Advanced Gas-Phase Catalytic Chlorination for Commercial Scale-Up of 2-Chloro-5-Trifluoromethylpyridine
The global demand for fluorinated heterocyclic compounds continues to surge, driven by their critical role in modern agrochemicals and pharmaceuticals. A pivotal advancement in this sector is detailed in Chinese Patent CN110003096B, which discloses a highly efficient method for preparing 2-chloro-5-trifluoromethylpyridine. This compound serves as an indispensable building block for synthesizing high-value active ingredients such as fluazinam, acetamiprid, and quizalofop-p-ethyl. The patent introduces a novel gas-phase catalytic chlorination strategy that fundamentally shifts the manufacturing paradigm from energy-intensive thermal processes to a more sustainable, catalyst-driven approach. By utilizing supported palladium or specific alkaline earth metal catalysts, the technology achieves superior conversion rates and selectivity while operating at significantly lower temperatures. For R&D directors and procurement strategists, this represents a tangible opportunity to optimize supply chains for high-purity agrochemical intermediates, ensuring both cost efficiency and environmental compliance in an increasingly regulated market.
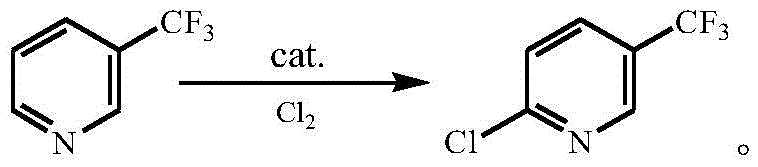
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-5-trifluoromethylpyridine has been plagued by severe operational inefficiencies and safety concerns inherent to older chlorination technologies. As documented in prior art such as European Patent EP0013474, traditional thermal chlorination methods necessitate reaction temperatures exceeding 380°C to achieve acceptable conversion. Such extreme thermal conditions not only result in exorbitant energy consumption but also promote the formation of coke and tar, which foul reactor internals and necessitate frequent, costly shutdowns for maintenance. Alternatively, photochlorination routes require specialized reactor equipment equipped with high-intensity light sources and often rely on radical initiators and organic diluents to manage reaction kinetics. These auxiliary chemicals complicate the downstream purification process, requiring extensive distillation columns to separate the product from the diluent, thereby inflating both capital expenditure (CapEx) and operating expenditure (OpEx). Furthermore, other catalytic methods reported in patents like CN104610137 utilize ferric chloride on activated carbon but still operate at excessively high temperatures around 420°C and frequently employ carbon tetrachloride as a solvent, introducing significant toxicity and waste disposal challenges that are untenable for modern green manufacturing standards.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110003096B offers a transformative solution by leveraging advanced heterogeneous catalysis under milder conditions. The core innovation lies in the use of supported palladium catalysts or specific magnesium, calcium, and barium salts which activate chlorine gas effectively at temperatures ranging strictly between 220°C and 360°C. This substantial reduction in thermal load directly translates to lower energy utility costs and significantly extends the operational lifespan of the reactor materials by minimizing thermal stress and coking. Crucially, this novel approach operates in the gas phase without the need for any organic solvents or diluents. The elimination of solvents like carbon tetrachloride removes the entire unit operation associated with solvent recovery and recycling, simplifying the process flow diagram to essentially just reaction, neutralization, and distillation. This streamlining not only reduces the physical footprint of the manufacturing plant but also drastically lowers the risk profile associated with handling volatile organic compounds, aligning perfectly with the stringent EHS (Environment, Health, and Safety) protocols required by top-tier multinational agrochemical companies seeking a reliable agrochemical intermediate supplier.
Mechanistic Insights into Pd-Catalyzed Electrophilic Substitution
From a mechanistic perspective, the success of this synthesis relies on the precise activation of the chlorine molecule by the transition metal centers on the catalyst surface. In the case of supported palladium catalysts, the palladium atoms act as Lewis acid sites that polarize the Cl-Cl bond, generating a highly electrophilic chlorine species capable of attacking the electron-deficient pyridine ring. The presence of the trifluoromethyl group at the 3-position exerts a strong electron-withdrawing inductive effect, which deactivates the ring towards electrophilic aromatic substitution generally, but directs incoming electrophiles specifically to the 2-position due to steric and electronic factors governed by the catalyst surface geometry. The catalyst support, whether it be activated carbon, alumina, or aluminum fluoride, plays a non-innocent role by stabilizing the active metal species and preventing sintering at the operating temperatures of 270-320°C. This stabilization is critical for maintaining consistent activity over long run times, ensuring that the selectivity for the mono-chlorinated product remains high while suppressing the formation of di-chlorinated impurities which are difficult to separate. The interaction between the basic sites of the alkaline earth metal promoters (such as MgF2 or CaO) and the acidic HCl byproduct generated during the reaction may also serve to buffer the local acidity, further protecting the catalyst structure from degradation and enhancing the overall atom economy of the transformation.
Impurity control is another critical aspect where this catalytic system excels compared to free-radical thermal chlorination. In thermal processes, the lack of directional control often leads to a statistical distribution of chlorinated isomers, including 4-chloro and 6-chloro derivatives, as well as poly-chlorinated species that degrade the quality of the final API intermediate. The heterogeneous catalyst in this patent creates a specific microenvironment that favors the kinetic formation of the 2-chloro isomer. By optimizing the contact time between the vaporized 3-trifluoromethylpyridine and the chlorine gas within the catalyst bed to between 5 and 30 seconds, the process minimizes residence time enough to prevent over-chlorination while ensuring complete conversion of the starting material. This precise kinetic control results in a crude product stream with exceptionally high purity, reducing the burden on the final distillation columns. For quality assurance teams, this means a much cleaner impurity profile, facilitating easier compliance with rigorous pharmacopeial or agrochemical specifications and reducing the risk of batch rejection due to trace isomeric contaminants that could affect the biological efficacy of the downstream pesticide.
How to Synthesize 2-Chloro-5-Trifluoromethylpyridine Efficiently
The implementation of this synthesis route requires careful attention to reactor engineering and catalyst preparation to fully realize the benefits described in the patent literature. The process begins with the activation of the chosen catalyst, typically involving a thermal treatment under nitrogen or chlorine flow to ensure the active sites are clean and accessible. Once the reactor, preferably a fluidized bed or fixed bed tubular reactor made of corrosion-resistant alloys like Inconel, reaches the target temperature zone of 220-360°C, the vaporized feedstocks are introduced. The molar ratio of 3-trifluoromethylpyridine to chlorine is a critical parameter, optimally maintained between 1:4 and 1:10 to drive the equilibrium forward without excessive waste of chlorine gas. Detailed standardized synthetic steps, including specific flow rates, catalyst loading densities, and workup procedures involving water and alkali washing followed by steam distillation, are essential for reproducibility.
- Preparation of Catalyst Bed: Load a fixed or fluidized bed reactor with supported palladium (0.1-10wt% on carbon/alumina) or magnesium/calcium salt catalysts.
- Gas-Phase Reaction: Vaporize 3-trifluoromethylpyridine and mix with chlorine gas at a molar ratio of 1:4 to 1:10, passing through the catalyst bed at 220-360°C.
- Workup and Purification: Condense the effluent, neutralize with alkali, separate the organic layer, and distill to obtain high-purity 2-chloro-5-trifluoromethylpyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers profound strategic advantages that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic simplification of the manufacturing process, which directly correlates to reduced operational complexity and lower fixed costs. By eliminating the requirement for organic solvents and diluents, manufacturers can avoid the substantial costs associated with purchasing, storing, and recovering large volumes of volatile chemicals. This solvent-free approach also mitigates the regulatory burdens and environmental taxes often levied on facilities handling hazardous air pollutants, thereby improving the overall margin profile of the product. Furthermore, the lower operating temperatures significantly reduce the energy intensity of the process, leading to sustained utility cost savings over the lifecycle of the plant. These efficiencies collectively contribute to a more competitive pricing structure, allowing suppliers to offer cost reduction in fluorinated pyridine manufacturing without compromising on quality or reliability, a key differentiator in the volatile global chemical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of solvent recovery units and the reduction in energy consumption. Traditional methods requiring carbon tetrachloride necessitate complex distillation trains to separate the solvent from the product, which consumes significant steam and cooling water; removing this step simplifies the plant design and lowers variable production costs. Additionally, the use of robust heterogeneous catalysts that maintain activity over extended periods reduces the frequency of catalyst replacement and the associated downtime, further enhancing asset utilization. The high selectivity of the reaction minimizes the generation of heavy ends and tars, reducing waste disposal fees and maximizing the yield of saleable product per unit of raw material input. These factors combine to create a leaner, more cost-effective production model that is resilient against fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for downstream formulators of herbicides and insecticides, and this technology supports that need through its operational robustness. The ability to run the reaction in a continuous gas-phase mode allows for steady-state production that is less prone to the batch-to-batch variability seen in older liquid-phase methods. The feedstock, 3-trifluoromethylpyridine, is a commercially available commodity, and the reagent chlorine is ubiquitous, ensuring that raw material sourcing is not a bottleneck. Moreover, the simplified downstream processing means that turnaround times between batches or campaigns are minimized, enabling manufacturers to respond more agilely to spikes in market demand. This reliability makes the producer a preferred partner for long-term supply agreements, reducing the risk of production stoppages that could disrupt the global supply of critical crop protection agents.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial tonnage is straightforward due to the well-understood engineering principles of fixed and fluidized bed gas-phase reactors. The absence of solvent hazards simplifies the safety case for large-scale operations, facilitating faster regulatory approvals for capacity expansions. From an environmental standpoint, the process generates significantly less hazardous waste, as there are no spent solvent streams to treat and the primary byproduct is hydrogen chloride, which can be easily scrubbed and potentially valorized. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturer, meeting the increasingly strict ESG (Environmental, Social, and Governance) criteria demanded by multinational end-users. Consequently, this technology future-proofs the supply chain against tightening environmental regulations, ensuring long-term viability and market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this key intermediate, based on the specific embodiments and data provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this material into their existing supply networks. The answers reflect the optimized conditions and performance metrics achieved through the novel catalytic system, providing clarity on purity, scalability, and process safety.
Q: What are the primary advantages of this gas-phase method over traditional thermal chlorination?
A: Unlike traditional thermal chlorination which requires temperatures above 380°C leading to coking and high energy consumption, this patented method operates efficiently at 220-360°C. Furthermore, it eliminates the need for toxic organic solvents like carbon tetrachloride, drastically simplifying downstream separation and waste treatment.
Q: Which catalysts provide the highest selectivity for the 2-chloro isomer?
A: The patent highlights that supported palladium catalysts (specifically 1-2% Pd on activated carbon or alumina) and certain alkaline earth metal salts (like MgF2 or CaCl2) exhibit exceptional regioselectivity. These catalysts promote chlorination specifically at the 2-position of the pyridine ring, minimizing the formation of unwanted polychlorinated byproducts.
Q: Is this process suitable for large-scale continuous manufacturing?
A: Yes, the process is explicitly designed for scalability using fixed-bed or fluidized-bed reactors. The absence of solvents and the use of robust heterogeneous catalysts allow for continuous operation with extended catalyst life, making it highly suitable for multi-ton commercial production of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of 2-chloro-5-trifluoromethylpyridine in the synthesis of next-generation agrochemicals and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. By leveraging advanced catalytic technologies similar to those described in recent patents, we optimize our manufacturing processes to deliver superior quality while maintaining competitive lead times. Our facility is designed to handle complex fluorinated chemistries safely and efficiently, making us an ideal partner for your long-term sourcing strategies.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to verify specific COA data for a new formulation, our experts are ready to assist. We encourage you to request route feasibility assessments to explore how our manufacturing capabilities can enhance your product development timeline. Partnering with us ensures access to a stable, high-quality supply of essential building blocks, empowering your R&D and production teams to focus on innovation and market growth without supply chain interruptions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
