Advanced Nickel-Catalyzed Base-Free Suzuki Reaction for High-Purity Alcohol Intermediates
The landscape of organic synthesis is constantly evolving, driven by the need for more sustainable and efficient methodologies to construct complex molecular architectures. A significant breakthrough in this domain is documented in patent CN110724033B, which discloses a novel method for preparing alcohol compounds via a nickel-catalyzed Suzuki-type ring-opening coupling reaction. Unlike traditional approaches that rely heavily on stoichiometric amounts of strong exogenous bases, this innovation achieves high efficiency under base-free conditions. This technological leap addresses critical pain points in the manufacturing of pharmaceutical intermediates, specifically regarding functional group tolerance and reagent costs. By utilizing a robust nickel catalytic system, the process enables the coupling of diverse epoxides with aryl or alkenyl boronic acids, delivering high yields with exceptional purity profiles suitable for demanding industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alcohol compounds through the ring-opening of ternary epoxides has been dominated by copper-catalyzed protocols, such as those reported by Professor Fu's group. While effective for specific substrates, these conventional methods suffer from severe limitations that hinder their broad application in complex molecule synthesis. The most significant drawback is the absolute requirement for excessive amounts of lithium tert-butoxide (LiOtBu) as an exogenous base. This reagent is not only expensive but also possesses extremely strong alkalinity, which wreaks havoc on sensitive functional groups present in advanced intermediates. Furthermore, the substrate scope is notoriously narrow; typically, only terminal epoxides and simple aryl borons are tolerated, leaving chemists struggling to synthesize more complex 1,2-disubstituted alcohols or those bearing electrophilic moieties.
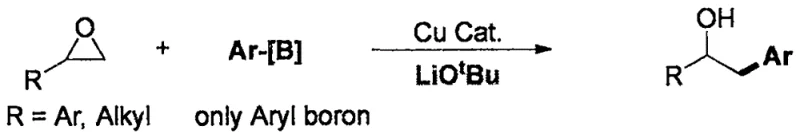
The Novel Approach
In stark contrast to the restrictive copper-based methodologies, the technology described in CN110724033B introduces a paradigm shift by employing a nickel-catalyzed system that operates entirely without exogenous base. This base-free protocol utilizes a combination of Nickel(II) bromide diglyme and a bulky bipyridine ligand (dtbpy) to facilitate the coupling. The elimination of strong alkali reagents fundamentally changes the reaction profile, allowing for the preservation of delicate functional groups that would otherwise be degraded. Moreover, this new approach dramatically expands the substrate scope to include 1,2-disubstituted epoxides and various substituted aryl or alkenyl boronic acids. The use of ethanol as a green solvent further enhances the environmental profile and operational safety of the process, making it a superior choice for modern chemical manufacturing.

Mechanistic Insights into Nickel-Catalyzed Epoxide Ring-Opening
The success of this base-free transformation lies in the unique mechanistic pathway enabled by the nickel catalyst and the iodide additive. The catalytic cycle likely involves the oxidative addition of the nickel species into the carbon-oxygen bond of the epoxide, activated by the iodide ion, followed by transmetallation with the boronic acid species. Crucially, the absence of an external base suggests that the activation of the boronic acid occurs through a distinct mechanism, possibly involving the formation of a reactive boronate species facilitated by the solvent or the metal center itself. This subtle mechanistic difference is what grants the reaction its remarkable chemoselectivity. By avoiding the harsh deprotonation events typical of base-mediated couplings, the system prevents side reactions such as aldol condensations or ester hydrolysis, ensuring that the final alcohol product retains the structural integrity of the starting materials.
From an impurity control perspective, this mechanism offers substantial advantages for the production of high-purity pharmaceutical intermediates. Traditional base-mediated reactions often generate complex impurity profiles due to non-selective nucleophilic attacks or base-catalyzed decomposition of the epoxide. In this nickel-catalyzed system, the reaction proceeds with high regioselectivity and minimal byproduct formation. The compatibility with electron-withdrawing groups is particularly noteworthy, as demonstrated by the successful coupling of substrates containing ketone and ester functionalities. For instance, the reaction tolerates acetyl groups and methoxycarbonyl groups without reduction or hydrolysis, which is a common failure mode in other transition-metal catalyzed systems. This level of precision reduces the burden on downstream purification processes, directly translating to higher overall process efficiency.
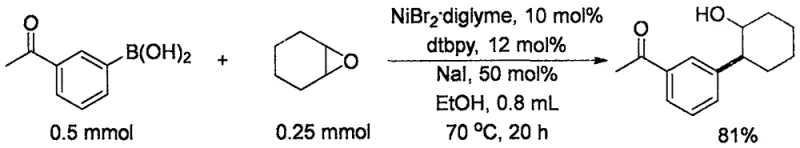
How to Synthesize Alcohol Intermediates Efficiently
Implementing this synthesis route requires careful attention to the catalytic loading and reaction atmosphere to ensure reproducibility and high yield. The process is designed to be operationally simple, utilizing readily available reagents and standard laboratory equipment, which facilitates easy translation from bench scale to pilot plant operations. The following guide outlines the standardized procedure derived from the patent examples, focusing on the optimal ratios of catalyst, ligand, and additives required to drive the reaction to completion. For detailed standard operating procedures and safety data sheets regarding the specific reagents mentioned, please refer to the technical documentation provided below.
- Prepare the catalytic system by mixing Nickel(II) bromide diglyme (10 mol%), dtbpy ligand (12 mol%), and Sodium Iodide (50 mol%) in a sealed reaction vessel under inert atmosphere.
- Add ethanol solvent followed by the arylboronic acid (2.0 equivalents) and the epoxide substrate (1.0 equivalent) to the reaction mixture.
- Heat the reaction mixture to 70°C for 12 hours, then perform standard aqueous workup and silica gel chromatography to isolate the pure alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-free nickel-catalyzed technology represents a strategic opportunity to optimize manufacturing costs and secure supply continuity. The removal of lithium tert-butoxide from the process equation eliminates a significant cost driver, as this reagent is both pricey and hazardous to handle in large quantities. Furthermore, the substitution of hazardous solvents with ethanol aligns with increasingly stringent environmental regulations, reducing waste disposal costs and simplifying compliance reporting. The robustness of the reaction conditions means that raw material specifications can be slightly relaxed without compromising yield, allowing for greater flexibility in sourcing bulk chemicals from multiple vendors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive stoichiometric bases and the use of earth-abundant nickel instead of precious metals like palladium. By removing the need for lithium tert-butoxide, manufacturers avoid the high procurement costs associated with this specialized reagent and the safety infrastructure required to store and handle it. Additionally, the high atom economy and reduced formation of side products mean that less raw material is wasted, leading to a lower cost of goods sold (COGS) per kilogram of final product. The simplified workup procedure, which avoids complex neutralization steps, further reduces labor and utility consumption during the isolation phase.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethanol, sodium iodide, and commercially available boronic acids ensures a stable and resilient supply chain. Unlike specialized catalysts or reagents that may have single-source suppliers and long lead times, the components of this reaction system are widely produced by global chemical manufacturers. This diversity of supply sources mitigates the risk of production stoppages due to raw material shortages. Furthermore, the mild reaction temperature of 70°C reduces energy consumption and places less stress on reactor equipment, extending the lifespan of capital assets and minimizing unplanned maintenance downtime.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage scale is straightforward due to the absence of exothermic base-addition steps, which are often a bottleneck in batch processing. The use of ethanol as a solvent significantly lowers the environmental footprint of the process, facilitating easier permitting and reducing the regulatory burden associated with volatile organic compound (VOC) emissions. The high selectivity of the reaction minimizes the generation of hazardous waste streams, simplifying effluent treatment and lowering the overall environmental compliance costs for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this nickel-catalyzed methodology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable foundation for process development decisions. Understanding these nuances is critical for R&D teams evaluating this technology for integration into existing synthetic routes.
Q: What is the primary advantage of this nickel-catalyzed method over traditional copper-catalyzed Suzuki reactions?
A: The primary advantage is the elimination of exogenous strong bases like lithium tert-butoxide. This base-free condition significantly improves functional group compatibility, allowing sensitive groups like ketones and esters to remain intact during the synthesis.
Q: What are the optimal reaction conditions for this transformation?
A: The optimal conditions involve using NiBr2·diglyme (10 mol%) and dtbpy (12 mol%) as the catalyst system with NaI (50 mol%) as an additive in ethanol solvent at 70°C for approximately 12 hours.
Q: Does this method support diverse substrate types beyond simple phenyl rings?
A: Yes, the method demonstrates excellent substrate scope, successfully tolerating electron-withdrawing groups such as acetyl and methoxycarbonyl functionalities, as well as alkenyl boronic acids, yielding high-purity products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alcohol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of base-free nickel catalysis in the synthesis of complex alcohol intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alcohol intermediate delivered meets the highest industry standards for pharmaceutical and fine chemical applications.
We invite you to leverage our technical expertise to evaluate the feasibility of this cost-effective synthesis route for your specific projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this technology can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in your supply chain.
