Advanced Synthesis of 9-Aryl-10-Aryloxyanthracene Derivatives for Commercial OLED Manufacturing
Advanced Synthesis of 9-Aryl-10-Aryloxyanthracene Derivatives for Commercial OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit superior photophysical properties but also possess synthetic routes amenable to large-scale production. Patent CN112939813B introduces a groundbreaking class of 9-aryl-10-aryloxyanthracene derivatives that address the critical bottleneck of Aggregation-Caused Quenching (ACQ) through robust Aggregation-Induced Emission (AIE) characteristics. This technology represents a significant leap forward for R&D directors seeking high-purity luminescent materials with exceptional thermal stability and quantum yields reaching up to 93.2% in the solid state. By leveraging a novel one-step nucleophilic substitution strategy, this patent provides a viable pathway for the commercial scale-up of complex anthracene derivatives, ensuring that supply chain heads can secure reliable OLED material supplier partnerships without compromising on quality or delivery timelines.
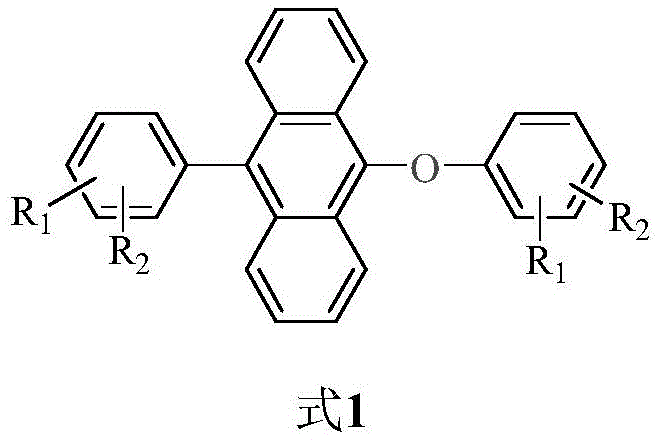
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance anthracene-based luminescent materials has been plagued by multi-step procedures that rely heavily on expensive transition metal catalysts and toxic reagents. Traditional routes often involve complex cross-coupling reactions that require stringent anhydrous conditions and extensive purification processes to remove residual metal impurities, which can be detrimental to the longevity and efficiency of OLED devices. Furthermore, many conventional anthracene derivatives suffer from the ACQ effect, where fluorescence is significantly quenched in the solid state or high-concentration solutions, limiting their practical application in thin-film devices. These synthetic complexities not only drive up the cost reduction in electronic chemical manufacturing but also introduce significant variability in batch-to-batch consistency, posing a severe risk for procurement managers aiming to stabilize their supply chains for high-value display components.
The Novel Approach
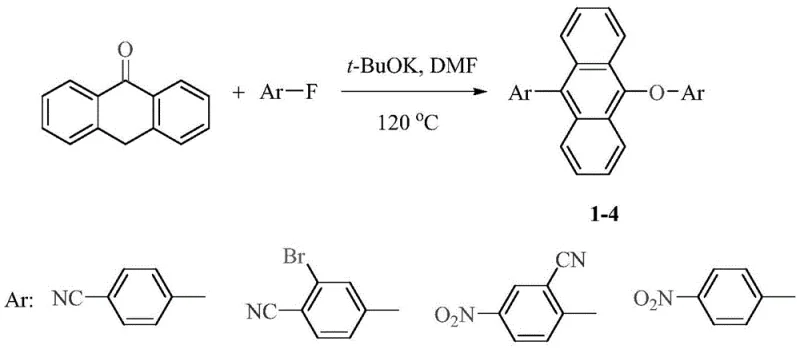
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN112939813B utilizes a streamlined one-step nucleophilic substitution reaction between 9(10H)-anthrone and fluorobenzene compounds. This approach eliminates the need for precious metal catalysts in the primary bond-forming step, utilizing instead a cost-effective alkali promoter such as potassium tert-butoxide. The reaction conditions are remarkably mild, typically proceeding at temperatures around 120°C in DMF solvent, which simplifies the engineering requirements for reactor setups. This methodological shift drastically reduces the operational complexity and waste generation associated with traditional synthesis, thereby enhancing the overall sustainability profile of the manufacturing process. For supply chain stakeholders, this translates to reducing lead time for high-purity OLED materials, as the simplified workflow allows for faster turnaround from raw material intake to finished product isolation without the need for extensive metal scavenging steps.
Mechanistic Insights into t-BuOK-Promoted Nucleophilic Substitution
The core innovation of this patent lies in the detailed mechanistic pathway that enables the efficient formation of the 9-aryl-10-aryloxyanthracene scaffold. The reaction initiates with the deprotonation of 9(10H)-anthrone by the alkali promoter, generating a reactive anionic species that undergoes nucleophilic attack on the fluorobenzene substrate. Unlike standard substitution reactions, this process involves a sophisticated intramolecular isomerization step where the intermediate anion rearranges to form an anthracenoxy anion before the final substitution occurs. This unique mechanistic feature ensures high regioselectivity and minimizes the formation of unwanted by-products, which is crucial for achieving the high purity specifications required in electronic grade chemicals. Understanding this mechanism allows R&D teams to fine-tune reaction parameters such as base strength and solvent polarity to maximize yield and minimize impurity profiles.
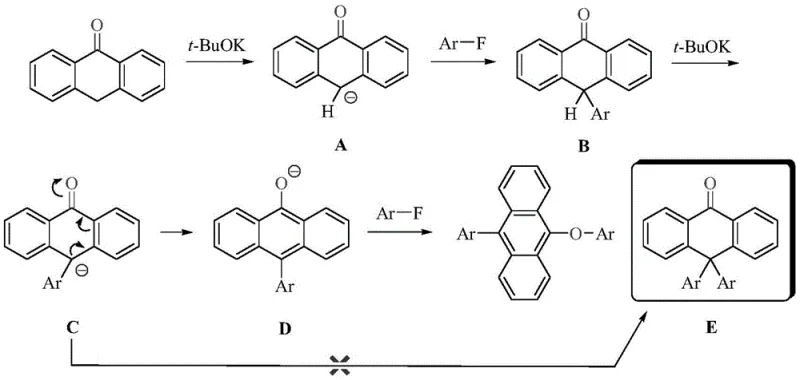
Furthermore, the control over impurity generation is inherently built into the reaction design through the selection of specific fluorobenzene derivatives with electron-withdrawing groups like cyano or nitro substituents. These groups activate the aromatic ring towards nucleophilic attack while simultaneously stabilizing the intermediate species, preventing side reactions that could lead to complex impurity spectra. The patent data indicates that the resulting derivatives exhibit excellent photoluminescence quantum yields and thermal stability, with decomposition temperatures exceeding 265°C for most variants. This level of thermal robustness is essential for maintaining device integrity during the vacuum deposition processes used in OLED panel fabrication. By mastering these mechanistic nuances, manufacturers can ensure consistent product quality that meets the rigorous standards of international display manufacturers.
How to Synthesize 9-Aryl-10-Aryloxyanthracene Derivatives Efficiently
The practical implementation of this synthesis route is designed for scalability, allowing chemical engineers to transition from gram-scale laboratory experiments to ton-scale commercial production with minimal re-optimization. The process begins with the precise stoichiometric mixing of 9(10H)-anthrone and the selected fluorobenzene compound in a polar aprotic solvent, followed by the controlled addition of the alkali promoter under an inert atmosphere. Detailed standardized synthesis steps are provided below to guide process development teams in establishing robust manufacturing protocols that ensure safety and efficiency. Adhering to these guidelines is critical for maintaining the high purity and yield metrics that define the commercial viability of this technology.
- Mix 9(10H)-anthrone with fluorobenzene compounds and potassium tert-butoxide in DMF solvent.
- Heat the reaction mixture to 120°C and stir for 12 hours under nitrogen atmosphere.
- Cool, precipitate in ethanol/water, filter, and recrystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis technology offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for electronic chemicals. The elimination of expensive transition metal catalysts in the primary synthetic step significantly lowers the raw material costs and reduces the dependency on volatile precious metal markets. Additionally, the simplified workup procedure, which often involves simple precipitation and filtration rather than complex chromatography, reduces solvent consumption and waste disposal costs. These factors collectively contribute to substantial cost savings in electronic chemical manufacturing, making the final luminescent materials more competitive in the global market while maintaining high performance standards.
- Cost Reduction in Manufacturing: The use of readily available starting materials like 9(10H)-anthrone and substituted fluorobenzenes, combined with inexpensive alkali promoters, drastically reduces the bill of materials compared to palladium-catalyzed routes. The absence of heavy metal catalysts also removes the need for costly metal scavenging resins and extensive purification steps, leading to a leaner and more cost-effective production process. This economic efficiency allows for better margin management and pricing flexibility when supplying high-purity luminescent materials to downstream device manufacturers.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals that are widely available from multiple global suppliers, the risk of raw material shortages is significantly mitigated. The robust nature of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent output and reliable delivery schedules. This stability is crucial for maintaining the continuity of supply for critical OLED components, preventing production delays that could impact the entire display manufacturing value chain.
- Scalability and Environmental Compliance: The one-step nature of the synthesis facilitates easier scale-up from pilot plants to full commercial production without the need for complex reactor modifications. Furthermore, the reduced use of toxic reagents and the potential for solvent recovery align with increasingly stringent environmental regulations, enhancing the sustainability profile of the manufacturing operation. This compliance not only avoids regulatory penalties but also appeals to eco-conscious partners seeking green chemistry solutions in their supply base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced luminescent materials. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their product lines. Understanding these details is essential for making informed decisions about material selection and process adoption.
Q: How does this patent solve the ACQ effect in solid-state lighting?
A: The 9-aryl-10-aryloxyanthracene derivatives exhibit Aggregation-Induced Emission (AIE) properties, meaning they emit strong fluorescence in the solid state rather than quenching, solving the traditional Aggregation-Caused Quenching (ACQ) limitation.
Q: What are the thermal stability characteristics of these luminescent materials?
A: The derivatives demonstrate excellent thermal stability with decomposition temperatures (Td5) ranging from 265°C to 357°C, making them highly suitable for the rigorous processing conditions of OLED device fabrication.
Q: Is the synthesis method scalable for industrial production?
A: Yes, the method utilizes a one-step nucleophilic substitution with readily available reagents like 9(10H)-anthrone and fluorobenzenes, avoiding expensive transition metal catalysts in the primary step, which facilitates commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Aryl-10-Aryloxyanthracene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercially viable products that meet the exacting standards of the global electronics industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising synthesis routes described in CN112939813B can be effectively implemented at an industrial level. We are committed to delivering products with stringent purity specifications and supporting our clients with rigorous QC labs that verify every batch against the highest performance metrics for OLED applications.
We invite you to collaborate with us to explore how these advanced anthracene derivatives can enhance your product portfolio and drive efficiency in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
