Advanced Synthesis of Diamine Monomers for High-Performance Polyimide Commercial Scale-Up
The rapid evolution of the flexible copper clad laminate (FCCL) industry demands insulating materials with exceptional thermal stability and mechanical strength, driving the need for advanced polyimide precursors. Patent CN101265206A introduces a breakthrough preparation method for 2,2-bis[4-(2,4-diaminophenoxy)-3,5-dimethylphenyl]propane, a critical monomer for high-performance polyimide resins. This technical insight report analyzes the proprietary catalytic transfer hydrogenation process detailed in the patent, highlighting its potential to revolutionize the supply chain for electronic chemical intermediates. By replacing traditional high-pressure hydrogenation with a hydrazine-based reduction system, this method offers a safer, more efficient pathway to achieving ultra-high purity standards required in microelectronics. For R&D Directors and Procurement Managers, understanding the nuances of this synthesis route is essential for optimizing cost structures and ensuring the reliability of high-purity electronic chemical supplies in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for aromatic diamines often rely on catalytic hydrogenation using molecular hydrogen under high pressure, which presents significant safety hazards and capital expenditure challenges for manufacturing facilities. These conventional methods typically require specialized high-pressure reactors, rigorous safety protocols, and complex gas handling systems that drastically increase operational overhead and lead time for high-purity electronic chemical production. Furthermore, the removal of residual metal catalysts from the final product can be notoriously difficult, often necessitating multiple recrystallization steps that reduce overall yield and increase solvent waste. The reliance on high-pressure infrastructure also limits the flexibility of production lines, making it difficult to adapt quickly to fluctuating market demands for specialty chemical intermediates. Consequently, manufacturers face substantial bottlenecks in scaling up production while maintaining the stringent purity specifications demanded by the semiconductor and display industries.
The Novel Approach
The method disclosed in patent CN101265206A circumvents these industrial bottlenecks by utilizing a catalytic transfer hydrogenation system with hydrazine hydrate and palladium on carbon in a saturated fatty alcohol medium. This innovative approach operates at atmospheric pressure within a moderate temperature range of 70°C to 85°C, eliminating the need for expensive high-pressure equipment and significantly enhancing operational safety. The process simplifies the workup procedure, as the catalyst can be easily removed by hot filtration, and the product crystallizes directly from the mother liquor upon cooling. This streamlined workflow not only reduces energy consumption but also minimizes the generation of hazardous waste, aligning with modern green chemistry principles. For supply chain heads, this translates to a more robust and flexible manufacturing process that can be rapidly deployed to meet the commercial scale-up of complex polymer additives and monomers without compromising on quality or safety standards.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthesis lies in the efficient reduction of the nitro groups to amino groups using hydrazine hydrate as the hydrogen donor, facilitated by the palladium on carbon catalyst. In this mechanism, hydrazine adsorbs onto the palladium surface where it decomposes to release nitrogen gas and active hydrogen species, which are then transferred to the nitro groups of the substrate. The use of a saturated fatty alcohol solvent system is critical, as it provides a stable medium that solubilizes the organic substrate while maintaining the dispersion of the heterogeneous catalyst. This specific solvent environment helps to control the reaction kinetics, preventing over-reduction or side reactions that could compromise the structural integrity of the sensitive aromatic backbone. The moderate temperature range of 70°C to 85°C ensures sufficient activation energy for the reduction while avoiding thermal degradation of the product, resulting in a clean reaction profile with minimal byproduct formation.
Controlling impurity profiles is paramount for applications in FCCL and advanced composites, where even trace contaminants can affect the dielectric properties of the final polyimide resin. The described method achieves purity levels exceeding 99% by leveraging the differential solubility of the product and impurities in the chosen solvent system during the crystallization phase. The hot filtration step effectively removes the palladium catalyst and any insoluble particulate matter, while the subsequent cooling of the mother liquor induces selective crystallization of the target diamine. This intrinsic purification capability reduces the need for downstream chromatographic separation, which is often a major cost driver in fine chemical manufacturing. By optimizing the molar ratio of hydrazine hydrate to substrate between 1:20 and 1:50, the process ensures complete conversion of the dinitro precursor, thereby minimizing the presence of partially reduced intermediates that could act as impurities in the final polymer matrix.
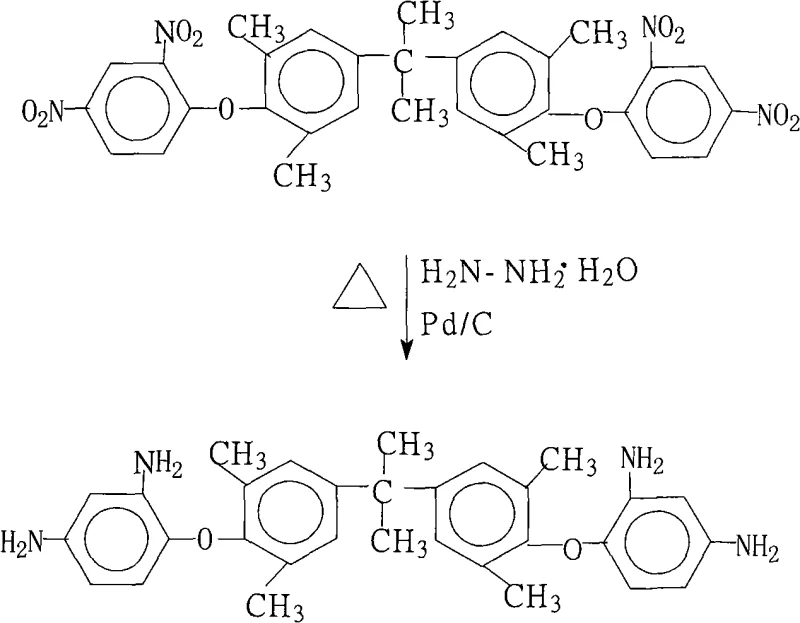
How to Synthesize 2,2-bis[4-(2,4-diaminophenoxy)-3,5-dimethylphenyl]propane Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and ensure consistent product quality suitable for industrial applications. The process begins with the preparation of the reaction mixture, where the dinitro precursor is suspended in a saturated fatty alcohol solvent along with the palladium on carbon catalyst. Operators must carefully monitor the temperature during the addition of hydrazine hydrate to maintain the optimal range of 70°C to 85°C, as deviations can impact reaction kinetics and safety. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance during scale-up operations.
- Prepare the reaction system by mixing the dinitro precursor with Pd/C catalyst and saturated fatty alcohol solvent.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution while maintaining temperature between 70°C and 85°C.
- Filter the hot solution, cool the mother liquor to precipitate crystals, and vacuum dry to obtain high-purity diamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis method offers tangible strategic advantages in terms of cost structure and operational reliability. By eliminating the need for high-pressure hydrogenation infrastructure, manufacturers can significantly reduce capital expenditure and maintenance costs associated with specialized reactor systems. The simplified workup process, which relies on standard filtration and crystallization rather than complex purification techniques, drastically shortens the production cycle time and reduces labor requirements. This efficiency gain allows for a more responsive supply chain capable of adapting to the dynamic demands of the electronic materials market without incurring prohibitive operational expenses. Furthermore, the use of readily available raw materials such as hydrazine hydrate and common fatty alcohols ensures a stable supply base, mitigating the risks associated with sourcing exotic or constrained reagents.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the simplification of the purification process lead to substantial cost savings in both capital investment and daily operations. By avoiding energy-intensive high-pressure conditions and reducing solvent usage through efficient crystallization, the overall manufacturing cost per kilogram is significantly optimized. This economic efficiency allows suppliers to offer more competitive pricing for high-purity electronic chemicals while maintaining healthy profit margins. Additionally, the reduced need for downstream processing minimizes waste disposal costs, further contributing to the overall financial viability of the production route.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available reagents such as hydrazine hydrate and saturated fatty alcohols ensures a robust and resilient supply chain. Unlike processes that depend on specialized catalysts or high-purity gases that may face supply constraints, this method utilizes materials that are easily sourced from multiple vendors. This diversification of the supply base reduces the risk of production interruptions due to raw material shortages, ensuring consistent delivery schedules for critical polyimide monomers. The operational simplicity also means that production can be easily transferred between facilities if necessary, enhancing overall supply continuity.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction conditions that are easily manageable in large-scale reactors without compromising safety or yield. The reduction in hazardous waste generation and the avoidance of high-pressure risks align with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing sites. This compliance advantage facilitates faster permitting and expansion of production capacity, enabling suppliers to meet growing market demand for sustainable electronic materials. The efficient use of resources and minimization of waste also support corporate sustainability goals, making this route attractive for environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this diamine monomer, based on the specific data and claims found in the patent documentation. These insights are intended to clarify the feasibility and advantages of the described method for potential partners and technical stakeholders. Understanding these details is crucial for making informed decisions about integrating this material into your supply chain or product development pipeline.
Q: What are the purity specifications for this polyimide monomer?
A: According to patent CN101265206A, the described method consistently achieves product purity exceeding 99%, with specific examples demonstrating purity levels up to 99.8% without requiring complex post-reaction purification steps.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process is explicitly designed for industrialization. It utilizes standard filtration and crystallization techniques, avoids high-pressure hydrogenation equipment, and uses readily available raw materials, ensuring robust commercial scale-up of complex electronic chemicals.
Q: How does this method improve environmental compliance?
A: The method significantly reduces the generation of three wastes compared to traditional reduction methods. The use of hydrazine hydrate in a closed system with simple filtration minimizes hazardous byproduct discharge, aligning with strict environmental regulations for specialty chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-bis[4-(2,4-diaminophenoxy)-3,5-dimethylphenyl]propane Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of polyimide monomer synthesis and is equipped to implement the advanced reduction techniques described in patent CN101265206A with precision. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for electronic applications. Our commitment to quality and process optimization ensures that our clients receive materials that consistently perform in high-end polymer formulations.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our specialized expertise. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our production capabilities can support your long-term strategic goals. Let us help you secure a reliable supply of high-performance monomers for your next generation of electronic materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
