Advanced Synthesis of 2-Chloro-4-Fluoro-5-Nitrobenzoic Acid for Commercial Scale-Up
The chemical landscape for producing critical agrochemical intermediates is undergoing a significant transformation, driven by the urgent need for greener, more efficient synthetic pathways. Patent CN114105772A introduces a groundbreaking methodology for the synthesis of 2-chloro-4-fluoro-5-nitrobenzoic acid, a pivotal building block in the manufacture of the herbicide saflufenacil. This innovative protocol departs from traditional, harsh nitration techniques by employing a strategic sequence of photochlorination followed by controlled mixed-acid nitration and a streamlined one-pot hydrolysis-oxidation. For R&D directors and process chemists, this represents a paradigm shift towards higher atom economy and superior impurity control. By utilizing 2-chloro-4-fluorotoluene as the foundational feedstock, the process mitigates the formation of troublesome isomers that have historically plagued direct nitration routes, thereby ensuring a robust supply of high-purity material essential for downstream drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-chloro-4-fluoro-5-nitrobenzoic acid has been fraught with significant technical and economic challenges that hinder scalable production. Traditional methods often rely on the direct nitration of 2-chloro-4-fluorobenzoic acid using mixed acids, a process notoriously difficult to control regarding regioselectivity. Literature and experimental data indicate that this conventional approach frequently generates substantial quantities of isomeric byproducts, ranging from 10% to 20%, which necessitates multiple, yield-depleting recrystallization steps to achieve acceptable purity. Furthermore, alternative pathways involving 2-chloro-4-fluorotrichlorotoluene require extreme reaction conditions, such as intense light exposure and elevated temperatures, imposing severe demands on reactor materials and safety systems. These legacy processes not only suffer from low overall yields, often hovering around 40%, but also generate complex waste streams containing heavy metal catalysts that complicate environmental compliance and wastewater treatment protocols.
The Novel Approach
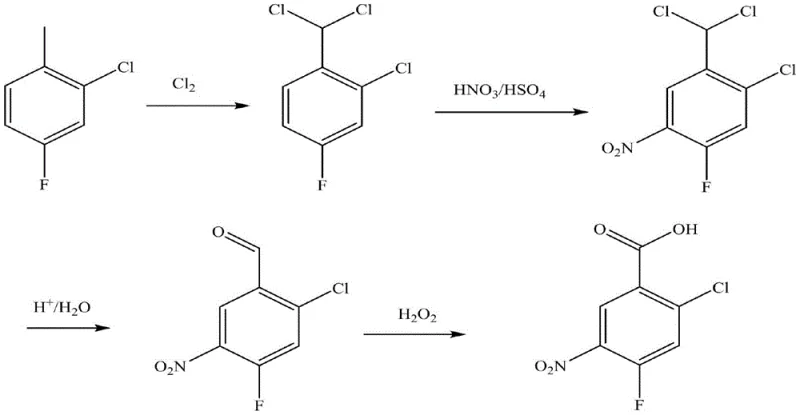
In stark contrast, the novel synthetic route detailed in the patent data offers a sophisticated solution by reversing the order of functionalization to exploit electronic effects for better selectivity. By initially subjecting 2-chloro-4-fluorotoluene to photochlorination, the methyl group is activated to a dichloromethyl moiety under mild thermal conditions, avoiding the degradation associated with high-energy inputs. This intermediate then undergoes nitration where the electron-withdrawing nature of the dichloromethyl group, combined with the specific solvent environment, effectively suppresses the formation of unwanted isomers. The subsequent conversion to the final acid is achieved through an elegant one-pot hydrolysis and oxidation sequence, eliminating the need for isolating unstable aldehyde intermediates. This streamlined approach not only boosts the total reaction yield to over 80% but also drastically simplifies the operational workflow, making it an ideal candidate for cost reduction in agrochemical manufacturing.
Mechanistic Insights into Photochlorination and Regioselective Nitration
The core innovation of this synthesis lies in the precise manipulation of radical and electrophilic substitution mechanisms to achieve unparalleled purity profiles. The initial photochlorination step utilizes UV or fluorescent light to generate chlorine radicals that selectively abstract hydrogen atoms from the benzylic position of the toluene derivative. This radical chain reaction is carefully monitored to ensure the formation of the gem-dichloride species while minimizing polychlorination or ring chlorination, which are common side reactions in unoptimized processes. The resulting 2-chloro-4-fluorobenzylidene dichloride serves as a superior substrate for the subsequent nitration because the dichloromethyl group exerts a strong meta-directing influence that aligns perfectly with the desired substitution pattern relative to the existing halogen substituents. This electronic alignment is crucial for preventing the formation of the 3-nitro or 6-nitro isomers that typically contaminate batches produced via older methodologies.
Furthermore, the final transformation stage employs a tandem hydrolysis-oxidation mechanism that exemplifies process intensification principles. In the presence of acidic catalysts like zinc chloride or ferric chloride, the gem-dichloride moiety is hydrolyzed to the corresponding benzaldehyde intermediate without isolation. Immediately following this, the introduction of hydrogen peroxide acts as a green oxidant to convert the aldehyde directly into the carboxylic acid functionality. This one-pot strategy is mechanistically advantageous because it prevents the accumulation of the aldehyde, which can be prone to polymerization or further unwanted side reactions if left exposed. The use of hydrogen peroxide ensures that the only byproduct of the oxidation step is water, significantly reducing the toxic load of the effluent compared to chromium or manganese-based oxidants used in historical contexts, thus aligning with modern green chemistry mandates.
How to Synthesize 2-Chloro-4-Fluoro-5-Nitrobenzoic Acid Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature control and reagent stoichiometry to maximize the benefits of the novel pathway. The process begins with the photochlorination of the toluene starting material at temperatures between 60-100°C, ensuring the complete conversion to the dichloride while monitoring for residual monochloride. Following this, the nitration is conducted at low temperatures (0-30°C) in a solvent system such as dichloromethane or chloroform to manage the exotherm and maintain selectivity. The final step involves heating the nitrated intermediate in a carboxylic acid solvent with a Lewis acid catalyst, followed by the controlled addition of oxidant. For a comprehensive understanding of the specific operational parameters, safety precautions, and workup procedures required to replicate these results in a pilot or production setting, please refer to the standardized synthesis guide below.
- Perform photochlorination on 2-chloro-4-fluorotoluene using UV or fluorescent light at 60-100°C to obtain 2-chloro-4-fluorobenzylidene dichloride.
- Conduct mixed acid nitration on the dichloride intermediate at 0-30°C in a solvent like dichloromethane to introduce the nitro group selectively.
- Execute a one-pot hydrolysis using formic or acetic acid followed by oxidation with hydrogen peroxide to convert the dichloromethyl group to carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route translates into tangible strategic advantages regarding cost stability and supply continuity. The elimination of heavy metal catalysts and the reduction in nitric acid consumption directly correlate to a significant decrease in raw material costs and waste disposal fees. By avoiding the complex purification trains required to remove isomeric impurities found in conventional batches, manufacturers can achieve higher throughput with existing infrastructure. This efficiency gain means that production cycles are shortened, allowing for faster response times to market demand fluctuations without the need for massive capital expenditure on new specialized equipment. The reliance on commodity chemicals like chlorine, hydrogen peroxide, and standard organic solvents further insulates the supply chain from the volatility associated with exotic or highly regulated reagents.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substantial increase in overall yield, which exceeds 80% compared to the roughly 40% yield of traditional methods. This doubling of efficiency effectively halves the raw material cost per kilogram of the final active intermediate. Additionally, the removal of expensive heavy metal catalysts eliminates the need for costly scavenging steps and specialized wastewater treatment facilities, leading to drastic operational expenditure savings. The mild reaction conditions also reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and reduced utility bills for the manufacturing site.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved because the starting material, 2-chloro-4-fluorotoluene, is a widely available commodity in the fine chemical sector. The process does not depend on scarce or geopolitically sensitive reagents, ensuring a stable input flow even during global supply disruptions. Furthermore, the robustness of the reaction conditions means that production is less susceptible to batch failures caused by minor deviations in temperature or pressure, guaranteeing consistent delivery schedules for downstream customers. This predictability allows supply chain planners to maintain leaner inventory levels while still meeting strict Just-In-Time delivery commitments.
- Scalability and Environmental Compliance: From a scalability perspective, the process is designed for seamless transition from laboratory to multi-ton production without requiring high-pressure reactors or exotic metallurgy. The use of standard glass-lined or stainless steel reactors makes it accessible for most contract development and manufacturing organizations (CDMOs). Environmentally, the process generates significantly less hazardous waste, particularly by avoiding heavy metals and minimizing acidic effluent volume. This facilitates easier compliance with increasingly stringent environmental regulations, reducing the risk of production shutdowns due to permit violations and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthetic pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is becoming the preferred choice for high-volume production. Understanding these nuances is critical for technical teams evaluating technology transfer opportunities or assessing the feasibility of integrating this intermediate into their existing portfolio of agrochemical actives.
Q: Why is the photochlorination-first route superior to direct nitration of the benzoic acid?
A: Direct nitration of 2-chloro-4-fluorobenzoic acid typically results in significant isomer formation (10-20%) and lower yields around 40%. The novel route nitrates the dichloromethyl intermediate, which allows for better regioselectivity and inhibits isomer generation, achieving yields above 80%.
Q: What are the environmental benefits of this synthesis method?
A: This method avoids the use of heavy metal catalysts often required in traditional pathways, simplifying wastewater treatment. Additionally, the mild reaction temperatures and reduced consumption of nitric acid contribute to lower three-waste emissions and energy usage.
Q: What purity levels can be achieved with this process?
A: Through optimized recrystallization steps using solvents like ethanol/water or methanol/water, the final product consistently achieves HPLC purity levels exceeding 99%, meeting stringent requirements for pharmaceutical and agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4-Fluoro-5-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-chloro-4-fluoro-5-nitrobenzoic acid meets the exacting standards required for herbicide synthesis. Our commitment to quality assurance means that we can consistently deliver material with purity profiles that exceed industry norms, minimizing risks for your downstream processing.
We invite you to collaborate with us to optimize your supply chain for saflufenacil production. By leveraging our technical proficiency in photochlorination and nitration chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than estimates. Let us help you secure a sustainable, high-quality supply of this critical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
