Revolutionizing D-Gluconic Acid-Gamma-Lactone Production: A High-Yield Silylation Strategy for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical intermediates like D-gluconic acid-γ-lactone (CAS: 1198-69-2), a compound pivotal in both food additive applications and emerging anti-diabetic drug research. Patent CN110759941A introduces a transformative synthetic methodology that addresses the longstanding inefficiencies of traditional production routes. By leveraging a strategic silylation-protection strategy starting from D-glucolactone, this technology enables the production of high-purity intermediates with exceptional stability and yield. Unlike conventional oxidative pathways that struggle with selectivity and waste generation, this novel approach utilizes accessible silane reagents and mild basic conditions to construct the target molecular architecture. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in process chemistry, offering a reliable foundation for securing the supply of high-purity pharmaceutical intermediates while drastically simplifying the operational complexity associated with carbohydrate derivative synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of D-gluconic acid-γ-lactone has been plagued by inefficient multi-step sequences that compromise both economic viability and environmental sustainability. Traditional literature methods, such as those described in the Journal of Carbohydrate Chemistry, typically initiate from D-(+)-glucose, requiring harsh oxidation conditions using calcium salts followed by complex lactonization steps in dioxane-water systems. These legacy processes are characterized by a dismal overall yield of approximately 48.8%, meaning more than half of the valuable starting material is lost to side reactions or purification losses. Furthermore, alternative high-temperature aqueous methods often result in intractable mixtures of γ-lactone and 1,5-lactone isomers, creating severe downstream purification bottlenecks that increase solvent consumption and waste disposal costs. For supply chain managers, these inefficiencies translate into volatile pricing and unreliable delivery schedules, as the low throughput of these archaic methods struggles to meet the growing demand from the nutraceutical and pharmaceutical sectors.
The Novel Approach
In stark contrast, the methodology disclosed in CN110759941A circumvents these oxidative hurdles by employing D-glucolactone as a direct precursor, utilizing a sophisticated silylation strategy to protect hydroxyl functionalities selectively. This route operates under mild conditions, typically between -20°C and 80°C, using inexpensive silane reagents like trimethylchlorosilane in the presence of catalytic DMAP and stoichiometric bases. The result is a dramatic improvement in process efficiency, with experimental data demonstrating yields consistently exceeding 90% and product purities reaching upwards of 99%. By shifting the synthetic logic from oxidation to protection-deprotection, the process eliminates the formation of difficult-to-separate isomeric byproducts, thereby streamlining the isolation procedure to simple filtration and crystallization steps. This technological pivot not only enhances the atom economy of the synthesis but also provides a robust platform for the commercial scale-up of complex carbohydrate derivatives, ensuring a steady supply of high-quality material for global markets.
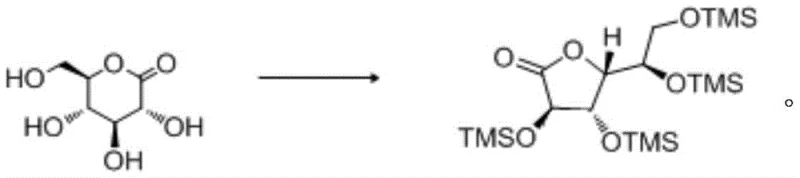
Mechanistic Insights into Silylation-Mediated Lactone Synthesis
The core innovation of this process lies in the precise control of nucleophilic substitution at the silicon center, facilitated by the synergistic action of organic bases and nucleophilic catalysts. In the initial step, the hydroxyl groups of the D-glucolactone substrate act as nucleophiles, attacking the silicon atom of the silane reagent (e.g., TMSCl) to form stable silyl ethers. The presence of 4-dimethylaminopyridine (DMAP) is critical here, as it acts as a nucleophilic catalyst that accelerates the silylation rate by forming a highly reactive silyl-pyridinium intermediate, which is then rapidly displaced by the alcohol oxygen. This mechanism ensures rapid and exhaustive protection of the hydroxyl groups, preventing unwanted intermolecular reactions or polymerization that could degrade the lactone ring. The choice of base, such as N-methylcyclohexylamine or triethylamine, serves to scavenge the generated hydrochloric acid, driving the equilibrium towards the product while maintaining a pH environment that preserves the integrity of the sensitive lactone moiety. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or optimize the process, as slight deviations in base strength or catalyst loading can significantly impact the impurity profile of the resulting intermediate.
Following the formation of the fully protected intermediate, the subsequent deprotection step relies on the high affinity of fluoride ions or protons for silicon, effectively cleaving the Si-O bonds to regenerate the free hydroxyl groups. When using fluoride sources like potassium fluoride (KF) or tetraalkylammonium fluorides, the reaction proceeds via the formation of a hypervalent silicon-fluoride species, which weakens the silicon-oxygen bond and facilitates its rupture under mild conditions. Alternatively, acidic deprotection using carboxylic acids like acetic acid leverages protonation of the silyl ether oxygen, making the silicon center more electrophilic and susceptible to nucleophilic attack by the solvent. This dual-pathway flexibility allows manufacturers to select the most cost-effective or environmentally benign deprotection agent based on their specific facility capabilities. Crucially, the purification strategy employs n-heptane crystallization for the intermediate and ethanol recrystallization for the final product, exploiting solubility differences to exclude trace siloxanes and unreacted starting materials, thereby guaranteeing the stringent purity specifications required for pharmaceutical applications.
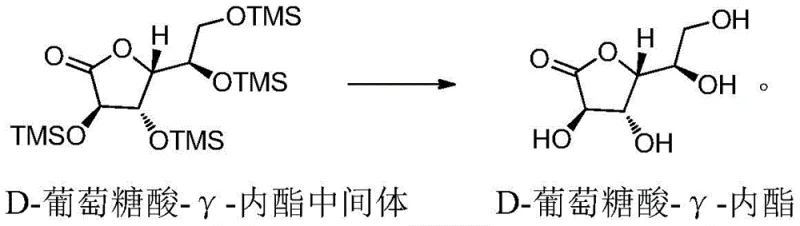
How to Synthesize D-Gluconic Acid-Gamma-Lactone Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity, allowing for seamless integration into existing fine chemical manufacturing lines without the need for specialized high-pressure or cryogenic equipment. The process begins with the dissolution of D-glucolactone in tetrahydrofuran (THF), followed by the controlled addition of the base and silane reagent at low temperatures to manage exotherms and ensure selectivity. Detailed standard operating procedures regarding stoichiometry, addition rates, and crystallization parameters are essential for maintaining batch-to-batch consistency. The following guide outlines the critical workflow derived from the patent data, serving as a foundational reference for process engineers looking to implement this superior synthetic route.
- React D-glucolactone with a silane reagent (e.g., TMSCl) and a base (e.g., N-methylcyclohexylamine) in THF at controlled low temperatures (-5 to 25°C) to form the protected intermediate.
- Purify the intermediate via filtration and crystallization using n-heptane to ensure high solid-state purity before the final deprotection step.
- Cleave the silyl protecting groups using a fluoride source (e.g., KF) or organic acid (e.g., acetic acid) in an alcoholic solvent, followed by recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silylation-based technology offers profound strategic advantages that extend far beyond simple yield improvements. By transitioning away from the erratic and low-yielding oxidative pathways, organizations can secure a more predictable and cost-effective supply of D-gluconic acid-γ-lactone. The elimination of expensive oxidants and the reduction of purification steps directly correlate to a significant reduction in manufacturing overheads, allowing for more competitive pricing structures in long-term supply contracts. Furthermore, the use of commodity chemicals like silanes and common organic solvents mitigates the risk of raw material shortages that often plague specialty reagent-dependent syntheses. This resilience is crucial for maintaining continuity in the production of downstream anti-diabetic medications and food additives, where supply disruptions can have cascading effects on global inventory levels.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic improvement in material throughput and the simplification of downstream processing. By achieving yields nearly double that of traditional methods, the effective cost per kilogram of the active intermediate is substantially lowered, as less starting material is wasted. Additionally, the avoidance of transition metal catalysts eliminates the need for costly and time-consuming heavy metal scavenging steps, which are often required to meet regulatory limits in pharmaceutical products. The ability to isolate the intermediate as a stable solid via simple filtration further reduces energy consumption associated with distillation or chromatography, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on D-glucolactone as a starting material, rather than D-glucose, shortens the synthetic timeline and reduces the number of unit operations required. This streamlined workflow minimizes the potential for equipment bottlenecks and scheduling conflicts within multipurpose production facilities. Moreover, the robustness of the silylation chemistry against minor variations in reaction conditions ensures high batch success rates, reducing the frequency of failed batches that can disrupt supply commitments. For supply chain planners, this reliability translates into shorter lead times and the ability to respond more agilely to fluctuations in market demand for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles by minimizing waste generation and solvent usage. The high selectivity of the reaction means fewer byproducts are formed, simplifying wastewater treatment and reducing the burden on effluent management systems. The solvents employed, such as THF, methanol, and heptane, are well-understood industrially and can be efficiently recovered and recycled, further lowering the environmental footprint. This compliance readiness facilitates smoother regulatory approvals and audits, ensuring that the manufacturing site remains operational without interruption due to environmental non-compliance issues, thus safeguarding the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of purity, yield, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for large-scale production.
Q: How does this silylation method improve yield compared to traditional glucose oxidation routes?
A: Traditional methods starting from D-glucose often suffer from multi-step oxidation and lactonization issues, resulting in yields as low as 48.8%. The patented silylation route starts directly from glucolactone, bypassing oxidation steps and achieving yields consistently above 90% through efficient protection-deprotection chemistry.
Q: What are the critical parameters for controlling impurity profiles in this synthesis?
A: Temperature control during the silylation step is paramount. Maintaining the reaction between -5°C and 25°C prevents side reactions. Additionally, using a precise molar excess of the base (optimally 4.5 equivalents) ensures complete conversion while minimizing degradation, leading to purities exceeding 99%.
Q: Is this process scalable for industrial manufacturing of food and pharma grade intermediates?
A: Yes, the process utilizes common industrial solvents like THF, methanol, and heptane, and avoids expensive transition metal catalysts. The isolation involves standard filtration and crystallization techniques, making it highly amenable to scale-up from pilot plants to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Gluconic Acid-Gamma-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-yield synthetic routes in maintaining a competitive edge in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the silylation technology described in CN110759941A and confirmed its immense potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of D-gluconic acid-γ-lactone we deliver meets the highest standards required for API and food-grade applications.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains using this advanced technology. By leveraging our expertise, you can achieve substantial cost savings and secure a stable supply of this vital intermediate. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
