Advanced Synthetic Route for Saxagliptin Intermediate A1 via Grignard Reaction
Advanced Synthetic Route for Saxagliptin Intermediate A1 via Grignard Reaction
The global demand for Dipeptidyl Peptidase-IV (DPP-IV) inhibitors continues to surge as Type 2 diabetes prevalence rises, placing immense pressure on the supply chains for critical active pharmaceutical ingredients (APIs) like Saxagliptin. At the heart of this complex molecule lies the adamantane scaffold, specifically the 3-hydroxyadamantane-1-carboxylic acid derivative known as intermediate A1, which serves as a pivotal chiral building block. Recent intellectual property developments, specifically patent CN102807487A, have unveiled a streamlined chemical synthesis strategy that bypasses the limitations of traditional multi-step pathways. This innovative methodology leverages a robust Grignard reaction followed by a controlled mixed-acid hydroxylation to deliver high-purity intermediates suitable for large-scale manufacturing. By optimizing reaction conditions and selecting cost-effective reagents, this approach addresses the critical needs of modern pharmaceutical production for efficiency and reliability.
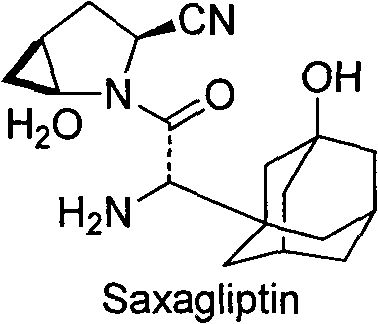
Understanding the structural complexity of Saxagliptin is essential for appreciating the synthetic challenges involved in constructing its adamantane core. The molecule features a unique fusion of a lipophilic adamantane cage with a polar nitrile-substituted pyrrolidine ring, requiring precise stereochemical control during assembly. Intermediate A1 represents the functionalized adamantane precursor that eventually couples with the amine component to form the final drug substance. The ability to produce this intermediate with consistent quality and high purity is paramount for downstream processing, as impurities at this stage can propagate through the synthesis, complicating purification and impacting the safety profile of the final medication. Consequently, the development of a robust, scalable route for A1 is a strategic priority for any reliable pharmaceutical intermediate supplier aiming to support the global generic and branded drug markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Saxagliptin intermediates often suffer from significant drawbacks that hinder their industrial viability, ranging from excessive step counts to the use of hazardous or expensive reagents. For instance, earlier routes described in literature such as WO2010032129 involve the oxidation of acetyl adamantane using alkaline potassium permanganate, a process that generates substantial manganese waste and requires difficult-to-source starting materials. Other pathways, like those utilizing trimethylsilyl cyanide or complex enzymatic resolutions, introduce additional operational burdens, including the need for strict anhydrous conditions, expensive biocatalysts, or toxic cyanide handling protocols. Furthermore, many of these conventional routes exhibit suboptimal overall yields and struggle to maintain high stereochemical purity without resorting to costly chromatographic separations. These inefficiencies translate directly into higher production costs and extended lead times, creating bottlenecks for procurement managers seeking cost reduction in API manufacturing.
The Novel Approach
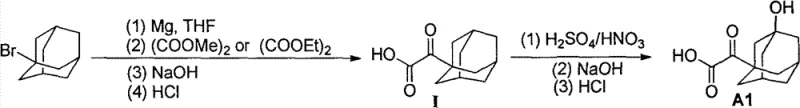
In stark contrast to these cumbersome legacy processes, the method disclosed in CN102807487A offers a remarkably concise two-step synthesis that dramatically simplifies the production landscape. This novel approach initiates with the formation of a Grignard reagent from readily available bromoadamantane, which then reacts with a diethyl oxalate ester at low temperatures to construct the keto-acid framework in a single pot. The subsequent step involves a direct hydroxylation using a mixed acid system (sulfuric and nitric acid) to install the crucial hydroxyl group on the adamantane cage. This strategy eliminates the need for protecting groups, enzymatic steps, or transition metal catalysts, thereby reducing the total number of unit operations and minimizing waste generation. The result is a streamlined workflow that achieves a total yield of 52% and a purity exceeding 99%, demonstrating superior process economics and operational simplicity compared to prior art.
Mechanistic Insights into Grignard Carbonylation and Mixed Acid Hydroxylation
The core of this synthetic breakthrough lies in the precise execution of the Grignard carbonylation step, where bromoadamantane is converted into a highly reactive organomagnesium species in a tetrahydrofuran (THF) solvent system. The reaction is meticulously controlled at cryogenic temperatures, typically around -70°C, to prevent side reactions such as Wurtz coupling or over-addition to the oxalate ester. Upon addition of the diethyl oxalate, the Grignard reagent attacks one of the carbonyl groups, forming a tetrahedral intermediate that collapses to a ketone upon acidic workup, effectively installing the alpha-keto acid functionality onto the rigid adamantane skeleton. This transformation is critical because it establishes the carbon-carbon bond necessary for the carboxylic acid moiety while maintaining the integrity of the bulky cage structure. The choice of THF as the solvent is particularly advantageous due to its ability to stabilize the Grignard reagent and facilitate heat transfer during the exothermic addition, ensuring consistent batch-to-batch reproducibility.
Following the formation of the adamantanone acid compound, the second mechanistic phase involves an electrophilic substitution reaction driven by the mixed acid system to introduce the hydroxyl group. In this step, the nitric acid acts as a source of nitronium ions or related reactive nitrogen species that facilitate the oxidation of the tertiary C-H bond on the adamantane ring. The sulfuric acid serves both as a dehydrating agent and a catalyst to enhance the electrophilicity of the oxidizing species. This direct C-H functionalization is chemically elegant as it avoids the need for pre-functionalized precursors, directly converting the ketone-acid intermediate into the desired 3-hydroxy derivative. The mechanism likely proceeds through a radical or cationic pathway typical of adamantane oxidations, where the bridgehead positions are activated selectively. Rigorous control of acid concentration and temperature is essential here to prevent over-oxidation or degradation of the sensitive carboxylic acid group, ensuring the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Saxagliptin Intermediate A1 Efficiently
Implementing this synthesis requires careful attention to moisture exclusion during the Grignard formation and precise thermal management during the exothermic quenching phases. The process begins with the activation of magnesium turnings in dry THF under an inert nitrogen atmosphere, followed by the slow addition of bromoadamantane to generate the organometallic reagent. Once the Grignard solution is stabilized at -70°C, the diethyl oxalate is introduced dropwise to maintain temperature control, followed by a hydrolysis step using sodium hydroxide solution to liberate the free acid. The resulting crude adamantanone acid is then isolated and subjected to the nitration mixture, where reaction time and acid ratios are optimized to maximize the yield of the hydroxylated product. For detailed standard operating procedures and specific parameter ranges, please refer to the technical guide below.
- Prepare Grignard reagent from bromoadamantane and Mg in THF, react with diethyl oxalate at -70°C, then hydrolyze to obtain adamantanone acid compound I.
- Subject compound I to mixed acid (sulfuric/nitric acid) conditions to introduce the hydroxyl group, yielding the target intermediate A1.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of supply chain volatility and rising manufacturing costs in the pharmaceutical sector. By relying on commodity chemicals like bromoadamantane, magnesium, and oxalic acid esters, the process decouples production from the supply constraints often associated with specialized chiral catalysts or biologically derived enzymes. This shift to chemically robust reagents ensures a more predictable procurement cycle, allowing supply chain heads to secure raw materials with greater confidence and reduced risk of disruption. Furthermore, the elimination of complex purification steps, such as chiral chromatography or extensive recrystallization sequences found in other routes, significantly lowers the operational expenditure per kilogram of output. The ability to recycle solvents like THF through distillation further enhances the economic profile, aligning with sustainability goals while driving down variable costs.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the avoidance of expensive biocatalysts and precious metal catalysts, which often constitute a significant portion of the bill of materials in alternative syntheses. By utilizing a purely chemical approach with high atom economy in the Grignard step, manufacturers can achieve substantial cost savings without compromising on yield. Additionally, the short reaction sequence reduces labor hours, energy consumption for heating and cooling, and equipment occupancy time, all of which contribute to a lower cost of goods sold (COGS). The high purity of the crude product (>99%) also minimizes the need for resource-intensive downstream purification, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals ensures that the supply chain remains resilient against market fluctuations that might affect niche reagents. Bromoadamantane and oxalate esters are produced at scale by multiple global suppliers, providing procurement teams with the flexibility to diversify their vendor base and negotiate better terms. This redundancy is crucial for maintaining continuous production schedules, especially when scaling up to meet the demands of commercial API manufacturing. Moreover, the stability of the intermediates allows for potential storage and inventory buffering, offering an additional layer of security against unexpected supply interruptions or logistical delays.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations and avoiding hazardous reagents that require specialized containment systems. The waste management protocol described in the patent, which involves neutralizing acidic waste streams and recycling organic solvents, demonstrates a commitment to environmental stewardship that simplifies regulatory compliance. Reducing the generation of heavy metal waste, common in oxidation routes using permanganate or chromium, lowers the burden on waste treatment facilities and reduces disposal costs. This environmentally friendly profile not only mitigates regulatory risk but also enhances the corporate social responsibility (CSR) standing of the manufacturing entity, making it a more attractive partner for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, drawing directly from the experimental data and beneficial effects reported in the patent literature. Understanding these nuances is vital for R&D directors evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The answers provided reflect the specific advantages of the Grignard-based approach over competing technologies, highlighting its suitability for industrial application.
Q: What is the overall yield of the novel Saxagliptin intermediate A1 synthesis?
A: According to patent CN102807487A, the total yield for this specific two-step route can reach 52%, with a final product purity exceeding 99%.
Q: Why is the Grignard route preferred over enzymatic methods for this intermediate?
A: While enzymatic methods offer high selectivity, the chemical Grignard route described avoids the high costs and supply chain complexities associated with biocatalysts, utilizing cheap and easily accessible raw materials like bromoadamantane.
Q: How does this process handle environmental waste?
A: The process includes solvent recycling (THF) through distillation and neutralizes waste liquid in a soda acid pond before purification, significantly reducing environmental pollution compared to heavy metal oxidation routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Saxagliptin Intermediate A1 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Saxagliptin depends on a partnership grounded in technical excellence and supply chain reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Saxagliptin Intermediate A1 with stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of Grignard chemistry, guaranteeing a consistent and high-quality supply for your downstream synthesis needs.
We invite you to collaborate with us to optimize your supply chain and achieve significant competitive advantages in the marketplace. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your strategic goals. Let us be your trusted partner in navigating the complexities of API intermediate production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
