectively removes benzyloxycarbonyl protecting groups without affecting the selenium moiety, showcasing the chemoselectivity of the method. Furthermore, the purification protocols involve standard techniques like silica gel column chromatography and recrystallization, which are well-understood and easily validated in a GMP environment. This ensures that the final product meets stringent purity specifications required for pharmaceutical intermediates. The structural diversity is further expanded by the ability to vary the R groups on the sugar ring, as illustrated in the general formula below, allowing for a broad library of compounds to be generated for screening purposes.
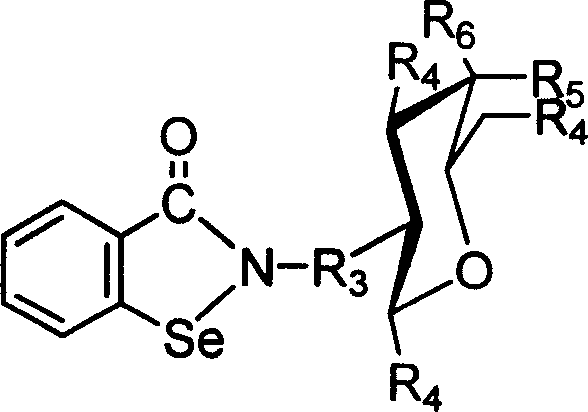
How to Synthesize Benzisoselenazolone Amino Sugar Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure high yields and purity. The process begins with the preparation of the key selenium building block, 2-chloroselenobenzoyl chloride, which is synthesized from anthranilic acid via a diazonium salt intermediate and subsequent reaction with sodium diselenide. This precursor is then coupled with protected amino sugars, such as 1,3,4,6-tetra-O-benzoyl-2-deoxy-β-D-glucopyranosamine, under basic conditions. The detailed standardized synthesis steps, including specific molar ratios, solvent choices like chloroform or dichloromethane, and workup procedures, are critical for reproducibility. For a comprehensive guide on executing this synthesis with optimal efficiency and safety, please refer to the technical protocol provided below.
- Preparation of 2-chloroselenobenzoyl chloride via diazotization of anthranilic acid followed by reaction with sodium diselenide and thionyl chloride.
- Protection of 2-amino sugars (e.g., glucosamine) using benzoyl or acetyl groups to ensure regioselectivity during subsequent coupling.
- Cyclization reaction between the protected amino sugar or sugar-amide intermediate and 2-chloroselenobenzoyl chloride in the presence of triethylamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the manufacturing process for benzisoselenazolone amino sugar derivatives offers significant advantages over more complex biologic or semi-synthetic routes. The reliance on commodity chemicals means that the raw material supply is secure and less susceptible to geopolitical disruptions or seasonal fluctuations. For procurement managers, this translates into a more predictable cost structure and the ability to negotiate long-term contracts with confidence. The synthetic route is linear and convergent, minimizing the number of unit operations required to reach the final intermediate. This simplicity directly correlates to reduced manufacturing costs, as fewer processing steps mean lower labor, energy, and equipment utilization expenses. Additionally, the avoidance of transition metal catalysts in the final cyclization step eliminates the need for costly and time-consuming heavy metal scavenging processes, further streamlining the production workflow and reducing waste disposal costs.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes widely available starting materials such as D-glucosamine hydrochloride and anthranilic acid, which are produced on a multi-ton scale globally, ensuring a stable and low-cost supply base. The reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, allowing for production in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. By eliminating the need for expensive chiral catalysts or enzymatic resolutions, the overall cost of goods sold (COGS) is significantly optimized. The high yields reported in the patent examples suggest that material throughput is efficient, minimizing the loss of valuable intermediates and maximizing the return on raw material investment.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures that the manufacturing process is resilient to minor variations in operating conditions, which is crucial for maintaining consistent supply continuity. Since the synthesis does not depend on single-source proprietary reagents, there is flexibility to qualify multiple vendors for key inputs, thereby mitigating supply risk. The scalability of the process from gram to kilogram and eventually to ton scale is straightforward, as the exothermic profiles of the reactions are manageable and the workup procedures involve standard phase separations and crystallizations. This scalability assures supply chain heads that the production capacity can be rapidly ramped up to meet increasing demand from downstream pharmaceutical partners without compromising quality.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is relatively low compared to other organometallic processes, as the primary byproducts are inorganic salts and benign organic solvents that can be recovered and recycled. The absence of persistent heavy metal contaminants simplifies the wastewater treatment process, ensuring compliance with increasingly stringent environmental regulations. The solid waste generated, primarily consisting of silica from chromatography or filter cakes, is non-hazardous and can be disposed of using standard industrial waste management protocols. This alignment with green chemistry principles not only reduces regulatory burden but also enhances the sustainability profile of the supply chain, a key metric for modern corporate responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized selenium intermediates. These insights are derived directly from the technical disclosures in the patent literature and our internal expertise in process chemistry. Understanding these aspects is crucial for stakeholders evaluating the feasibility of incorporating these derivatives into their drug development pipelines. We encourage potential partners to review these details to gain a clearer picture of the operational and scientific landscape surrounding this technology.
Q: What is the primary advantage of benzisoselenazolone amino sugar derivatives over traditional Ebselen?
A: Unlike traditional Ebselen which faces challenges regarding toxicity, these derivatives utilize amino sugar moieties to potentially enhance bioavailability and target tumor cells more specifically while reducing systemic toxicity.
Q: Are the starting materials for this synthesis commercially scalable?
A: Yes, the synthesis relies on readily available bulk chemicals such as anthranilic acid, selenium powder, and D-glucosamine hydrochloride, ensuring a robust and scalable supply chain for commercial production.
Q: What are the key purification steps in this manufacturing process?
A: The process involves standard purification techniques including recrystallization from solvents like ether or ethanol, and silica gel column chromatography to ensure high purity of the final organoselenium intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzisoselenazolone Amino Sugar Derivative Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of novel oncology therapeutics depends on a reliable and high-quality supply of advanced intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage clinical trials through to full-scale market launch. We operate with stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzisoselenazolone derivatives meets the highest industry standards. Our commitment to quality assurance means that you can trust our materials to perform consistently in your biological assays and formulation studies, reducing the risk of delays in your development timeline.
We invite you to collaborate with us to unlock the full potential of this promising chemical class. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us today to request specific COA data and route feasibility assessments for your project. By partnering with us, you gain access to a supply chain partner dedicated to driving innovation and efficiency in the pharmaceutical industry, ensuring that your breakthrough therapies reach patients faster and more economically.
