Advanced L-Proline Amide Derivatives for Efficient Asymmetric Aldol Synthesis
The landscape of asymmetric synthesis is undergoing a significant transformation, driven by the urgent need for environmentally benign and operationally simple methodologies in the production of chiral pharmaceutical intermediates. Patent CN101891668A introduces a groundbreaking class of L-proline amide derivatives, specifically designed to overcome the limitations of traditional catalytic systems. These novel organocatalysts, characterized by the general Formula I shown below, represent a major leap forward in metal-free catalysis, offering a robust solution for constructing carbon-carbon bonds with exceptional stereocontrol. 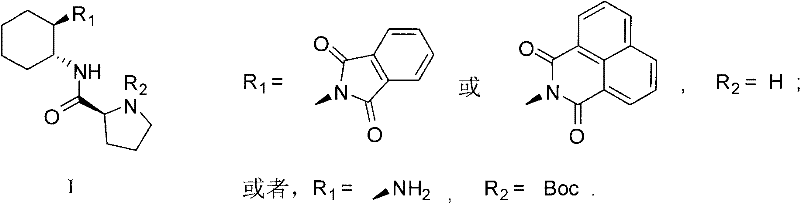 By integrating a rigid cyclohexane diamine backbone with the proven proline motif, these derivatives achieve a delicate balance of steric hindrance and electronic activation that is crucial for high-performance asymmetric induction. This technological advancement addresses the critical pain points of modern drug discovery, where purity profiles and regulatory compliance regarding heavy metal residues are becoming increasingly stringent.
By integrating a rigid cyclohexane diamine backbone with the proven proline motif, these derivatives achieve a delicate balance of steric hindrance and electronic activation that is crucial for high-performance asymmetric induction. This technological advancement addresses the critical pain points of modern drug discovery, where purity profiles and regulatory compliance regarding heavy metal residues are becoming increasingly stringent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral beta-hydroxy ketones via the Aldol reaction has relied heavily on stoichiometric chiral auxiliaries or transition metal complexes. While effective, these conventional methods suffer from inherent drawbacks that hinder their scalability and economic viability in large-scale manufacturing. Metal-catalyzed processes often necessitate rigorous exclusion of moisture and oxygen, requiring specialized equipment and inert atmosphere techniques that drastically increase operational costs. Furthermore, the removal of trace metal contaminants from the final active pharmaceutical ingredient (API) is a costly and technically challenging purification step, often requiring additional chromatography or complexation agents. The environmental burden of disposing of heavy metal waste streams also poses a significant liability for manufacturers aiming to adhere to green chemistry principles, making these traditional routes less attractive for sustainable industrial applications.
The Novel Approach
The innovative strategy presented in this patent utilizes L-proline amide derivatives to catalyze intermolecular asymmetric direct Aldol reactions with remarkable efficiency. Unlike previous organocatalytic attempts that required high catalyst loadings to achieve acceptable conversion rates, these specific derivatives, particularly compounds b and c, demonstrate superior activity at significantly lower concentrations. The structural rigidity imparted by the cyclohexane ring system enhances the facial selectivity of the enamine intermediate, leading to outstanding diastereomeric ratios (dr) and enantiomeric excess (ee) values. This approach eliminates the need for toxic metals entirely, allowing reactions to proceed in common organic solvents or even water under ambient atmospheric conditions. The ability to operate at mild temperatures, typically ranging from -40°C to 25°C, further underscores the practicality of this method for sensitive substrates that might decompose under harsher conditions.
Mechanistic Insights into Organocatalytic Asymmetric Aldol Reaction
The efficacy of these L-proline amide derivatives stems from their ability to form a well-defined chiral enamine intermediate with the ketone donor. The secondary amine of the proline moiety condenses with the ketone, while the adjacent amide group participates in a hydrogen-bonding network that organizes the transition state. This dual activation mode ensures that the incoming aldehyde electrophile approaches the enamine from a specific trajectory, dictated by the bulky substituents on the cyclohexane scaffold.  Experimental data indicates that the presence of a Brønsted acid cocatalyst, such as 3-methylbenzoic acid, further accelerates the reaction by protonating the carbonyl oxygen of the aldehyde, thereby increasing its electrophilicity. This synergistic effect between the chiral amine and the acidic additive is critical for achieving the high turnover frequencies observed in the patent examples, allowing for rapid bond formation without compromising stereochemical integrity.
Experimental data indicates that the presence of a Brønsted acid cocatalyst, such as 3-methylbenzoic acid, further accelerates the reaction by protonating the carbonyl oxygen of the aldehyde, thereby increasing its electrophilicity. This synergistic effect between the chiral amine and the acidic additive is critical for achieving the high turnover frequencies observed in the patent examples, allowing for rapid bond formation without compromising stereochemical integrity.
Impurity control is inherently superior in this metal-free system due to the absence of metal-ligand complexes that can lead to side reactions or racemization. The reaction pathway is highly selective, minimizing the formation of syn-diastereomers and favoring the anti-product with ratios often exceeding 95:5. The robustness of the catalyst structure prevents degradation under the reaction conditions, ensuring that the impurity profile of the crude product remains clean and manageable. This predictability is invaluable for process chemists who must validate purification protocols early in the development timeline, as it reduces the risk of unexpected byproducts that could complicate downstream processing or fail regulatory specifications for genotoxic impurities.
How to Synthesize Chiral Aldol Products Efficiently
To leverage the full potential of this technology, manufacturers should adopt the optimized protocol detailed in the patent, which balances reaction time, temperature, and catalyst loading for maximum throughput. The procedure involves a straightforward mixing of reagents in acetonitrile, followed by a controlled addition of the aldehyde to manage exotherms and maintain selectivity. Detailed standardized synthesis steps for implementing this catalytic cycle in a GMP environment are provided in the guide below, ensuring reproducibility and safety.
- Prepare the reaction mixture by dissolving the ketone substrate, L-proline amide derivative catalyst (5 mol%), and 3-methylbenzoic acid cocatalyst in acetonitrile at -20°C.
- Add the aldehyde substrate to the stirred mixture and maintain the temperature at -20°C for approximately 30 to 40 hours to ensure complete conversion.
- Quench the reaction with saturated ammonium chloride, extract with dichloromethane, and purify the crude product via flash column chromatography to isolate the high-purity Aldol adduct.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-proline amide derivative technology offers substantial strategic benefits beyond mere technical performance. The shift from metal-based to organocatalytic processes fundamentally alters the cost structure and risk profile of manufacturing chiral intermediates. By removing the dependency on precious metals like palladium or rhodium, companies can insulate themselves from the volatile pricing fluctuations of the commodities market. Furthermore, the simplified workup procedures reduce the consumption of expensive scavenging resins and specialized filtration media, leading to a leaner and more cost-effective production workflow that enhances overall margin potential.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts removes the necessity for expensive and time-consuming metal scavenging steps, which often involve costly silica-bound thiol resins or complex aqueous extractions. Additionally, the ability to recover and reuse the organocatalyst multiple times without significant loss of activity dramatically lowers the effective cost per kilogram of the final product. This recyclability feature transforms the catalyst from a consumable expense into a reusable asset, driving down the variable costs associated with raw materials and waste disposal significantly.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can be subject to geopolitical instability and supply bottlenecks, whereas the precursors for these L-proline derivatives are abundant and commercially available fine chemicals. The robust nature of the catalyst allows for storage and transport under standard conditions without the need for specialized cold chain logistics or inert gas blanketing. This resilience ensures a continuous supply of critical reagents, minimizing the risk of production stoppages due to material shortages and enabling more accurate forecasting for long-term manufacturing campaigns.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent's successful transition from milligram-scale screening to multi-gram synthesis without loss of selectivity. The use of benign solvents like acetonitrile and the absence of toxic metal waste simplify the environmental permitting process and reduce the burden on wastewater treatment facilities. This alignment with green chemistry principles not only lowers regulatory compliance costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major pharmaceutical partners who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this catalytic technology in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable foundation for process evaluation.
Q: What are the advantages of using L-proline amide derivatives over traditional metal catalysts?
A: Unlike traditional metal catalysts, these organic small molecule catalysts eliminate the risk of toxic metal contamination in the final product, which is critical for pharmaceutical applications. They operate under mild conditions without requiring strict anhydrous or oxygen-free environments, significantly simplifying operational complexity and reducing safety hazards associated with heavy metal handling.
Q: Can the L-proline amide catalyst be recovered and reused?
A: Yes, the patent data demonstrates that the catalyst can be effectively recovered from the aqueous phase after acid-base extraction. It maintains high catalytic activity and stereoselectivity over multiple cycles, with experimental results showing consistent performance through at least five reuse cycles with only negligible decreases in yield or enantiomeric excess.
Q: What is the substrate scope for this asymmetric Aldol reaction?
A: The catalyst exhibits broad substrate universality, successfully catalyzing reactions between cyclohexanone and various aromatic aldehydes, including those with electron-withdrawing or electron-donating groups. It also accommodates heteroaromatic aldehydes and different ketone substrates like cyclopentanone, delivering high diastereoselectivity and enantioselectivity across diverse chemical structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Proline Amide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in the synthesis of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our L-proline amide derivatives meets the exacting standards required for GMP manufacturing, providing our partners with the confidence needed to integrate these materials into their critical drug synthesis routes.
We invite you to collaborate with us to optimize your synthetic routes and achieve substantial cost savings through the adoption of this cutting-edge technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our high-purity organocatalysts can enhance your operational efficiency and product quality.
